INTRODUCTION
The epididymis is a critical organ in the male reproductive tract where immature spermatozoa go through several maturation processes and mature spermatozoa are stored before ejaculation. The epididymis is structured by a highly convoluted tubule and is separated into three distinct regions, namely, the caput, corpus, and cauda epididymis, based on their morphology, histology, physiology, and function [1,2]. The spermatozoon produced in the testis is transcriptionally and translationally silent and lacks fertility ability [3]. Therefore, this cell must undertake post-testicular maturation steps by traveling through the entire epididymal lumen, which is arranged with the pseudostratified columnar epithelium [4–9]. The epididymal epithelium comprises a few different cell types including principal cells (PCs), clear cells (CCs), narrow cells (NCs), and basal cells (BCs). Various proteins that are produced and released from these major cell types can regulate and create a special luminal condition that is attributed to sperm maturation and acts as a reservoir in the epididymis [10–14].
NCs and CCs express high levels of vacuolar ATPase (V-ATPase) in their apical membranes and are required for the formation of an acidic luminal pH that provides to sustain spermatozoa in a deactivated status during their maturation and storage in the epididymis. These two cell types are important for luminal acidification of the epididymis; yet, they are present in very different distributions along the epididymis. NCs are comparatively small in number and are situated only in the initial segments. CCs are widely distributed in the caput, corpus, and cauda epididymis and dramatically increase in number from the proximal to the distal regions [14,15]. The morphological characteristics of CCs differ depending on the regions of the epididymis where they are found. For example, CCs are bowl and cuboidal-shaped in the caput/corpus and cauda, respectively, in mice [16]. Recently, we reported that bowl-shaped CCs are only pertained in the epididymis of bats [17], whereas, in pigs, bowl-shaped CCs are only located in the caput regions and are absent in other regions of the epididymis [18]. Therefore, the distribution and localization of CCs show species-specific patterns. In addition, recent study has reported that a loss of CCs can lead to elevation of luminal pH in the epididymis and result in decreased sperm fertility [19]. BCs display a hemispherical morphology and are placed underneath other epithelial cells and are in contact with the basement membrane. Recently, however, it was observed that BCs appear in different morphological features in the epithelium of the epididymis and trachea. They can extend their long and narrow projections, cross tight junctions, and scan luminal environments in rodents [20,21]. In addition to luminal acidification, BCs are an indirect player in regulating luminal pH via communication with CCs in the rat epididymis [22]. Therefore, the interactions between these two cell types, CCs and BCs, is necessary to create an ideal luminal condition for maturation and storage processes of spermatozoa. However, limited knowledge exists regarding the regulation and establishment mechanisms of the optimal luminal environment in the epididymis of bulls. To improve our understanding of these mechanisms, more studies related to the localization and distribution of these epididymal epithelial cells are required.
Here, we examined the presence and distribution of the B1 subunit of V-ATPase (B1-VATPase) and cytokeratin 5 (KRT5) (also known as CC and BC markers, respectively) in the bovine epididymis. Determining the specific distribution of epididymal epithelial cells may help understand the regulatory system to create a perfect internal milieu for maturation and storage of spermatozoa in the bovine epididymis. As far as we know, this study is the first to apply immunohistochemical markers to investigate their expression and localization into epithelial cells and spermatozoa of the adult bovine epididymis.
MATERIALS AND METHODS
Five bulls between 26 and 28 months of age were used for this experiment. The epididymides were collected from a local slaughterhouse (Yesan, Chungnam, Korea). All animal procedures used in this study were approved by the Animal Care and Use Committee of the National Institute of Animal Science (NIAS 2018-290).
The bovine epididymides were dissected and immediately placed in 4% paraformaldehyde (4% PFA) for 48 h at room temperature (4% PFA was replaced with fresh fixative every 12 hours), followed by three × 15-min washes in PBS. The tissues were transferred to 30% sucrose solution in PBS until the tissues sink to the bottom of the containers. The tissues were then placed in OCT compound (Thermo Scientific, Rockford, IL, USA), embedded in molds, and frozen with dry ice. The tissues were cut at 10 µm using a Leica 3050S cryostat (Leica Biosystems, Wetzlar, Germany), placed onto Muto New Silane III coating slides (Muto Pure Chemical, Tokyo, Japan), and stored at −80°C until subsequent use. For histological observation, hematoxylin and eosin (H&E) staining was performed as described before [23].
The sections were thawed at room temperature for 10 min, and rehydrated in PBS for 10 min. To unmask antigens, the slides were placed in a microwave-resistant jar containing an alkaline buffer (Vector Laboratories, Burlingame, CA, USA) and microwaving for 2 min on high power (repeat three times with 5-min intervals between steps). After antigen retrieval, the slides were allowed to cool slowly to room temperature for at least 30 min. The slides were then washed three times with PBS and were incubated with the blocker solution (Thermo Scientific) to block non-specific antibody binding for 30 min at room temperature. The slides were then incubated with the primary antibodies. After some washing steps, secondary antibodies were applied. The slides were rinsed between antibody treatments with PBS containing 0.1% Tween-20 (Sigma-Aldrich, St. Louis, MO, USA). The incubation times of primary and secondary antibodies vary from 60 min at room temperature to overnight at 4°C. The slides then counterstained and mounted with Vectashield antifade mounting medium (Vector Laboratories) containing 4′,6-diamidino-2-phenylindole (DAPI). Coverslips were sealed with clear nail polish. To observe CCs and BCs, the slides were stained with a chicken polyclonal antibody against the B1-VATPase (diluted 1:1,500) [18] and a rabbit polyclonal antibody against the KRT5 (diluted 1:300; Thermo Scientific) [21], respectively. For the co-localization studies, primary and secondary antibodies were applied sequentially, as described above. The following all secondary antibodies used in this experiment were purchased from Jackson ImmunoResearch Laboratories (West Grove, PA, USA): Cy3-conjugated donkey anti-chicken IgG (1:600) and Alex Fluor®488-conjugated donkey anti-rabbit IgG (1:300). Dako antibody diluent (DAKO, Carpinteria, CA, USA) was used for dilution of primary and secondary antibodies. For negative control, the same staining protocol without the addition of primary antibodies was performed. The slides were observed under an LSM800 confocal microscope (Carl Zeiss, Jena, Germany) and acquired confocal images were analyzed using Zen Blue (Lite) software.
B1-VATPase and KRT5 antibodies were used to perform immunofluorescence staining of CCs and BCs, respectively. At least three epididymides obtained from different individual males were examined. The numbers of CCs and BCs were measured by manually counting the number of B1-VATPase- and KRT5-positive cells per square millimeter epithelium area. To measure B1-VATPase-positive BCs, double labeled epithelial cells were also examined as described above. Zeiss confocal microscope was used to acquire digital images (20× objective).
Tissues were lysed for 30 min on ice in RIPA buffer (Thermo Scientific) containing complete protease inhibitors and phosphatase inhibitors (Roche Applied Science, Indianapolis, IN, USA). The lysate was centrifuged at 16,000×g for 30 min at 4°C and the supernatant was collected. For electrophoresis, samples were prepared in LDS Sample Buffer (Invitrogen) with 4% b-mercaptoethanol and incubated for 30 min at room temperature. The protein was loaded on LDS polyacrylamide gel and electrophoresed as described before [24]. The separated proteins were transferred onto Immun-Blot polyvinylidene difluoride (PVDF) membranes. Antibodies against B1-VATPase (diluted 1:2,500), and KRT5 (diluted 1:1,000) were used and the labelled antigens were detected by chemiluminescence and Kodak imaging films.
The epididymides from at least three bulls were used for all experiments, and for each epididymis, three sections were examined. The numbers of CCs and BCs were analyzed using one-way analysis of variance (ANOVA) test and the significant difference between means of analyzed data was analyzed using Duncan’s multiple range test. A significant difference was indicated by p < 0.01. Measurements of the epithelial area were performed by using Zen Blue (Lite) software.
RESULTS
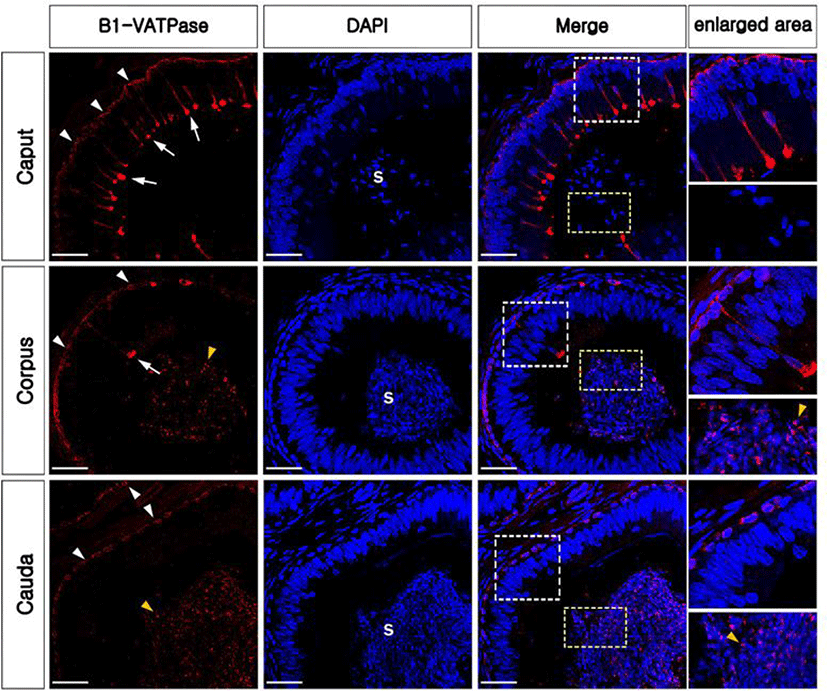
In the present study, the epididymis of bulls was labeled to examine the presence and localization of B1-VATPase. As shown in Fig. 1, B1-VATPase was expressed and localized in two different cell types, with both cell types lying on the epithelium. Epithelial cells with nuclei positioned close to the lumen were observed with a narrow-shaped morphological characteristic (Fig. 1, arrows) and the other type of epithelial cells was located beneath the basal lamina where BCs were commonly present (Fig. 1, arrowheads). Specialized cells with high expression of V-ATPase in their apical membrane and the nucleus adjacent to the luminal aspect are classical characteristics of epididymal CCs in many species, e.g., mice, rats, pigs, and bats. Therefore, we called these specialized cells narrow-shaped CCs. These narrow-shaped CCs were present from the caput to the corpus; however, they were disappeared in the cauda regions of the epididymis (Fig. 1). The number of cells was much less in the corpus than that in the caput (Fig. 2A). On the other hand, V-ATPase positive cells located in the lower parts of the epithelium were observed in all epididymal regions from the caput and cauda (Fig. 1, arrowheads). The number of V-ATPase positive cells located in the lower parts of the epithelium was highest in the corpus, followed by the caput and cauda epididymal segments (Fig. 2B). V-ATPase-positive spermatozoa were observed in the corpus and cauda (Fig. 1, yellow arrowheads) but not in the caput epididymis (Fig. 1). No labeling was detected from negative controls (Supplementary Fig. S1A and D). Western blotting confirmed the expression of B1-VATPase in the bovine epididymis (Supplementary Fig. 1G).
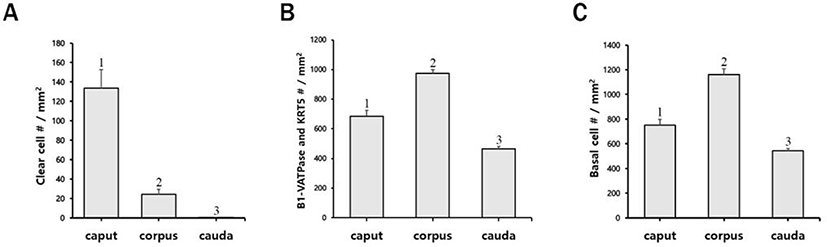
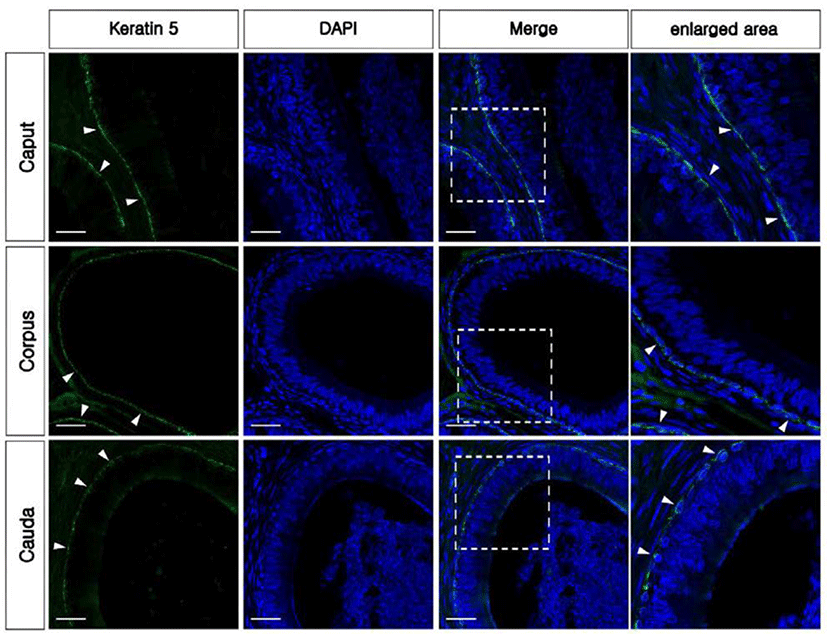
The KRT5 antibody was used as a marker of BCs in the epididymis. KRT5 positive cells were present in the base of the epithelium and had a dome-shaped morphology from all regions of the epididymis (Fig. 3). No axopodia cells displaying a long cytoplasmic projection were present in any regions of the epididymis. No KRT5 expressed spermatozoa were detected in the epididymis. The largest number of BCs was observed in the corpus, followed by the caput and cauda (Fig. 2C). No labeling was detected from negative controls (Supplementary Fig. 1B and E). Western blotting confirmed the expression of KRT5 in the bovine epididymis (Supplementary Fig. 1G).
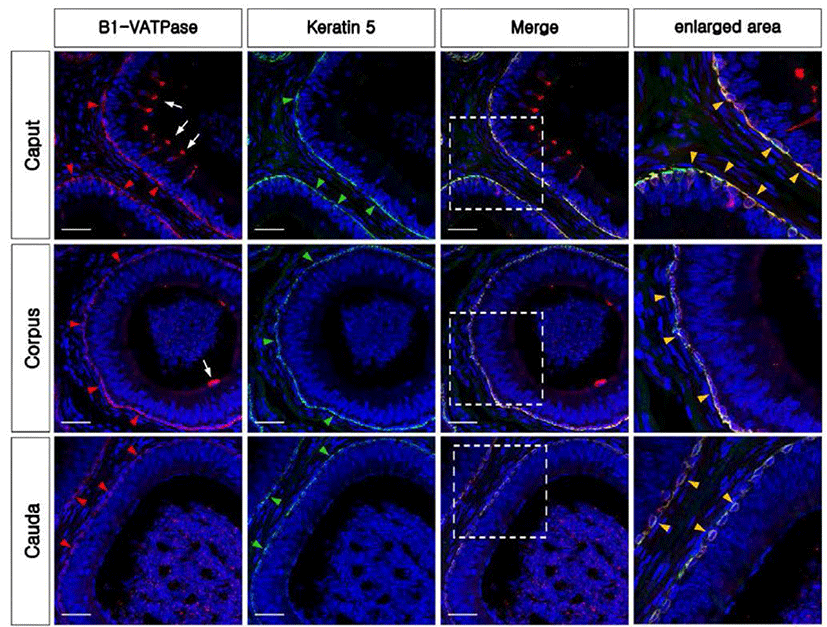
To confirm whether V-ATPase was expressed in the BCs, B1-VATPase and KRT5 were double-labeled in the bovine epididymis. B1-VATPase was clearly expressed in two different localizations in both the caput and corpus (Fig. 4, white arrows and red arrowheads), whereas KRT5 was only present at the lower part of the epithelium from all regions of the epididymis (Fig. 4, green arrowheads). Epithelial Cells co-labeled with B1-VATPase and KRT5 antibodies were detected in the lower part of the epithelium from the caput to the cauda regions of the epididymis (Fig. 4, yellow arrowheads). Only B1-VATPase positive cells (narrow-shaped CCs) were detected in the caput and corpus (Fig. 4G and H, arrows). Thus, V-ATPase was expressed in BCs as well as in CCs in the bovine epididymis. No labeling was detected from negative controls (Supplementary Fig. 1C, and F). Histological analysis revealed that the epididymal epithelium is pseudostratified and contains CCs and BCs with different locations (Supplementary Fig. S1H-M).
DISCUSSION
V-ATPase and KRT5 have been used as CC and BC markers, respectively, in many species including mice, rats, pigs, and bats. We first observed the expression and localization of CCs and BCs using B1-VATPase and KRT5 antibodies in the adult bovine epididymis. BCs were maintained in all portions of the epididymis and had a dome-shaped morphology. This result is in agreement with earlier researches in pigs and bats that do not have any projected BCs but have only dome-shaped BCs [17,18]. In rodents, however, the BCs were present with region-specific morphological characteristics. For example, the observation of BCs with long and narrow projections were confirmed in the initial segment and distal corpus/proximal cauda in mice [21] and rats [22], respectively. Thus, the morphological characteristics of the epididymal BCs are species-specific. In this study, we suggested that CCs were observed with a narrow-shaped morphology from the caput to the corpus; however, they were not present in the cauda region of the epididymis. Earlier studies have reported that CCs are localized differently depending on the species. In pigs, CCs were only localized in the caput region [18], but CCs in bats have been observed in all regions of the epididymis [17]. Regarding the morphological features of CCs, bowl-shaped CCs were observed in pigs and bats [17, 18]; however, in mice, the CCs were shown with various morphologies such as narrow-, bowl-, and cuboidal-shaped morphologies in their regions of the epididymis [15]. Thus, CCs have species/region-specific distribution and morphology. In the present study, both B1-VATPase and KRT5 were co-expressed in the BCs of the adult bovine epididymis. This observation is consistent with our goat study where B1-VATPase and KRT5 were co-localized in adult goat epididymis but not before puberty [25]. According to previous studies, however, B1-VATPase and KRT5 were not co-expressed and co-localized in the epididymal epithelium in pigs, mice, rats, and bats [15,17,18,26]. Therefore, these co-localization patterns of V-ATPase and KRT5 in BCs could be specific to ruminant animals.
The V-ATPase is confined to the apical membranes of CCs of the epididymis and is a major contributor to luminal acidification by secreting protons into the epididymal luminal. Recent studies on the epididymis have shown an elaborate communication network exists between CCs and BCs, and between CCs and PCs, to establish and maintain luminal acidification that is necessary for sperm maturation and storage [22,27]. The pH in bovine cauda epididymis was reported to be approximately 6.8 by Wales et al. [28] and approximately 5.8 as reported by Acott and Carr [29]. These acidic conditions in the epididymis could be controlled by epithelial cells and other cells. No CCs were present in the cauda epididymis in the present study; rather, BCs expressed B1-VATPase. However, we still have insufficient data to interpret the acidification mechanism of the bovine cauda epididymis. The mechanism could be complicatedly regulated by many factors. We speculate that in the bovine epididymis, there are two hypotheses for maintaining the acidic luminal condition: 1) V-ATPase-positive BCs might directly secrete protons into the lumen and 2) PCs might secrete less HCO3− to alleviate luminal pH in the bovine epididymis. Additional researches are required to elucidate the fundamental mechanisms for the establishment and regulation of luminal acidification in the bovine epididymis.
Here, we report that V-ATPase was highly expressed in the cauda spermatozoa but less expressed in the caput spermatozoa. These results are in agreement with a previous study that the a2 isoform of V-ATPase (ATP6V0A2) was highly expressed in normal human spermatozoa (motile) but was not detected in spermatozoa (immotile) from infertile men [30]. The epididymal transit from the caput to the cauda is necessary for sperm maturation, the acquisition of motility, and the ability to fertilize eggs. Therefore, V-ATPase in bovine spermatozoa may be a vital molecule for controlling sperm maturation and could be used as a biomarker candidate for the evaluation of bull infertility in the AI center.
In conclusion, B1-VATPase is expressed in CCs, BCs, and cauda spermatozoa in the bovine epididymis. These highly specific expression patterns in the bovine epididymis might be important for the establishment and maintenance of an ideal internal environment for the maturation and storage of spermatozoa. However, to identify a novel function of V-ATPase to regulate the unique luminal environment in the bovine epididymis, it is essential to increase our understanding of the regulatory mechanism by systemic cell-cell communications and intense cell-sperm interactions.
SUPPLEMENTARY MATERIALS
Supplementary Materials Are Only available online from: https://doi.org/10.5187/jast.2021.e32.
