INTRODUCTION
Age at first calving (AFC) is a period when a cow reaches its maturity and reproduces for the first time. AFC determines the reproductive capacity of cows, i.e., how early they are able to conceive, gestate, and calve. The primary determinant of pregnancy success of a heifer is age at puberty. Subsequently, this affects her ability to rebreed in subsequent years, remain in the herd, and her lifetime productivity. The sexual maturity of a cow considerably depends on body weight [1] and fat percentage [2]. In this study, we focused on the interrelationship between AFC, conformation traits, and calving interval (CI).
AFC can be manipulated by altering growth rates. Growth is affected by genetic potential, nutrition, and environmental conditions. The relationship between body development, subsequent reproduction, and ability to produce milk is important for the final economic evaluation of a reduced rearing period [3]. One strategy to reduce the cost of milk production is to shorten the rearing period. Thus, heifers can calve when they are not older than 24 months and have adequate body size.
Hanwoo cows have been raised for labor for thousands of years; however, in recent decades, they have been bred as beef cattle, and their productivity of beef became important [4]. Delayed first calving is a major loss in replacement cost of Korean beef farms under the circumstance of absence of seasonal breeding. However, early calving may make heifers experience precocious puberty. When they become pregnant at this time, they usually calve late or have smaller body size than heifers calving at the proper time; in addition, they have a greater propensity for dystocia, calf death at calving, and an extended postpartum recovery period [5,6]. The physiological endpoint for first calving is affected by factors related to the management of farm as well as the physiology and genetics of the female. The objective of this study is to investigate the genetic relationship between AFC, body development, and reproduction capacity in female Hanwoo.
MATERIALS AND METHODS
All Hanwoo producers voluntarily inform Korea Animal Improvement Association (KAIA) and Livestock Cooperative Federation certain details related to calving events including the date of birth and pedigree information of Hanwoo cows. In addition, KAIA legally records conformation traits of Hanwoo cows. This information is held in the national database. All abovementioned information was downloaded from the pre-existing national database for the purpose of this study. Therefore, it was not necessary to obtain animal ethics committee approval before conducting this study.
Conformation and reproduction traits of 19,565 Hanwoo cows (born between 1997 and 2017, affiliated to 18 local Livestock Cooperative Federations [LCFs] participating in the Beef Cattle Breeding Program of Korea Animal Improvement Association, Seoul Korea) were used for the analysis. In the analysis, conformation traits were the body condition score (BCS), height (HT), body width (BW), hip bone length (HL), hip bone width (HW), and final score (FS). The FS of a cow was based on the six major classification categories such as the general appearance, soundness, front, middle and rear section of body, and feet and legs. The scores from each category are based on the level of desirability exhibited by individual cows compared to the ideal model having a total of 100 point. The descriptions and scores of type traits are shown in Table 1, and the range of FS is from 68.2 to 87.1.
The 52,299 calving records consisted of AFC and CI of cows mated through artificial insemination and weaned at approximately three months after delivery. Calving records from 2011 to 2019 were collected, and calving occurred throughout the year. AFC was determined by the birth date of the heifer and the date of its first calving, while CI was computed using the date of the previous calving and the current calving date. AFC and CI were recorded in months and days, respectively. The records of AFC with the range from 16 to 32 months and the records of CI with the range from 300 to 600 days were included in the analysis. The structure of data is shown in Table 2.
Identifiable environmental factors were initially analyzed to determine whether they should be included in the model by simultaneously considering numbers in contemporary groups. Basic analysis was performed by the SAS software 9.4 version (SAS Institute, Cary, NC, USA). For genetic analysis, the Wombat [7] program for Linux OS was used.
The mixed animal model with the assumptions of expectations and variances was
where y is the vector of observations for each trait; b is the vector of fixed effects; a is the vector of random additive genetic effects of the animals; p is the vector of random permanent environmental effects only for the repeated CIs through parities; e is the vector of random residual effects associated with the observations. Meanwhile, X, and Z are incidence matrices related to fixed, additive genetic, and permanent environmental effects, respectively. G includes the relationship matrix of individuals and additive variance of the traits. P and R are the identity matrix with permanent environmental and residual variance, respectively. For the traits of conformation, the fixed effects were the birth year (2002–2017), region-classification year (107 levels, classification year 2014–2019), and classifier (4 levels). A total of 23 classifiers rated type traits, of which only 3 had a significant number of type evaluations. The rest of them were considered to be 1. The conformation traits of Hanwoo cows at the age of 12.6–250.0 months were recorded along with calving. The average age month at measurement was 57.5; however, the highest frequency was observed at 39 months. Age month was treated as the second polynomial covariate in the model for type analysis. The fixed effects of birth year (2002–2016) and region-calving year (225 levels, calving year 2014–2019) were included in the model for the reproduction traits; in addition, parity (8 levels) was a fixed effect for repeated records of CI. The models for both conformation and reproduction traits secured proper numbers in contemporary groups.
RESULTS AND DISCUSSION
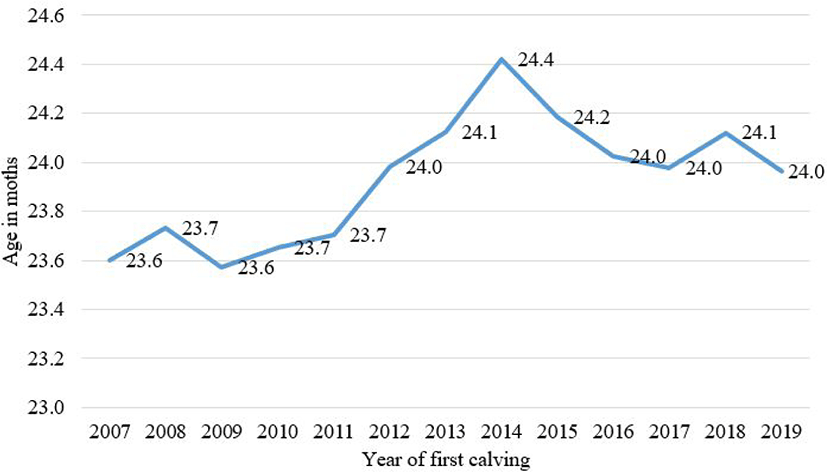
Fig. 1 shows AFC trends for Hanwoo. Between the years of approximately 2007 to 2014, there was an increase in the average AFC. This was likely associated with an industry-wide oversupply of cows, which led producers to wait longer to calve heifers. However, since this time, the average AFC has been decreasing. From 2014 to 2019, the average first calving age has decreased from 0.4 months to the average of 24.0 months in 2019.
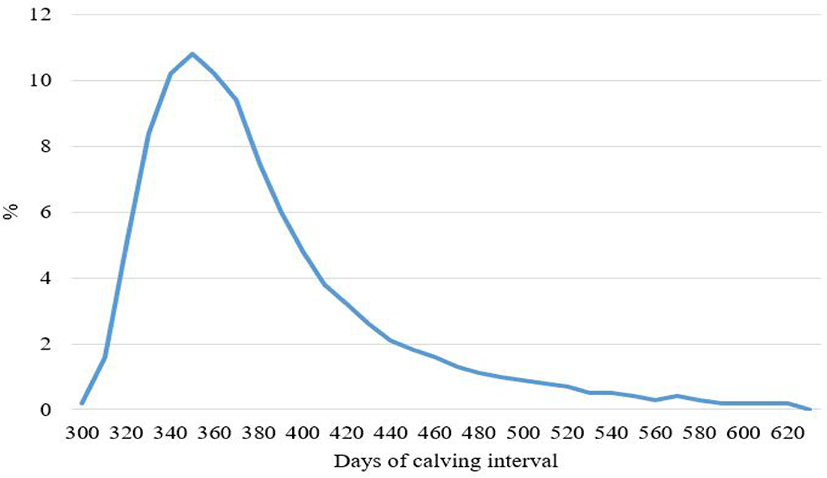
Fig. 2 shows the distribution of AFC for Hanwoo. The average and standard deviation is 24.1 and 2.3 months (Table 2), respectively, which is lower than 24.7 and 25.1 age month obtained by Eum et al. [8] and Lopez et al. [9]. The AFC in Hanwoo heifers seems to be lower than that of most beef cattle breeds [10–12], which ranges from 28 to 34 months old with seasonal breeding.
The two groups, determined from the age of 24 months at first calving, had approximately the same rate (50%) and frequency of age month and showed a peak at 24 month, as shown in Fig. 2. Analysis through grouping assumed that genetic factors affecting AFC could vary from group to group, and that all datasets had normal distribution.
Variation in the old group was greater than that in the young group, as shown in Fig. 2. AFC encompasses puberty and ability to conceive, gestate, and deliver a calf. The expression of AFC is limited by the breeding season in the countries using pasture for beef cattle. However, there is no grazing land in Korea; therefore, Hanwoo cattle are reared in the barn and bred using artificial insemination throughout the year [13,14]. It is speculated that the factors affecting young and old groups are different and are shown in Fig. 2. Young calving is mostly affected by physiology, genetics, and management at puberty, while old calving is affected by pregnancy. The cows reaching sexual maturity earlier than others may result in young calving. If sexual maturity is reached at approximately the age of 24 months, then the majority of calving after that age may have problems with pregnancy, and there may be more than one breeding attempt owing to impaired fertility or poor management.
The heritability of AFC in the young group was higher than AFC in the old group. It was 0.716 and 0.087, respectively (Table 3). The heritability of AFC in the old group was similar to or slightly lower than that of other reproduction traits such as CI (Table 4); however, AFC with the dataset before 24 months was highly heritable. This result indicated that the nature of young and old AFC is different, as has been previously described. Specifically, young AFCs are due to high levels of inheritance, and old AFCs have the same genetic patterns as low heritability of other reproductive traits. Similar results have been reported in beef cattle with high heritability for sexual maturity and AFC [15–17]. Moreover, the abovementioned studies have suggested to select for precocious maturity to calve at 24 months of age.
Conformation traits in the analysis were the BCS, HT, BW, HL, HW, and FS. The mean and standard deviation of age at classified date was 25.4 ± 13.6 months, ranging from 12.6 to 250.0 months of age. The classification started in 2014 and ended in 2019. BCS is used to estimate energy reserves in the form of fat and muscle of beef cows. BCS ranges from 1 to 9, with a score of 1 being extremely thin and 9 being very obese. HT is the vertical distance score from the apex of shoulder to the ground. BW is measure at the front leg, middle, and rear leg. The scores of conformation traits were multiplied by 10 for the analysis, except for FS (Table 2), and the scores were assumed to have a normal distribution in the analysis. This multiplied score was used to increase the amount of variance when estimating the genetic parameter to facilitate convergence during repetitive estimation. After estimating the genetic parameters, the breeding value was reduced to the original scale by dividing 10.
The calving age of primiparous cows affects their body development and reproduction capacity during their lifetime. Positive genetic correlation between sexual maturity and body weight in young and adult ages of the animals has been reported [18–20]. Furthermore, selection for high body weight at young age can improve sexual maturity and reproductive performance of cows owing to the favorable genetic association with these traits [21]. The ages of onset of puberty, breeding, and sexual maturity heavily rely on the body development of cattle [22].
The conformation traits of Hanwoo cows at the age of 13 month to 250 month were recorded along with AFC. In the model, age in month was treated as the second polynomial covariate to remove age effects from body development and to investigate genetic relationship of first calving age with conformation traits. Genetic analysis (Table 5) was performed using conformation data separated before and after 24 months of AFC, as shown in Fig. 2.
In the phenotypic correlation of AFC with body morphology (Table 5), the conformation data sets with young and old AFC did not show a significant difference (ranged from −0.038 to 0.001) but their genetic correlations showed large differences (ranged from −0.778 to 0.261). The genetic correlation of AFC with BCS in the dataset with young AFC was −0.778 but only −0.149 with old AFC. This result suggests that low BCS leads to early maturity at young age of heifers and reproductive disorders at old age of heifers. Young AFC showed a positive genetic correlation (0.442) with HT, but old AFC showed a high negative correlation (−0.826). This result shows that HT was increased along with the calving age for young heifers but HT appeared in reverse for old heifers. Old AFC BW showed a negative genetic correlation (−0.600 and −0.706) in both young and old AFC. BW was measured at front, middle, and rear legs, and acted like BCS.
In general, high BCS, and BW indicate an increase in body weight. Consequently, high body weight accelerates sexual maturity and reduces AFC, thus showing negative correlations. Heifers that are bred later have an increased incidence of dystocia at parturition [23]. In old AFC, it can be understood that high HT ease the reproductive problems, but in the young AFC, it can be interpreted that the higher the HT, the slower the sexual maturity. The result of young AFC is not clear.
HL and width showed a low positive correlation (0.010) in young AFC but a high negative genetic correlation (−0.623) in old AFC. Long hip bone has a genetic effect of delaying the calving age in the case of calving after 24 months of age. HW showed a similar tendency to HT and HL, and genetic correlation was 0.261 and −0.456 in young and old AFC, respectively. FS showed a low genetic correlation (−0.015) at young AFC but a high negative genetic correlation (−0.675) at old AFC. In the entire dataset, BCS, BW, and FS showed median values of genetic correlation between young and old AFC; HT showed a value that was close to that of old AFC; HL showed a positive correlation, which was higher than that for young AFC; HW showed a higher negative correlation than that of old AFC.
The obtained results demonstrate that the genetic effects of body type are different for young and old AFC. Young AFC is dominated by the sex maturation gene, and old AFC is dominated by the pregnancy-related gene. The genes related to sexual maturity are less affected by the environment, while the genes related to pregnancy are significantly affected by the effects of the environment. For example, the non-genetic effects for pregnancy (e.g., effects of artificial insemination agents and appropriate breeding time for fertilization) may not be trivial. The high heritability of young AFC and the low heritability of old AFC dataset support these facts.
Based on these results, high BCS and BW and low HT, HL, and HW were determined to be beneficial for sexual maturation. Lower values of all conformation traits increase AFC for the aged heifers. BCS, BW, and HT are closely related to body weight. Except for the HT of heifers, these results are consistent with the results of positive genetic correlation between sexual maturity and body weight in young and adult ages of the animals [18–20]. Moreover, selection for high body weight at young age can improve sexual maturity and reproductive performance of the cows owing to favorable genetic association with these traits [21]. However, because the standard errors of these correlation estimates are large, it is appropriate to accept these results as trend values.
CI is defined as the period of time elapsing between two consecutive parturitions. CI comprises the interval from calving to first heat (postpartum interval), number and length of services of the cow until pregnancy (conception length) which are often referred as open period in dairy cattle, and the length of gestation.
Normal postpartum interval of dairy cows has been reported to be 32.1 days for the experimental herd [24]. After an approximately 30-d period of uterine repair and involution, a normal postpartum ovarian cycle is resumed in beef cows [25]. On average, the conception length in Hanwoo is 69.52 ± 9.20 days [26], and the average gestation length is 285.35 ± 6.13 days. However, most variation in CI is due to the interval between the first service and conception, which may be greatly affected by environmental factors rather than genetic effects. Miscarriage or stillbirth is also a major factor when abnormally extending CIs.
Fig. 3 shows the distribution of CIs, ranging from 300 to 620 days, with the mean and standard deviation of 399.0 and 101.1 days (Table 2). Cho et al. [27] have reported similar values for the mean of CIs, while Eum et al. [8] and Lopez et al. [9] have reported much lower values of 360.8 and 363.1 days in Hanwoo.
The most important factor that affects the heifer rearing cost is AFC. Furthermore, AFC affects the lifetime productivity of dairy [28] and beef cattle [29–31]. CI is a complex trait that is affected by several reproductive traits. Therefore it is an effective indicator for monitoring the reproductive capacity of cows. Moreover, CI is economically important in Hanwoo beef production. Statistic Korea [32] has reported that a delay in pregnancy by 1 month causes a 350 USD loss, which covers feed and opportunity cost.
The heritability of CI and the correlation coefficients between CI and AFC are shown in Table 4. The CIs of cows were recorded along with AFC. Genetic analysis (Table 4) was performed using the CI data separated before and after 24 months of AFC, as shown in Fig. 2. In the two data sets, the heritability of CI was not significantly different but the heritability of the data set of old AFC was slightly higher (Table 4). In this study, the heritability of CI is higher than the values reported by Lopez et al. [11] and Oyama et al. [33], which are 0.03 and 0.05, respectively.
The genetic correlations between CI and AFC were higher than phenotype correlations in all datasets (Table 4). The phenotype and genetic correlation of AFC with CI were positive in the old AFC data, but negative in the young AFC data. Selecting for both young and old AFC groups towards the age of 24 months suggests that CI may decrease. When AFC approaches 24 months, CI decreases, which indicates that premature calving and delaying of first calving are not helpful in reducing CI. Thus, it is important to have calving of primiparous cows at an appropriate time. Of note, these results appear not only in phenotypes but also as genetic factors.
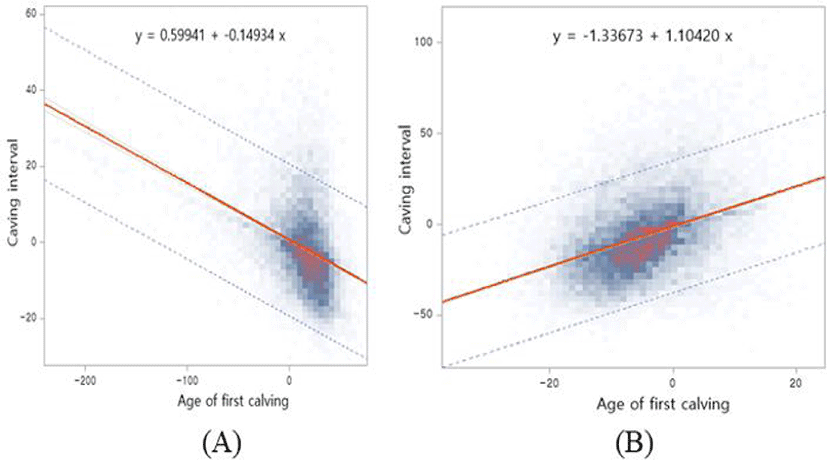
The breeding values of AFC and CI in the two groups (A: young AFC and B: old AFC) are shown in Fig. 4. The color in Fig. 2 shows the degree of density from grey to red. The negative and positive genetic correlations (−0.116 and +0.307) in the two groups are shown in Table 4, and the regression coefficients of both groups have the same negative and positives signs (−0.149 and +1.104). Fig. 4 shows the same genetic relationship between CI and AFC, as shown in Table 5. The R-squared values of each regression equation are 0.128 and 0.154 for A and B, as shown in Fig. 4. When cows were selected based on AFC, the corresponding genetic response of the A group showed a 0.149-day decrease in CI. However, the old group (B) showed an increase in CI by 1.104 days.
CONCLUSION
The production capacity of beef cattle is closely related to the reproductive capacity. Because the age of first birth is genetically related to the growth and reproduction of Hanwoo cattle, it is important to evaluate the proper genetic ability for AFC and use it for selection. Proper AFC may vary from individual to individual. However, because selection is performed for a population, it is necessary to set an appropriate time for AFC of a specific breed or population. Because the genetic characteristics before this point and the genetic characteristics after this point are different, it is necessary to select them separately. Proper genetic evaluation methods should be developed by considering the genetic nature of AFC.