INTRODUCTION
Influenza A viruses are negative-sense, single-stranded RNA (-ssRNA) viruses belonging to theOrthomyxoviridae family [1]. Influenza A virus infections are the only type of infection in birds [2]. Severe outbreaks of highly pathogenic avian influenza viruses (HPAIVs) have been reported worldwide from 2004 to the present, causing tremendous damage to the poultry industry [3-5]. The HPAI H5N1 virus is a fatal zoonotic disease that occurs in humans and has a mortality rate of approximately 53% [6]. Hemagglutinin (HA) protein binds to the cell and triggers endocytosis to enter the host cell [7]. Moreover, influenza A virus enters the nucleus to replicate the virus [8]. The HPAI H5N1 virus usually infects the trachea, in addition to other organs, especially the lungs, which are a major site of H5N1 replication in chickens [9,10].
The Vietnamese indigenous Ri chicken, which was used as an experimental animal in this study, is a yellow-fathered Vietnamese poultry [11]. HPAIV-resistant and susceptible lines belonging to the Ri chicken were distinguished by the genotypes of the Mx dynamin-like GTPase (Mx) gene and the BF2 gene, a major histocompatibility complex (MHC) class 1 molecule. Specifically, a substitution at the nucleotide 2032 (amino acid replacement at position 631) of the Mx gene allele, from A to G (amino acid serine to asparagine), demonstrated that chickens with allele A (Asn) have antiviral activity, i.e., they are HPAIV-resistant, and chickens with allele G (Ser) lack antiviral activity, i.e., they are HPAIV-susceptible [12,13]. MHC is a group of genes encoding different structures and functions [14]. Chickens containing the BF2-B21 haplotype have a high survival rate and those containing the BF2-B13 haplotype have a low survival rate against H5N1 avian influenza virus infection [15].
MicroRNAs (miRNAs) are non-coding endogenous RNAs approximately 22–24 nucleotides in size. miRNAs play various roles, one of which is to regulate gene expression [16]. They function as key regulators of various physiological and cellular activities and immune processes such as immune cell development, differentiation, and activation [17]. Moreover, miRNAs play crucial roles in the immune response of chickens to avian viral infections, such as leukemia, Marek’s disease, and infectious bursal disease [18].
A previous study investigated miRNA expression in the thymus, spleen, and bursa of Fabricius of H5N1-infected ducks and White Leghorns via high-throughput RNA sequencing to explore the disparate immunity between ducks and chickens [19]. Moreover, gga-miR-133c, gga-miR-1710, and gga-miR-146c target the PB1,PB1-F2, and N40 genes in H5N1-infected chicken lungs [20]. In our previous studies, RNA sequencing revealed immune-related genes involved in cytokine-cytokine interactions and mitogen-activated protein kinase (MAPK) signaling pathways in the lung and tracheal tissues of H5N1-infected Vietnamese indigenous Ri chickens [21,22]. Various cytokines and chemokines are induced by influenza A virus infection and some cytokines are essential for antiviral activity [23]. HPAI virus plays important role in MAPK signaling pathway by modulating MAPKs that have crucial role in innate and adaptive immune response [24].
Although there are studies on the immune function of HPAIV-infected chickens, including miRNA profiling studies, study on miRNA expression profiles between HPAIV-resistant and susceptible lines does not exist. In the present study, we compared differentially expressed (DE) miRNAs in Vietnamese indigenous Ri-resistant and susceptible lines against H5N1 virus infection. In this study, we revealed miRNA expression patterns and the potential of miRNAs that modulate the immune system through the regulation of candidate immune genes in resistant and susceptible lines of chickens infected with the HPAI H5N1 virus.
MATERIALS AND METHODS
Twenty specific-pathogen-free (SPF) chickens belonging to 10 resistant and susceptible lines were used in this study (Table 1). The Mx and BF2 genes were used to differentiate between the resistant and susceptible lines. High-resolution melting analysis confirmed the Mx gene genotyping results (Supplementary Fig. S1). The Mx gene, which has an adenine (A) at nucleotide 2032, was genotyped as the resistant line of Ri chicken, while the presence of guanidine (G) at this position was genotyped as the susceptible line. Based on BF2 genotyping, chickens possessing the B21 haplotype were determined to be resistant, and individuals possessing the B13 haplotype were identified as being susceptible. Thus, susceptible line chickens have the Mx (G)/B13 haplotype and resistant chickens have the Mx (A)/B21 haplotype. Ten 4-week-old Ri chickens (five resistant and five susceptible) were inoculated intranasally with 200 µL of 104 50% egg infectious dose (EID50) of A/duck/Vietnam/QB1207/2012 (H5N1), following OIE instructions [25]. Chickens were observed for symptoms of disease daily after infection with the H5N1 influenza virus. All chicken management and experiments were conducted in the Department of Biochemistry and Immunology at the National Institute of Veterinary Research (NIVR), Vietnam (TCVN 8402:2010/TCVN 8400-26:2014).
Lung tissues were collected on day 1 and day 3 from Ri chickens, following the WHO Manual on Animal Influenza Diagnosis and Surveillance. The chickens were euthanized after 1-day and 3-day virus infection. All sterilized lung samples were crushed and completely homogenized by cryogenic grinding in liquid nitrogen. RNAs were extracted from the lung tissue using TRIzol reagent (Invitrogen, Carlsbad, CA, USA) follow by the manufacturer’s guidelines. Isolated total RNA was checked for quality using Trinean Dropsense96 (Trinean, Gentbrugge, Belgium) and a Bioanalyzer RNA Chip (Agilent Technologies, Santa Clara, CA, USA).
All lung samples, infected and uninfected with HPAIV, were collected (day 1 and day 3 of each 5 samples). In this study, However, based on QC check and RIN values, some samples that did not meet the criteria were excluded (ratio < 1 or RIN < 7) and samples that passed the quality check were used to sequencing. The miRNA libraries were produced using the TruSeq Small RNA Sample Preparation Kit (Illumina, San Diego, CA, USA). The miRNA was separated by gel electrophoresis. miRNAs were ligated by their 3ʹ- and 5ʹ-end and then reverse transcribed and expanded to create miRNA libraries. The concentration and distribution of the eluted miRNA library were determined using a Bioanalyzer High-Sensitivity DNA Chip (Agilent Technologies). The expanded products were sequenced by LAS Company (Gimpo, Republic of Korea) on an Illumina NextSeq 500 System following Illumina’s recommended protocol to obtain single-end data of 75 bases.
After high-throughput small RNA sequencing, bioinformatic preprocessing and genome mapping were performed. Raw quality bases and adapters were trimmed using Skewer 0.2.2 [26]. The cleaned high-quality reads were mapped to the GRCg6a chicken reference genome, using QuickMIRSeq [27]. All known mature miRNAs and hairpins were obtained from the miRBase (https://www.mirbase.org/). QuickMIRSeq was used to estimate the mapped reads with the reference genome based on miRNA expression levels [27]. The hairpin and miRNA expression values were quantified in units of reads per million (RPM). Between the two selected biological conditions, DE miRNAs were analyzed using edgeR (empirical analysis of DGE in R, https://bioconductor.org/packages/release/bioc/html/edgeR.html). DE miRNAs with log2 fold change (FC) > 1 or < −1 with false discovery rate (FDR) less than 0.05 were considered DE miRNAs.
miRNA target genes were predicted to reveal miRNA functions. To predict mRNA targets, miRDB v6.0 (http://mirdb.org/), a miRNA target gene prediction database, was used. Candidate target genes with scores > 80 were used for bioinformatic analyses, such as Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichments. GO analyses were analyzed by Gene Ontology Resource (http://geneontology.org/) and GO terms with p-values less than or equal to 0.05 were summarized by REVIGO (http://revigo.irb.hr/). KEGG pathway analysis was analyzed by DAVID (https://david.ncifcrf.gov/summary.jsp).
Complementary DNA (cDNA) synthesis of miRNAs was conducted using the miScript® II Reverse Transcription Kit (Qiagen, Hilden, Germany) and Mir-X miRNA First-Strand Synthesis Kit (Takara, Kusatsu, Japan), following the manufacturer’s protocols. The miRNA cDNAs were used as template for qRT-PCR analysis. miRNA real-time PCR was performed on a LightCycler® 96 (Roche, Basel, Switzerland) and using the miScript® SYBR Green PCR Kit (Qiagen) and Mir-X miRNA qRT-PCR TB Green® Kit (Takara) following the manufacturer’s guidelines. Known miRNA primers used for qRT-PCR were derived from the miRNA database miRBase (Table 2). Primers for miRNA were synthesized by Genotech (Daejeon, Republic of Korea). qRT-PCR data were standardized relative to the U1A expression levels. Each qRT-PCR analysis was independently performed three times.
The target gene primers were designed using the NCBI primer design tool (Table 2). The cDNA synthesis process using total RNA was as follows. Total RNA (2 µg) was treated with 2 µL DNase I (Sigma-Aldrich, St. Louis, MO, USA) and incubated at 37 °C for 30 min. cDNAs were synthesized using the Revert Aid First Strand cDNA Synthesis kit (Thermo Fisher Scientific, Waltham, MA, USA) following the manufacturer’s instructions. First-strand cDNAs were used as templates for qRT-PCR amplification using AMPIGENE® qPCR Green Mix Lo-ROX (Enzo Life Sciences, Farmingdale, NY, USA) on a LightCycler® 96 System (Roche Life Science, Basel, Switzerland). cDNA was added to a mixture including 10 µL 2 × Power SYGR Green Master Mix, 1 µL of each forward and reverse primer, and nuclease-free water up to a total of 20 µL volume. The qRT-PCR results were normalized relative to the expression level of GAPDH. Each qRT-PCR experiment was performed in triplicate.
Statistical analyses were conducted using the IBM SPSS software (SPSS 26.0 for Windows, IBM, Chicago, IL, USA). Statistical data were confirmed using the Student’s t-test, and statistical significance was p < 0.05. All miRNAs and genes expression levels in the qRT-PCR experiment were calculated using the 2−ΔΔCt method [28]. qRT-PCR was replicated three times, and the mean ± standard error of the mean values for each set were validated.
RESULTS
After H5N1 infection, we observed symptoms such as emphysema and congestive lungs in the chickens. The quality of the 40 lung RNA samples was checked, and small RNA sequencing was performed on 29 samples (Supplementary Fig. S2). Among them, we focused on the comparison of resistant and susceptible lines after three days of infection with the HPAI H5N1 virus because clear symptoms after H5N1 infection showed at day 3 after infection (i.e., 3 days post-infection [dpi]). Small RNA sequencing of 13 libraries were performed at an average of approximately 41,323,928 read pairs per library. After trimming and quality checking, 35,833,365 read-pairs, on average, accounted for 90.1% of the clean read pairs with a Phred score of Q30 (Supplementary Table S1). After mapping with the chicken reference genome, 16,133,535 reads (51.91%) out of 31,081,156 total reads, on average, belonged to miRNA, whereas the others were hairpin loops (0.13%), small RNA (5.14%), mRNA (3.45%), and unaligned reads (39.37%) (Supplementary Table S2).
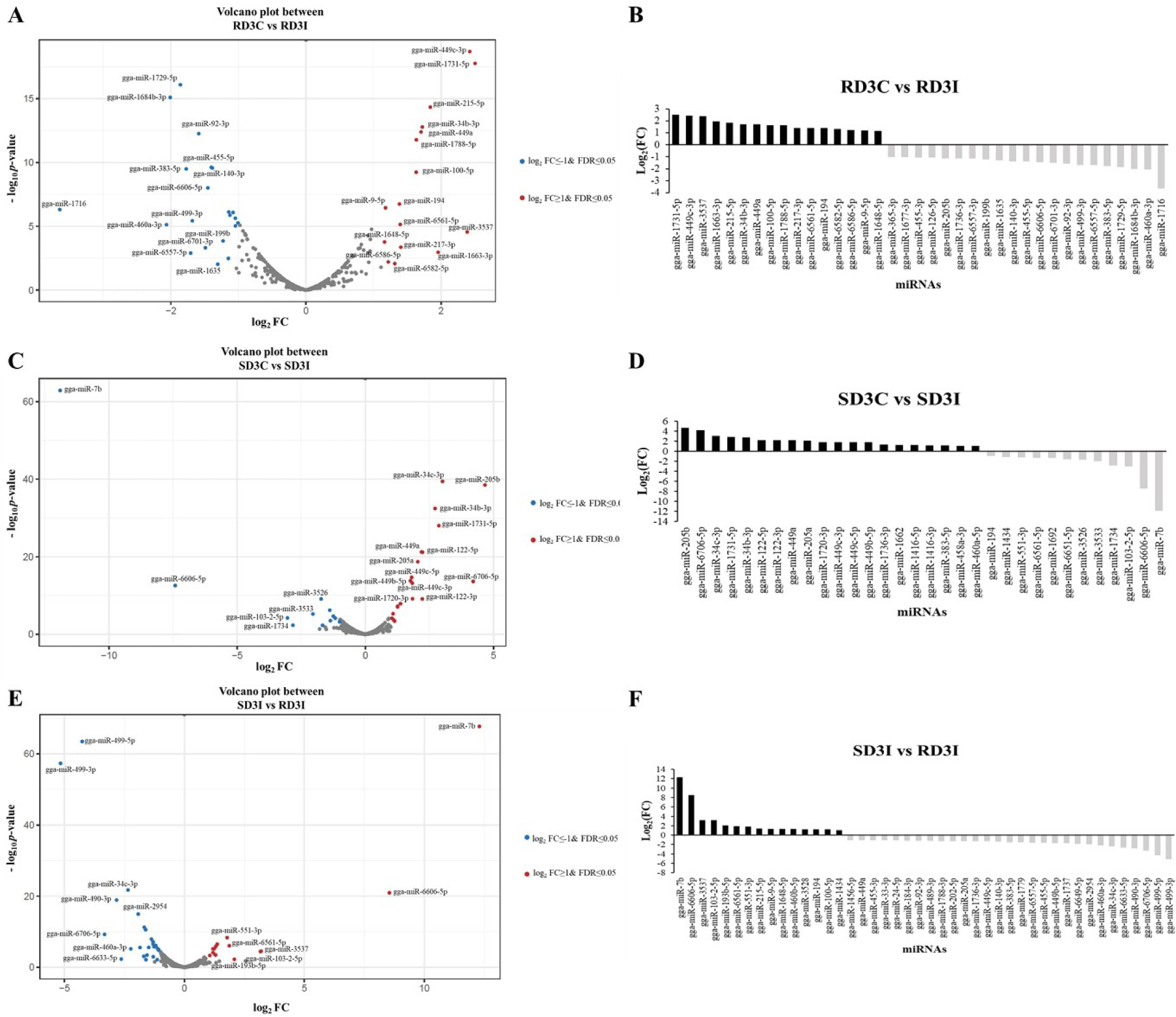
DE miRNAs in the lung tissue were described on volcano plots and bar graphs using log2FC and FDR, which are showed in Table 3 (Fig. 1). We compared the control and infected samples in the resistant and susceptible lines at 3 dpi. We also compared the H5N1 infection samples between resistant and susceptible lines at 3 dpi. The miRNAs with a log2FC >1 or < −1 and a FDR < 0.05 were considered DE miRNAs that are represented as blue dots and red dots in volcano plots. The miRNAs with a log2FC > 1 were denoted to have upregulated expression and those exhibiting a log2FC < −1 were denoted to have downregulated expression. Total 37 DE miRNAs were expressed in comparison between control and infected samples of 3 dpi resistant lines. The 16 DE miRNAs were upregulated in the infection samples compared control, while 21 DE miRNAs were downregulated. The gga-miR-1731-5p showed the highest log2FC and gga-miR-1716 showed the lowest log2FC among 37 DE miRNAs in comparison between control and infected samples of 3 dpi resistant lines (Figs. 1A and 1B). In addition, there were a total of 32 DE miRNAs between control and infection sample comparisons at 3 dpi in susceptible lines (Figs. 1C and 1D). The gga-miR-205b showed the highest log2FC among 12 DE miRNAs and gga-miR-7b showed the lowest log2FC among 20 DE miRNAs in this comparison group. Moreover, a total of 44 DE miRNAs were expressed in the infection samples between resistant and susceptible lines at 3 dpi (Figs. 1E and 1F). The 29 DE miRNAs were downregulated in the resistant line compared with the susceptible line, while 15 DE miRNAs were upregulated. In the resistant line, gga-miR-7b, gga-miR-6606-5p, and gga-miR-3537 showed high log2FC compared to the susceptible line at 3 dpi. Among them, gga-miR-7b showed the highest log2FC in the resistant line compared to the susceptible line. Furthermore, gga-miR-499-3p and gga-miR-499-5p were downregulated in the resistant line compared to the susceptible line at 3 dpi. Among them, gga-miR-499-3p showed the lowest log2FC in the resistant line compared to that in the susceptible line.
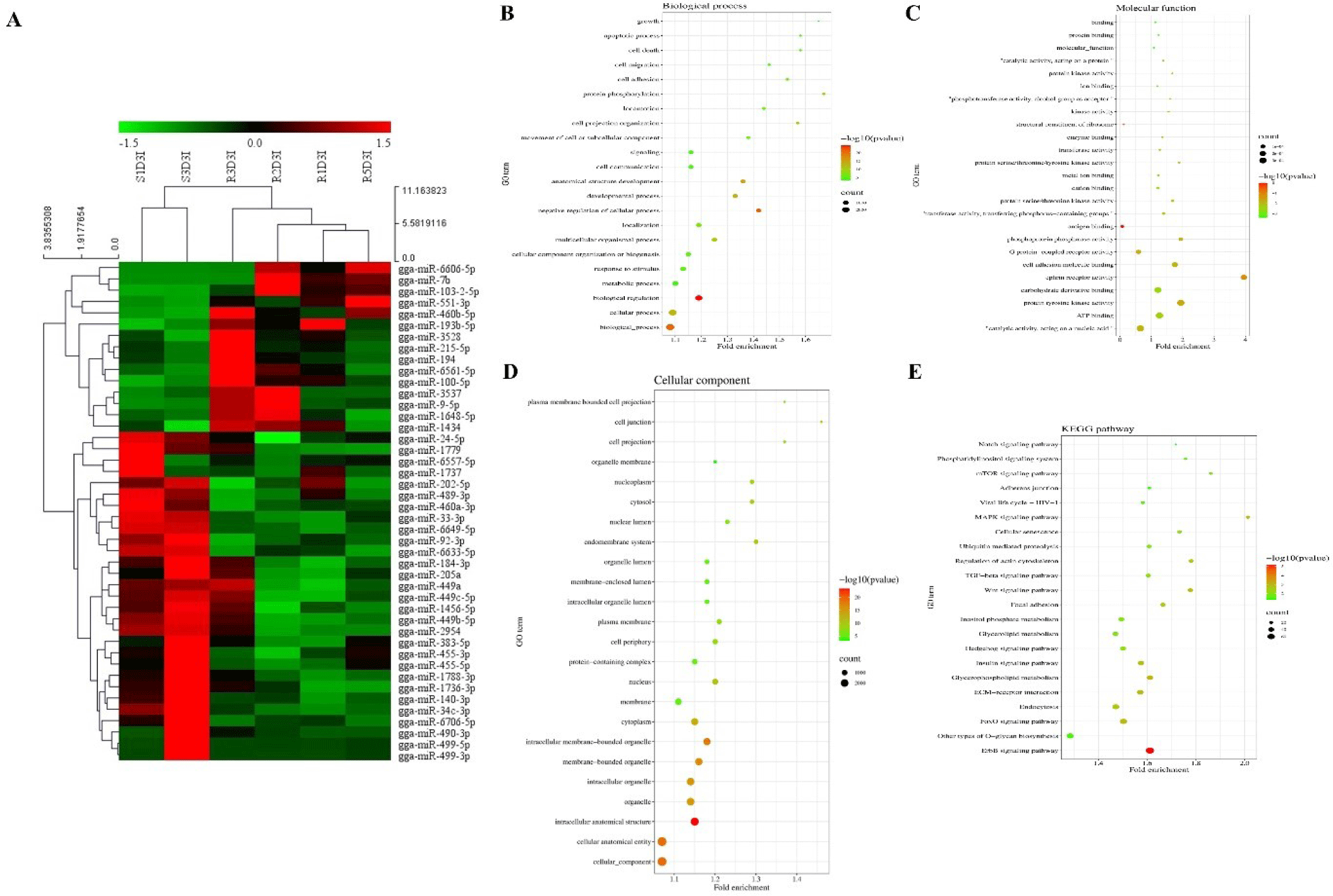
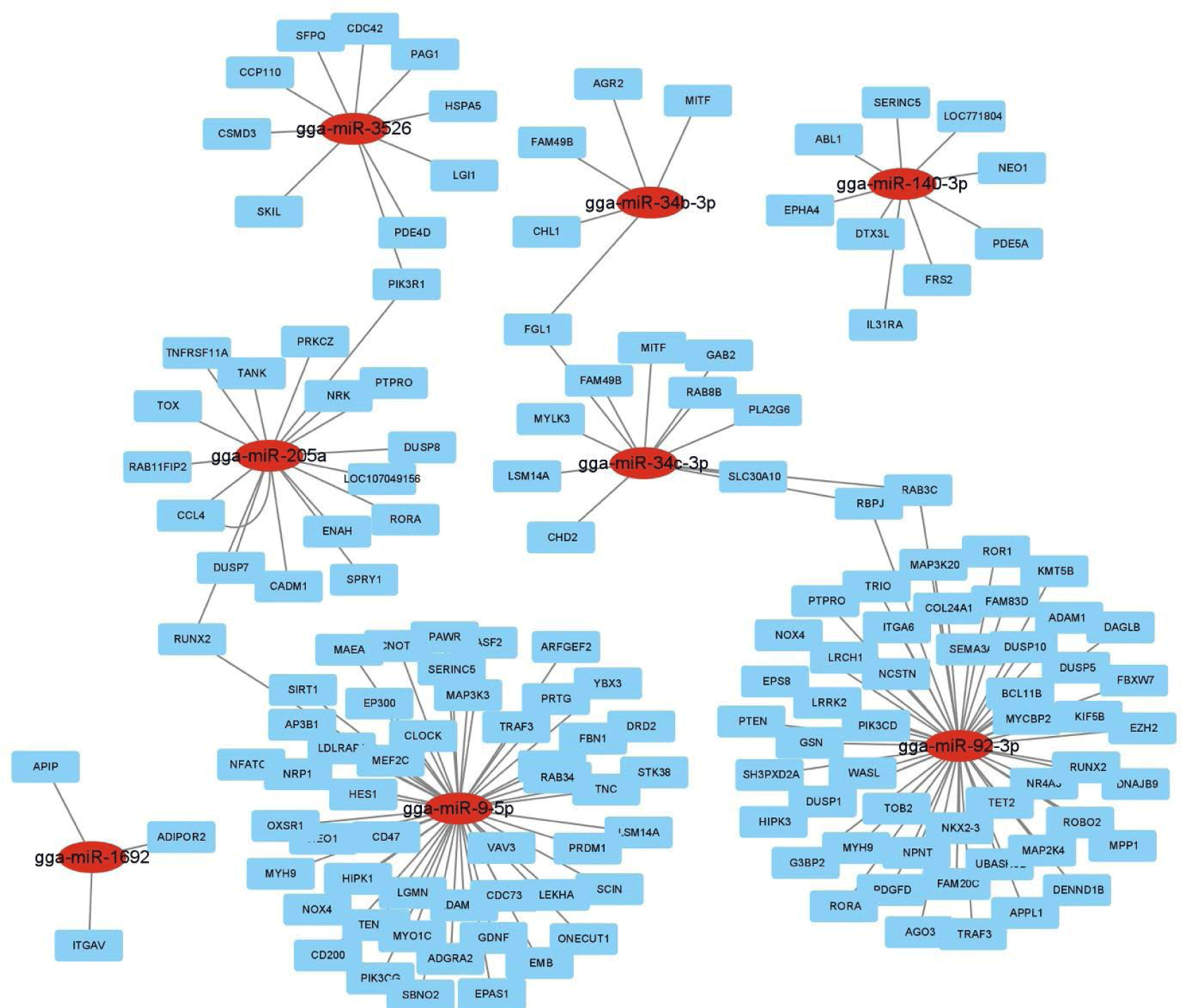
Hierarchical clustering analysis of 44 DE miRNAs was conducted between the two chicken lines at 3 dpi by the MeV program using Euclidean method (Fig. 2A). The Z-score was used to normalize values based on the expression levels. Moreover, the expression differences between resistant and susceptible could be confirmed through hierarchical clustering. Using miRNA target gene prediction tool miRDB, the target mRNAs of 44 DE miRNAs were predicted a score of 80 or higher. Around 25 GO terms with p-value <0.05 were enriched in biological process (BP), molecular function (MF), and cellular component (CC) categories using REVIGO and visualized by SRplot (Figs. 2B, 2C, and 2D) based on gene counts. In biological process GO terms, biological process, cellular process, and biological regulation were the most enriched terms. In molecular function GO terms, molecular function, binding, and protein binding were the most enriched terms. In cellular component GO terms, cellular component, cellular anatomical entity, and intracellular anatomical structure were the most enriched terms. Moreover, the target genes of the 44 DE miRNAs were involved in 22 KEGG pathways (Fig. 2E). The predicted target genes were involved in various immune-related pathways and signal transduction pathways such as the ErBb signaling pathway, MAPK signaling pathways, TGF-beta signaling pathway, Wnt signaling pathway, and mTOR signaling pathway. Furthermore, the interactions of these eight DE miRNAs and their predicted immune-related target genes were visualized by Cytoscape (Fig. 3). Red circles represent DE miRNAs, and blue rectangular boxes represent target genes. As shown in Fig. 3, various immune-related target genes were modulated by the DE miRNAs, and multiple immune target genes were modulated by more than one miRNA.
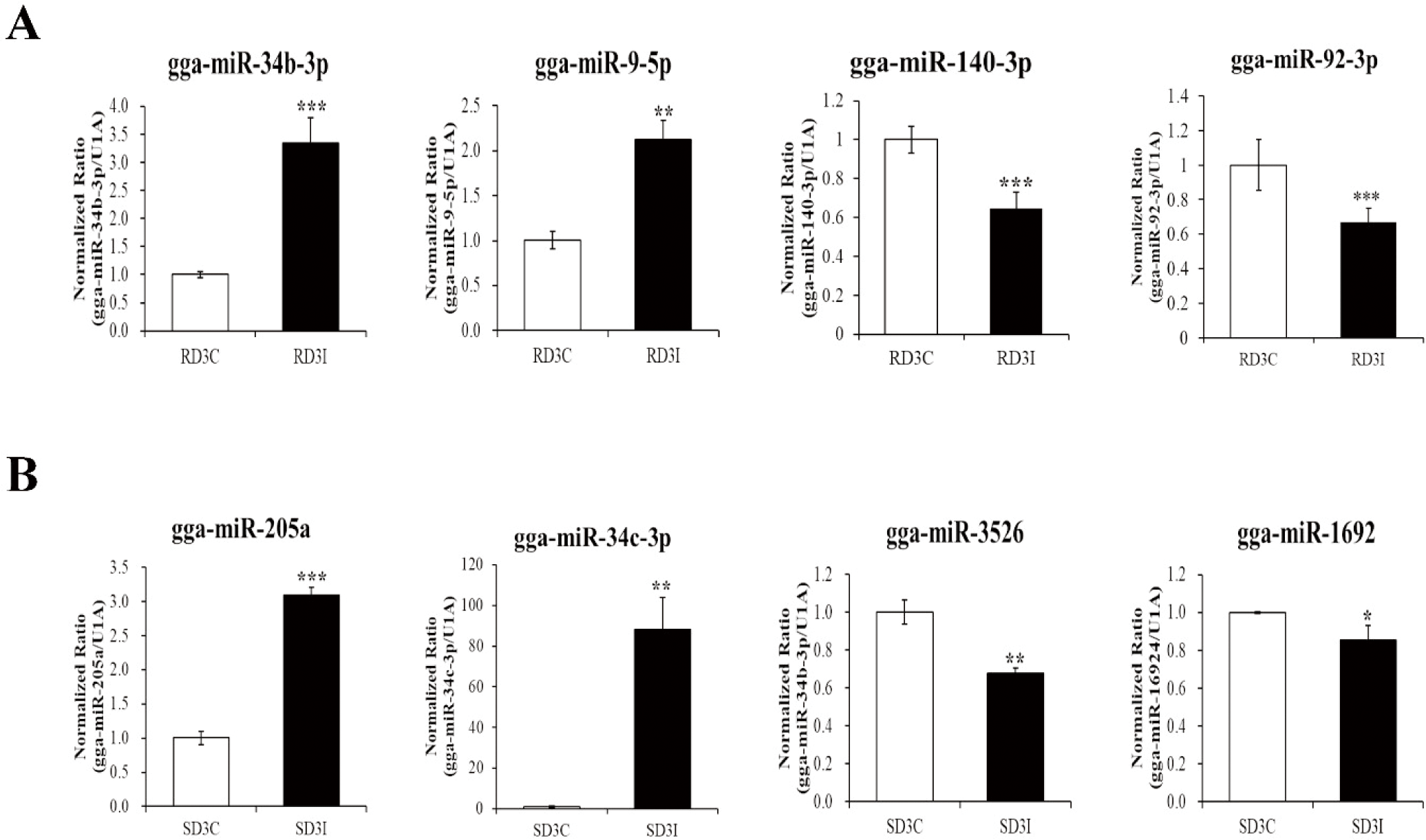
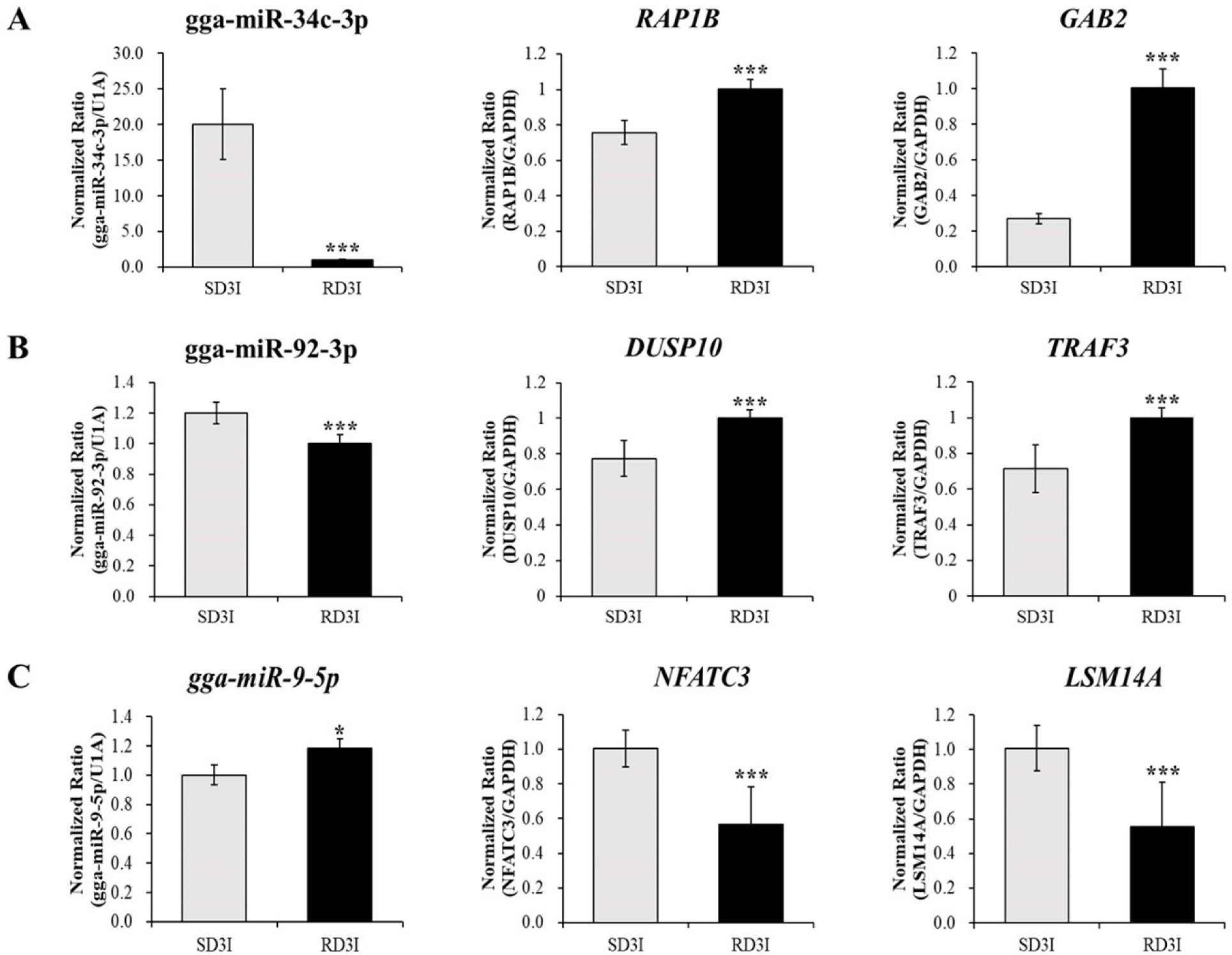
We validated the expression of DE miRNAs in the control and infected samples in the resistant and susceptible lines at 3 dpi via qRT-PCR (Fig. 4). Total of eight miRNAs were selected based on read counts, log2FC, and target genes. Four miRNAs, gga-miR-34b-3p, gga-miR-9-5p, gga-miR-140-3p, and gga-miR-92-3p, were validated comparison between control and infection in the resistant line at 3 dpi. The gga-miR-34b-3p, gga-miR-9-5p were up-regulated in the infection compared to control in the resistant line at 3 dpi. The gga-miR-140-3p, and gga-miR-92-3p down-regulated in the infection compared to control in the resistant line at 3 dpi. Moreover, four miRNAs gga-miR-34c-3p, gga-miR-205a, gga-miR-1692, and gga-miR-3526, were validated comparison between control and infection at 3 dpi of susceptible line. The gga-miR-34c-3p, gga-miR-205a were up-regulated in the infection compared to control in the susceptible line at 3 dpi. The gga-miR-1692, and gga-miR-3526 were down-regulated in the infection compared to control in the susceptible line at 3 dpi. These miRNAs’ immune related target genes were predicted by miRDB (Table 4). We also confirmed the expression of immune related target genes in the infected samples between resistant and susceptible lines at 3 dpi using qRT-PCR. The expressions of gga-miR-34c-3p and the predicted target genes, Ras-related Protein 1B (RAP1B) and Grb-associated binder 2 (GAB2) were confirmed via qRT-PCR (Fig. 5A). The expression of gga-miR-34c-3p was downregulated in the resistant line compared with that in the susceptible line. And the expression of target genes,RAP1B and GAB2 were upregulated in the resistant line. The expression level of gga-miR-92-3p was also downregulated in the resistant line and its target genes dual specificity phosphatase 10 (DUSP10) and tumor necrosis factor (TNF) receptor-associated factor 3 (TRAF3) were upregulated (Fig. 5B). In contrast, the expression of gga-miR-9-5p was upregulated in resistant line compared to those in the susceptible line and the expression of target genes, Nuclear factor of activated T cells 3 (NFATC3) and, Sm-like protein family 14A (LSM14A) were downregulated in resistant line (Fig.5C).
DISCUSSION
In this study, we analyzed miRNA profiles of Ri chickens, in the resistant and susceptible lines infected against HPAI H5N1 virus, using small RNA sequencing. Moreover, we predicted miRNA target genes using miRDB for DE miRNAs. Furthermore, various bioinformatic analysis, such as hierarchical clustering, GO, and KEGG pathway analyses, were conducted for miRNA target genes. The miRNAs and target mRNAs expression levels were validated via qRT-PCR.
The target genes RAP1B and GAB2 were found to be negatively correlated with gga-miR-34c-3p in this study. Expression levels of RAP1B andGAB2 were higher in resistant lines than in susceptible lines. Induction of miR-34c-3p was demonstrated in throat swab samples of H1N1-infected patients [29]. One of the target gene, RAP1B is a key signaling node in follicular thyroid carcinogenesis through PKA signaling in mice [30]. Moreover, RAP1B plays a crucial role in early T-cell humoral immunity and B-cell development [31]. A previous study suggested that after infection with the H5N1 virus, RAP1B expression may be involved in the host immune system by activating the T cell-dependent humoral immune system, B cell development, and biological processes [31]. The other target gene, GAB2, plays an important role in cell survival, differentiation, and growth by expressing a protein that interacts with various signaling pathways such as the PI3K, ERK, and JNK signaling pathways [32-36]. Previous studies have suggested that GAB2 has the potential to play a crucial role in cell immune signal transduction, but the mechanism ofGAB2 in avian influenza infection still requires further research [32,33,34]. This study suggests that gga-miR-34c-3p may activates the T cell-dependent humoral immune system, B cell development against avian influenza viruses and interacts with various signaling pathways such as the PI3K, ERK, and JNK signaling pathways by targeting the GAB2 and RAP1B genes after infected with H5N1.
In the present study, gga-miR-92-3p expression was downregulated in the resistant line compared with that in the susceptible line (Fig. 5). The most abundantly founded miRNA in chicken embryo fibroblasts upon H9N2 infection was the gga-miR-92-3p [37]. Moreover, this miRNA was also found in the various macrophage cell line, chicken HD11 and turkey IAH30 [38]. According to qRT-PCR results, gga-miR-92-3p targets TRAF3 and DUSP10. Expression of the immune target genes DUSP10 and TRAF3 was higher in the resistant line than in the susceptible line. DUSP10 (MKP5) is a regulator of MAP kinases such as JNK and p-38 kinases [39]. After influenza virus infection, numerous cytokines and pro-inflammatory cytokines are secreted by MAP kinases, which play a crucial role in the host innate antiviral response. The HPAI H5N1 virus has the potential to induce hypercytokinemia [9]. Therefore, the equilibrium between stimulating cytokine production and inactivating cytokine secretion is crucial to the host immune system. Unlimited secretion of cytokines occur various immune diseases [40]. Moreover, DUSP10 (MKP5) is also upregulated in avian influenza-infected chicken macrophages [41]. Previous studies suggest that DUSP10 (MKP5) inactivate cytokines and pro-inflammatory cytokines by inactivating MAP kinases to achieve equilibrium [39,41]. The other target gene, TRAF3, encodes a protein that activates the secretion of type I IFNs, such as IFN-α and IFN-β [42]. After infection with the avian influenza virus, TRAF3 interacts with Mitochondrial antiviral signaling protein (MAVS), which is associated with retinoic acid-inducible gene I (RIG-I) signaling against virus infection [43,44]. After influenza A virus interaction, TRAF3 activates IRF3, IRF7, and NF-κB to stimulate the production of type I IFN genes and pro-inflammatory cytokines, which are critical to the host immune response [45]. The present study suggests that gga-miR-92-3p may regulate MAP kinases and activate the secretion of type I IFNs as an active immune modulator in response to HPAIV infection by targeting DUSP10 and TRAF3. Moreover, the present study suggests that immune functions were more active in resistant line than in susceptible line through these predicted target genes results.
The gga-miR-9-5p expression was upregulated in the resistant line compared with that in the susceptible line (Fig. 5). According to previous study, gga-miR-9-5p was involved in various signal transduction and immune-related pathways by regulating target genes in the intestinal mucosal layer (IML) of necrotic enteritis (NE)-induced Fayoumi chicken lines [46]. The immune-related target genes of gga-miR-9-5p, LSM14A and NFATC3, were verified via RT-PCR. These target genes showed negative correlation with gga-miR-9-5p. LSM14A expression was higher in the susceptible line compared to resistant line in present study. LSM14A contributes to activation of IFN-β in the early period of virus infection [47]. The IFN-β expression was also upregulated in the H5N1 infected susceptible strain of mice compared to resistant [48]. IFN-β activates both pro-inflammatory and anti-inflammatory cytokines [49]. These reports suggest that gga-miR-9-5p may modulate immune responses such as activation of IFN-β via LSM14A. The other target gene NFATC3 (also known as NFAT4) mediates the various cytokines and immune modulatory gene expressions such as IFN-γ and TNF-α [50]. Moreover, NFAT4 plays a crucial role in the reproduction and survival of T cells [51]. These previous papers suggest that gga-miR-9-5p may mediate cytokines and T cell survival by NFATC3. However, since the function of LSM14A and NFATC3 in resistant and susceptible lines infected with AIV has not yet been elucidated, further research is needed. Furthermore, the miR-140-3p was downregulated in the infection sample compared to control in the resistant line at 3 dpi (Fig. 4). Previous study suggested that miR-140-3p regulates TNF-α-induced activation of MAPK and NF-κB by targeting CD38 [52].
Most candidate target genes of DE miRNAs were involved in BP GO terms, comprising biological processes, cellular processes, and metabolic process. Biological processes included the control of gene expression, protein modification, and interaction with proteins or substrate molecules. Cellular components, cellular anatomical entities, and intracellular anatomical structures were the most enriched cellular components obtained through the analysis of the candidate target genes of DE miRNAs upon HPAIV infection. Dendrites were also enriched in GO terms related to cellular components (data not shown). A previous study showed that chicken dendritic cells are involved in inflammation, which is induced during early HPAIV infection, triggering deregulation of the immune response [53]. In addition, dendritic cells participate in the dissemination of the H5N1 virus after the virus escapes viral-specific immunity that leading to cell death [54]. Moreover, various genes that related to the virus life cycle were involved in GO cellular component. The viral ribonucleoproteins (vRNP) gained entry to the host cell nucleoplasm and transported to the nucleus to replicate the influenza virus. After replication, the vRNP complex was exported to the cytoplasm and the plasma membrane for viral assembly. After viral assembly, the influenza virus was released [55].
DE miRNA candidate target genes were involved in various signal transduction and immune-related pathways such as ErbB signaling pathway, MAPK signaling pathway, TGF-beta signaling pathway, Wnt signaling pathway, and mTOR signaling pathway. Protein synthesis and actin cytoskeleton function in signaling pathways were induced by virus evasion [56]. These were modified upon influenza A virus infection, as observed in LLC-MK2 monkey kidney epithelial cells [57], and A549 human lung adenocarcinoma epithelial cell line [58]. Focal adhesion interacts with PI3K signaling and actin reconstitution after influenza A virus infection [59]. ErbB signaling pathway modulates immune responses by Interferon (IFN) –λ and CXCL10 against influenza A virus and Rhinovirus [60]. MAP kinase cascades are triggered upon influenza virus infection [61], which has been demonstrated as a novel approach for the development of antiviral drugs against the influenza virus [62]. MAPK signaling pathway modulates immune responses by regulation of pro-inflammatory cytokines [63]. The epithelial-derived TGF-β suppressed early immune responses during influenza virus infection [64]. The Wnt/β-catenin signaling may improve replication of influenza virus replication [65]. The PI3K/mTOR signaling pathway positively modulates immune cell activation. Moreover, in the dendritic cells, these pathways regulate type I IFN production by activating the IFN-regulatory factor 7 [66]. After infected against H5N1, miRNAs regulate immune responses via these various signaling pathways.
In this study, we compared control and infection samples in resistant and susceptible lines of Ri chickens especially infection samples between resistant and susceptible lines. The 44 DE miRNAs were confirmed to DE among the H5N1 infected susceptible and resistant lines at 3 dpi. Moreover, GO and KEGG pathway analysis identified their predicted target gene functions. Several DE miRNAs (gga-miR-92-3p, gga-miR-34b-3p, gga-miR-140-3p, gga-miR-205a, gga-miR-9-5p, gga-miR-3526, gga-miR-1692, and gga-miR-34c-3p) and some target genes expressions were validated using qRT-PCR. Therefore, this study revealed the potential regulation of miRNAs that mediate their candidate target genes related to the immune response against HPAIV infection. This may facilitate further studies on the overall understanding of the immune system regulation of miRNAs against HPAIV infection. Moreover, the present study may beneficial to the development of miRNA-based resistant and susceptible biomarkers corresponding to HPAI virus infection in poultry.
SUPPLEMENTARY MATERIALS
Supplementary materials are only available online from: https://doi.org/10.5187/jast.2022.e127.
