Background
Information and Communication Technology (ICT) is a valuable tool in decreasing management costs and early identification of problems at farms [1]. In the case of Korea, the introduction of ICT was increased on the pig industry in recent years. ICT technology of Korea was applied in the basic areas such as temperature and humidity change, and fire detection in the farm, the real-time monitoring system in the feed bin in the swine industry [2]. On the other hands, the development and application of ICT were far behind compared to other countries. Recently, the number of holding female pigs per farm continues to increase due to continuous scale-up, meat consumption, and specialization while a total of farms has decreased. Thus, precise detection of estrus is one of the critical factors for the farm’s economy, but it requires a lot of labor and time consuming of the production system [3,4]. There is various detecting method of estrus. First of all, detection using boar is widely investigated by many previous researchers [5–7]. Especially, Bressers et al. [5] and Korthals. [8] reported that 75%–96% estrus cases were classified. Secondly, the back-pressure test (BPT) is to investigate the standing response sow when the back was pressed [9]. Thirdly, physical activity, vulva swelling and redding is a visible sign of the upcoming estrus in sow [10,11]. These methods are the most widely used up to now, but the method spends a lot of money to maintain boars such as feed cost and space for the rearing compared with other methods. Also, detection using physical, physiological traits and BPT are not objective because people judge them subjectively, consequently the ordinary people cannot clearly distinguish the onset of estrus compared with technicians. Therefore, this study was conducted to investigate the feasibility and quantification of standing time, vulva and body temperature (BT) as methods of estrus detection using ICT technology.
Materials and Methods
The Animal Care and Use Committee of Chungbuk National University approved all the experimental protocols used in the current study.
A total of eighty sows were used in an experimental period between March 2018 and February 2019 in Chungbuk National University research farm. Yorkshire × Landrace sows were of good health at the beginning of the experiment. They were housed in temperature and humidity controlled environment. Sows were individually fed two times and had ad libitum access to water.
The estrus cycle of sows was synchronized using a progesterone-al-trenogest solution (Regumate®; Roussel-Uclaf, París, France). They administered at a dosage of 5 mL to each sow daily for 18 days. Estrus synchronization\was performed in March 2018 and July 2018.
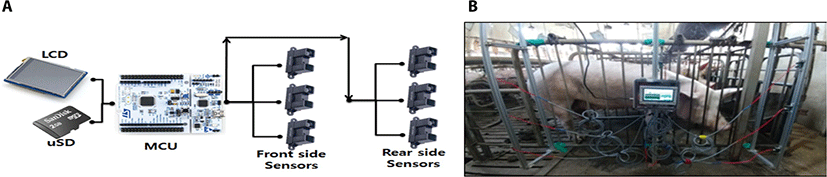
Ultrasonic sensor (GP2Y0A02YK0F, Sharp Inc, Japan) was connected to a micro-controller (NUCLEO F446, ST-LINK, Japan), so that the standing time of sow could be measured. Also, a small LCD module was built in the micro-controller to show the measurement content on the screen (Fig. 1A). Also, we used the C language to create the program in micro-controller. Ultrasonic sensor was installed on the front-end side according to height of the stall at 30 cm intervals (Fig. 1B). Standing time and number were measured into standing sensor in estrus synchronized sow, and collected 4 times in the experiment period. Data represented total standing time and total standing number of 24 hours and sanding number more than 10 minute in estrus and non-estrus period, respectively. Leg lesion of the sows were examined at the beginning and the ending experiment period. Leg lesion score were assigned when sows were in estrus and non-estrus period, resulting in a rating of 0 (no lesion), 1 (slight scratch) or 2 (erosion). The same individual recorded all scores.
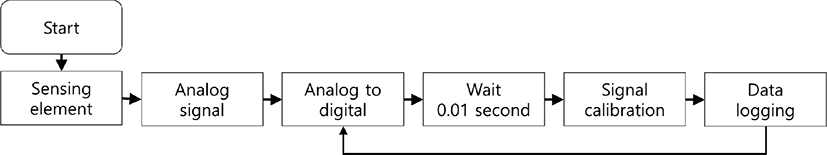
Each ultrasonic sensor produces an analog voltage signal from 0.0 V from 3.3 V corresponding to distance from a sow. The voltage signal indicates distance from 0.2 to 1.5 m through calibration curve provided by manufacturer. The microprocessor’s analog to digital conversion (ADC) function provides 12 bit of resolution and continuous measurement with 100 ns of conversion time in stable conversion, which could lead that the minimum distance resolution is 0.0003 m resulted from 1.3 m of physical property divided by 4,096 steps of signal decomposition. A fixed sampling frequency was determined 100 Hz (100 measurements in one second) to compare the periodic characteristics of sow’s movement. Data logging software was developed by a native C language and an executable application was compiled by gcc (GNU, version 5.4.0) in Linux operating system. The converted signal was recorded in a mass storage device with a common-separated values (CVS) format that measured date, time and distance values from sensor 1 to 6 in sequence. Total flow of signal processing is illustrated by Fig. 2.
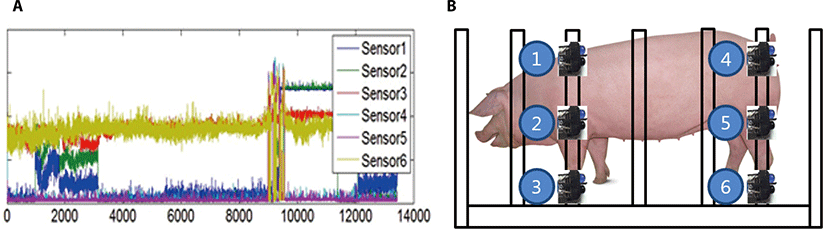
Based on the assumption that 100 Hz of measurement frequency is accurately performed, the dimension of logged data forms 8 by n (multiplied by 100 to elapsed seconds during measurement) matrix. Each distance value per every 0.01 second recorded from third column in the matrix was post processed to denoise exceptional fluctuations due to systematic error by calculating the mean of adjacent values. The recorded data was post-processed and visualized using MatlabTM (Mathworks, CA, USA). Data visualization was performed using a built-in plot function in the software. The plot of individual distance data from each sensor’s position was concurrently rendered for comparison between control groups in terms with variation and frequency in time domain with resolution of 0.01 second like Fig. 3A. The position of sensors is as illustrated by Fig. 3B. Thereafter, standing time, standing number and standing number over 10 minute were counted using this figure.
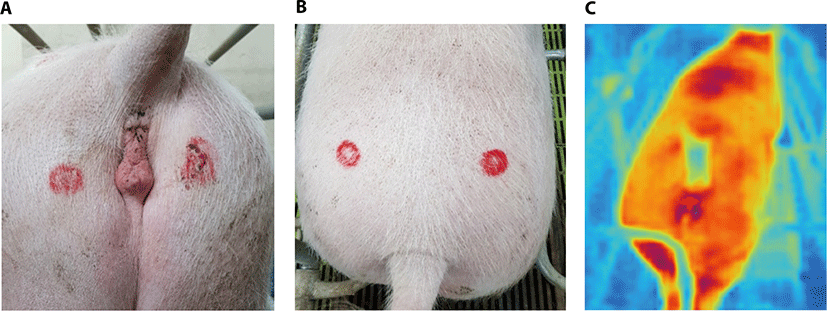
Vulvar temperature (VT) and BT were measured in estrus synchronized sows using the infrared thermography camera in estrus and non-estrus period at experiment 1 period. Each sow was monitored from a fixed distance of 1.9 m. The monitoring focused on the sow’s vulvar region or body. The camera focused on the vulvar area and top area of tail in the sows (Fig. 4). A red spot was marked on each vulva area (Fig. 4A) and the top of tail (Fig. 4B) of the female with the aim of taking measurements at the same location during each imaging event. When their measuring area was excessively dirty by feces, it was wiped using the paper towel to prevent a minor temperature change. Estrus VT measurement continued until no longer showing signs of estrus, and non-estrus VT measurement performed at normal days. The infrared images were downloaded into a computer and visualized using the Smart View™ Version 2.0 software (Fluke Thermography, Plymouth, USA).
A total of eighty (Landrace x Yorkshire) sows were consecutively used for this experiment in each treatment of twenty sows. They did not be synchronized. Estrus detection treatments were as follows: CON, visual evaluation; TRT1, standing time 110 or more minutes for 24 hours; TRT2, VT 31 °C or more; TRT3, BT 40 °C or more. The standards listed above were acquired from experiment 1. Sows were placed in an environmentally controlled farrowing facility. The temperature was maintained at a maximum of 22 °C. Sows were placed in farrowing crates (2.2 × 0.7 m) with an area (2.2 × 0.7 m) on either side of the crate for the piglets. When estrus was detected using these methods, sows were inseminated three times by 12 hours intervals using semen from Duroc (Darby Genetics Inc., Anseong, Korea). Artificial insemination (AI) was performed until the fertilization was finished. Mattress were put down for piglets to lie on before farrowing. The Lamp was dangled above the mattress to keep the temperature for newborn piglets constant at 35 °C. After the birth, individual pig weight was recorded at birth to determine litter birth weight. Also, individual numbers of piglets alive and death per litter were recorded until weaning to determine total number born and number of born alive per litter, respectively.
In experiment 1, data were analyzed using the General Linear models procedure of the SAS software (SAS Institute, 2006). Difference among the treatments were separated by a t-test. Variability in the data is expressed as the standard error and probability level of p < 0.05 was considered to be statistically significant. In experiment 2, all data were analyzed by ANOVA using the General Linear models procedure of the SAS software (SAS Institute, 2006). Difference among all treatments were separated by Ducan’s multiple range test. Variability in the data is expressed as the standard error and probability level of p < 0.05 was considered to be statistically significant.
Results
Recently, as interest in animal welfare has increased, pigs have been released from a variety of pain such as teeth clipping and tail docking, and have banned the usage of hormone. So far hormone associated with the growth was banned, but we think that usage of altrenogest also is banned in terms of animal welfare. Therefore, this study focused on finding new methods of estrus detection and whether quantified data are valuable.
These results of experiment showed that sows in estrus period had greater (p < 0.05) total standing time, total standing number and standing number 10 minute or more than sows in non-estrus period (Table 1). However, there was no significant difference (p > 0.05) on leg lesion among the treatments. For the temperature, sows in estrus period had greater (p < 0.05) vulva temperature than sows in non-estrus period (Table 2). Vulva temperature of sow in estrus increased by 4°C. Also, BT of sows tended (p = 0.06) to increase when they reached in estrus.
| Item1) | ES | NES | SE | p-value |
|---|---|---|---|---|
| Total standing time (m) | 129.15a | 103.44b | 4.07 | 0.03 |
| Total standing number (n) | 15.38a | 11.62b | 0.50 | 0.02 |
| Sanding number 10 minute or more | 6.75a | 3.00b | 0.48 | 0.03 |
| Leg lesion2) | 0.75 | 0.63 | 0.16 | 0.41 |
| Item | ES | NES | SE | p-value |
|---|---|---|---|---|
| Temperature | ||||
| Vulva | 34.95a | 29.55b | 1.65 | 0.03 |
| Body | 39.95 | 36.80 | 2.01 | 0.06 |
Table 3 showed the effect different estrus detection on number of AI and reproductive performance in sow. In the results of AI, standing time and vulva temperature tended (p = 0.06) to improve number of AI until fertilization was finished. However, no differences (p > 0.05) were observed on reproductive performance among treatments in sow.
| Item1) | CON | TRT1 | TRT2 | TRT3 | SE | p-value |
|---|---|---|---|---|---|---|
| Number (n) | ||||||
| Al | 2.17 | 1.53 | 1.42 | 1.93 | 0.32 | 0.06 |
| Performance | ||||||
| Litter birth weight (kg) | 16.13 | 16.83 | 16.53 | 16.67 | 0.39 | 0.24 |
| Total number born per litter (n) | 11.97 | 12.01 | 12.34 | 12.24 | 0.55 | 0.44 |
| Number of born alive per litter (n) | 11.47 | 11.42 | 11.58 | 11.47 | 0.24 | 0.38 |
| Mortality rate | 6.75 | 6.88 | 6.64 | 6.54 | 0.55 | 0.88 |
Discussion
There were no researches on the relationship standing behavior and onset of estrus in sow. Some researchers reported that standing reflex was observed at some sows in estrus, but it is behavior in response to the presence of a boar [12,13]. We hypothesized that standing reflex was observed due to their excitement, and change of standing behavior may be increased by this excitement. In the present study, results showed that time and number related with stand increased (p < 0.05) about 25%, 32%, and 125% when sows reached in estrus (Table 1). Estrogen level usually peaked as sows reached in estrus phase [14]. Many researchers showed that estrogen elevated cortisol level [15–17]. Cortisol is a steroid hormone, and it is released in response to stress. Ziegler. [18] reported that stress increased sympathetic nerve activities such as increasing the heart, respiratory rate and blood pressure. As a result, increasing of standing behavior was considered by increasing the sympathetic stimulation into estrogen when sows reached in estrus.
In the livestock industry, infrared thermal imaging has been used for measurement of VT to identify the fertility or estrus [3,19–22]. Increasing usage of Infrared temperature measurements is considered because it reduce the risk of spreading infection, since touching the subject is unnecessary [23]. Also, the reason why many researchers focused on VT the pig’s vulva is more noticeable than other livestock species and is not obstructed by the tail. In the present study, VT of sow in estrus increased (p < 0.05) about 20% compared with that of sow in non-estrus (Table 2). These results agree with previous researches that vulva temperature were greater at estrus than at non-estrus [3,24,25]. When sow reached in estrus, blood flow to the vulva increases via the internal pudendal artery due to the influence of increased estradiol form the developing follicles [26]. This increase in blood flow causes surface area temperature in the vulva [25]. Also, VT degree in this study was different from the temperature obtained above listed studies. These difference were considered by environmental conditions, such as temperature and air flow and obstacle in skin because they can affect the accuracy of thermal images [27]. However, there was no significant difference (p > 0.05) on BT among the estrus presence. No difference on BT is considered because sows had thick backfat depth.
Estrus detection using standing time and vulva temperature tended (p = 0.06) to improve number of AI until fertilization was finished while these detection did not affect reproductive performance in sow (Table 3). Visual evaluation was performed using redding and swelling vulva and back pressure test. During experimental period, we detected some sows in silent estrus. Soede et al. [28] showed that 18% of sows had an estrus duration of less than 24 h when estrus was detected by the back pressure test without a boar. Also, less intensive estrus detection caused the increasing incidence of silent estrus [29]. In experiment 2, boar and estrus synchronization were not used in terms of economic and welfare. Thus, new estrus detection using standing sensor and infrared Thermography can presumably be used for early detection with particular interest in animals with silent estrus in these conditions. Standing behaviors and vulva temperature increased by 25%, 32%, 125%, and 20% when sows reached in estrus, respectively. Therefore, new estrus detection are quantifiable when about standing behaviors and vulva temperature increased by 20% compared with non-estrus phase. Also, estrus detection using standing time and VT tended to improve number of AI until fertilization was finished. In conclusion, existing method be replaced to new method such as VT and standing time when detecting the estrus.
