INTRODUCTION
With the increasing emphasis on animal health and livestock product safety, the development of non-toxic, residue-free, and multifunctional “green” feed additives has become an inevitable trend in the development of the livestock industry [1]. Methylsulfonylmethane (MSM) is a stable metabolite of dimethyl sulfoxide (DMSO), which has the characteristics of low toxicity, low residue, and low susceptibility to pathogen resistance [2–4]. Research indicates that MSM has biological functions, including antioxidant, anti-inflammatory, immune regulation, and improvement of intestinal health [5,6]. At the same time, when used as an additive, MSM can improve animal growth performance and production quality [6–14], enhance immune function [6,8,9,15,16], regulate lipid metabolism [6,10,13,17–20], and relieve stress [7,13,21,22]. This article mainly reviews the biological functions and applications of MSM in animal production, in order to provide reference for the further development and utilization of MSM in animal husbandry.
BRIEF INTRODUCTION OF METHYLSULFONYLMETHANE
MSM is a small sulfur compound with the formula (CH)₂SO₂. It appears as a white acicular crystal at room temperature and can be soluble in water (150 g/L at 20°C) and organic solvents such as ethanol [5,6]. MSM is involved in the synthesis of methionine and cysteine and is of great significance for maintaining normal metabolic activities [23]. MSM is mainly absorbed by passive diffusion in the small intestine and can also be converted into sulfate by cecal flora [24]. The metabolites are excreted in urine and can penetrate the blood-brain barrier to protect nerves [24–26]. The synthesis of MSM depends on the metabolite DMSO, which can be rapidly converted into MSM by oral administration [26]. In the natural environment, DMSO is oxidized by ultraviolet light or lightning to generate MSM, forming a sulfur cycle, and artificial synthesis can be achieved by oxidizing DMSO by H₂O₂ or NO₂ [5,26,27]. MSM is a naturally occurring source of sulfate (accounting for 85% of sulfur supply) available in different fruits, vegetables, grains, and animal tissues, and is considered to provide health benefits when used to supplement the diet [6,24]. Cow’s milk is the richest source of MSM; other animal feed ingredients containing MSM include Swiss chard (0.05–0.18 ppm), corn (up to 0.11 ppm), and alfalfa (0.07 ppm) [28]. In addition, plants of the Allium genus contain a large amount of MSM, including garlic (Allium sativum L.), onion (Allium cepa), and chive (Allium tuberosum) [29]. The content of MSM in common feeds and foods is shown in Table 1 [28]. Thus, MSM could also be naturally synthesized in animals fed an MSM-free diet; the natural presence of MSM in animal products might be endogenous, exogenous, or both [6].
| Type | Content |
|---|---|
| Alfalfa | 0.77 |
| Corn | up to 0.11 |
| Swiss chard | up to 0.18 |
| Tomatoes | up to 0.86 |
| Cow’s milk | 3.30 |
| Tea | 0.30 |
| Coffee | 1.60 |
| Beer | 0.18 |
BIOGICAL FUNCTIONS AND MECHANISM OF METHYLSULFONYLMETHANE
Oxidative stress often causes animals to produce a large amount of reactive oxygen species (ROS), leading to oxidative damage, organ dysfunction, and reduced animal production performance [6]. MSM has been shown to possess strong antioxidant activities in animals such as broiler chickens, laying hens, ducks, and mice [5,6]. The mechanisms of MSM’s antioxidant activity includes increasing the activity of superoxide dismutase [6,8,20,30–32], enhancing total antioxidant capacity (TAC) [6–8,10,33,34], reducing malondialdehyde (MDA) [8,10,12,13,20,31,32,35], and regulating the glutathione system (e.g., glutathione synthesis and glutathione peroxidase activity) [7,8,13,20,32,35]. In addition, MSM can inhibit oxidative damage mediated by mitochondrial ROS (superoxide/H2O2/HClO), human immunodeficiency virus-1 transcription activator, oxidized oil, CoCl2, heat stress, and ethanol [7,13,21,22,36,37]. Cyclooxygenase-2 (COX-2) and nitric oxide synthase (iNOS) are two key enzymes that induce ROS production [13,38]. MSM can downregulate COX-2/iNOS expression through the nuclear factor kappa-B (NF-κB) pathway and indirectly block ROS generation [13,39]. Furthermore, MSM can regulate the balance between ROS and antioxidant enzymes by controlling other signaling pathways: Janus kinase/signal transducer and activator of transcription [40,41], nuclear factor (erythroid-derived 2)-like 2, and phosphoinositide 3-kinase/protein kinase B [37]. The antioxidant effect of MSM on various animal species is presented in Table 2 below.
| Items1) | Animals | Sample | Dietary MSM | Time | p-value2) | Dietary MSM decreased (–) or increased (+) items’ concentration compared with the non-supplemented control | Ref. |
|---|---|---|---|---|---|---|---|
| GPX (U/L) | 73-week-old laying hens (Lohmann Brown Lite) | Serum | 0, 0.1, 0.2, 0.3 and 0.4% | 4/8/12 wk | > 0.05 | No effects | [30] |
| CAT (U/mL) | |||||||
| 8-OHdG (ng/mL) | |||||||
| SOD (%) | 12 wk | 0.002 | +31.24 | ||||
| TAC (mM) | 8/12 wk | < 0.05 | +0.32/+0.44 | ||||
| MDA (µM) | 8 wk | 0.025 | –6.85 | ||||
| SOD (%) | 73-wk-old laying hens (Lohmann Brown-Lite) | Serum | 0 and 2 g/kg | 12 wk | 0.014 | +18 | [10] |
| TAC (mM) | 0.068 | No effects | |||||
| GPX (U/L) | 0.644 | ||||||
| CAT (U/mL) | 0.861 | ||||||
| MDA (μM) | 0.324 | ||||||
| 8-OHdG (ng/mL) | 0.886 | ||||||
| GPX (U/mg) | Liver | 0.199 | No effects | ||||
| CAT (U/mg) | 0.920 | ||||||
| TAC (nmol/mg) | 0.002 | +6.19 | |||||
| MDA (nmol/mg) | 0.026 | –0.51 | |||||
| GPX (U/mL) | Male Ross 308 broilers | Plasma | 0, 1 and 2 g/kg | 23/25/39 d | > 0.05 | No effects | [13] |
| SOD (%) | |||||||
| MDA (mL) | 25/39 d | < 0.05 | –1.6/–2.2 | ||||
| GSH (μmol/mL) | Erythrocytes | 23/39 d | < 0.05 | +0.34/+0.17 | |||
| GSSG (μmol/mL) | 23/25/39 d | > 0.05 | No effects | ||||
| GSSG/GSH | |||||||
| GPX (U/mL) | Liver | 23 d | 0.043 | –1.0 | |||
| SOD (%) | 23/25/39 d | > 0.05 | No effects | ||||
| MDA (mL) | |||||||
| GSH (μmol/mL) | |||||||
| GSSG (μmol/mL) | |||||||
| GSSG/GSH | |||||||
| MDA (nmol/mL) | 55-wk-old Jing-fen No.6 laying hens | Serum | 0, 350 and 700 mg/kg | 4 wk | < 0.05 | – | [20] |
| SOD (U/mL) | < 0.05 | + | |||||
| CAT (U/mL) | > 0.05 | No effects | |||||
| GPX (U/mL) | |||||||
| MDA (nmol/mg) | Liver | < 0.05 | – | ||||
| SOD (U/mg mg) | < 0.05 | + | |||||
| CAT (U/mL) | > 0.05 | No effects | |||||
| GPX (U/mg) | < 0.05 | + | |||||
| TAC (mM) | Broiler chicks (Ross 308) | Serum | 0 and 2 g/kg | 21 d | > 0.05 | No effects | [33] |
| MDA (µM) | |||||||
| TAC (nmol/mg) | Liver | < 0.05 | +13.07 | ||||
| MDA (nmol/mg) | > 0.05 | No effects | |||||
| TAC (nmol/mg) | Ileum | > 0.05 | No effects | ||||
| MDA (µM) | |||||||
| MDA (µM) | 73-week-old laying hens (Lohmann Brown-Lite) | Serum | 0 and 2.0 g/kg | 12 wk | 0.025 | –10.05 | [34] |
| SOD (%) | 4/8/12 wk | > 0.05 | No effects | ||||
| TAC (mM) | |||||||
| CAT (U/mL) | |||||||
| GPX (U/L) | |||||||
| 8-OHdG (ng/mL) | |||||||
| TAC (nmol/mg) | Liver | 12 wk | < 0.001 | +10.76 | |||
| GPX (U/mg) | > 0.05 | No effects | |||||
| CAT (U/mg) | |||||||
| MDA (nmol/mg) | |||||||
| SOD (U/mL) | Female Pekin ducklings | Serum | 0, 0.15 and 0.3% | 42 d | 0.03 | +23 | [8] |
| GPX (U/mL) | 0.02 | +33 | |||||
| MDA (nmol/mL) | 0.03 | –1.14 | |||||
| TAC (U/mL) | 0.02 | +6.4 | |||||
| IgG (μg/mL) | 0.38 | No effects | |||||
| TAC (mmol/L) | Ross 308 male broiler chicks | Serum | 0 and 0.05% | 7 d | 0.023 | +0.4 | [7] |
| TBARS (μmol/L) | 7/14/21 d | > 0.05 | No effects | ||||
| TGSH (μmol/mL) | |||||||
| GPX (nmol/mL) | |||||||
| GR (nmol/mL) | |||||||
| TBARS (μmol/g) | Liver | 7/14/21 d | > 0.05 | No effects | |||
| TAC (mmol/g) | |||||||
| TGSH (μmol/g) | 21 d | 0.012 | – | ||||
| GPX (nmol/mg) | 21 d | 0.003 | + | ||||
| GR (nmol/mg) | 7 d | 0.004 | + | ||||
| MDA (μmol) | Male Ross 308 chicks | Serum | 0 and 0.4% | 21/28 d | < 0.05 | –3.34/–3.26 | [12] |
| TAC (mmol) | 14/21/28 d | < 0.05 | –0.4/+0.53/–0.15 | ||||
| SOD (U/mL) | Ragdoll kittens | Serum | 0, 0.2 and 0.4% | 0/35/65 d | > 0.05 | No effects | [49] |
| MDA (nmol/mL) | |||||||
| GSH (U) | |||||||
| CAT (U/mL) | |||||||
| TAC (mM) | |||||||
| SOD (U/mg) | Rats | Liver | 0 and 400 mg/kg | 5 d | < 0.05 | +1.74 ± 0.1 | [31] |
| TAC (U/gm) | +14.59 ± 0.3 | ||||||
| MDA (µM/g) | –5.36 ± 0.1 | ||||||
| MDA (nmol/mg) | Rats | Colonic tissue | 0 and 400 mg/kg | 4 d | < 0.05 | – | [35] |
| MPO (U/mg) | – | ||||||
| GSH (nM/mg) | + | ||||||
| CAT (nmol/mg) | + | ||||||
| MDA (nmol/mg) | Rats | Liver | 0 and 100 mg/kg | 7 d | < 0.05 | –3.75 ± 0.59 | [32] |
| MPO (U/mg) | –1.7 ± 0.4 | ||||||
| SOD (U/mg) | +0.8 ± 0.3 | ||||||
| GSH (nmol/mg) | +10.32 ± 0.93 |
CAT, catalase; GPX, glutathione peroxidase; GR, glutathione reductase; GSH, glutathione; GSSG, glutathione disulphide; IgG, immunoglobulin G; MDA, malondialdehyde; MPO, myeloperoxidase; SOD, superoxide dismutase; TAC, total antioxidant capacity; TBARS, thiobarbituric acid reactive substances; TGSH, total glutathione; 8-OHdG, 8-hydroxydeoxyguanosine.
Animal studies on anti-inflammatory effects have mainly demonstrated the inhibitory effects of MSM on interleukin (IL)-1β, IL-2, IL-6, and tumor necrosis factor alpha (TNF-α) [21,23,35,42–44], though some reports suggested that MSM increased IL-2 and IL-6 in some poultry experiments [6,8,45]. The core mechanisms of MSM anti-inflammatory include: blocking the NF-κB signaling pathway and decreasing the expression of iNOS/COX-2, thereby downregulating the IL-1β, IL-6, and TNF-α [13,39,46]; directly bidirectionally regulating cytokines (inhibiting inflammatory cytokines and promoting anti-inflammatory factors) [47,48]; and regulating the activities of MDA, myeloperoxidase, glutathione, and TAC related to oxidative pathways [35]. In addition, MSM can also exert anti-inflammatory effects by inhibiting extracellular signal-related kinases/Jun amino terminal kinases-mitogen-activated protein kinases and leucine-rich repeat family pyrin domain containing 3 pathways [29,44]. The anti-inflammatory effect of MSM on various animal species is presented in Table 3 below.
| Items1) | Animals | Sample | Dietary MSM | Time | p-value2) | Dietary MSM decreased (–) or increased (+) items’ concentration compared with the non-supplemented control | Ref. |
|---|---|---|---|---|---|---|---|
| IL-10 | Male ross 308 chicks | Ceca | 0 and 0.4% | 12/19 d | > 0.05 | No effects | [12] |
| IL-1β | |||||||
| IFN-γ | |||||||
| IL-2 (ng/mL) | Female Pekin ducks | Serum | 0, 0.15 and 0.3% | 42 d | 0.03 | +14 | [8] |
| IL-6 (ng/mL) | 0.04 | +2.9 | |||||
| IFN-γ (ng/mL) | 0.02 | –2.6 | |||||
| TNF-α (pg/mL) | 0.03 | –2.7 | |||||
| IL-2 mRNA | Ross broiler chicks | Spleen | 0 and 0.10% | 35 d | 0.192 | No effects | [45] |
| IL-2 mRNA | Bursa | 0.008 | +0.686 | ||||
| IL-2 (pg/mL) | Serum | 0.029 | +19 | ||||
| IL-6 (pg/mL) | 0.018 | +25 | |||||
| TNF-α | Chickens | Intestinal epithelial cells | 0 and 100 mM | 24 h | < 0.05 | – | [42] |
| IL-1β | |||||||
| IL-6 | |||||||
| TNF-α | White leghorn chickens | Chicken like macrophages (HD11 cells) | 0 and 200 mmol/L | 12 h | < 0.05 | – | [44] |
| IL-1β | |||||||
| IL-6 | |||||||
| IL-8 | > 0.05 | No effects | |||||
| TNF-α | Pigs | IPEC-J2 cells | 0, 200 and 300 mmol/L | 24 h | < 0.05 | – | [43] |
| IL-6 | |||||||
| IL-1 | |||||||
| IL-1β (pg/mg) | Rats | Colonic | 400 mg/kg | 4 d | < 0.05 | – | [35] |
| TNF-α (pg/mg) | > 0.05 | No effects | |||||
| TNF-α (pg/mg) | Swiss albino mice | Gastric tissue | 0, 200 and 400 mg/kg | 1 h | < 0.05 | – | [23] |
| IL-1β (pg/mg) | |||||||
| IL-6 (pg/mg) | |||||||
| MCP-1 (pg/mg) | |||||||
| TNF-α (pg/mg) | Male C57BL/6 mice | Brain | 0, 200 and 400 mg/kg/day | 12 d | < 0.05 | – | [21] |
| IL-1β (pg/mg) | |||||||
| IL-6 (pg/mg) | |||||||
| MCP-1 (pg/mg) |
Mediating the interaction between oxidative stress and inflammatory response is one of the key mechanisms for MSM to regulate immune response [5,6]. For example, MSM has been shown to increase IL-2 and IL-6 expression levels in poultry [6,8,45], reduce the incidence of lymphadenopathy, splenomegaly, and anemia in mice with immune lymphoproliferative disorders [15], upregulate the mRNA expression of immune-related genes [16], and elevate alanine aminotransferase activity along with the quantity of white blood cells and lymphocytes in broilers [9].
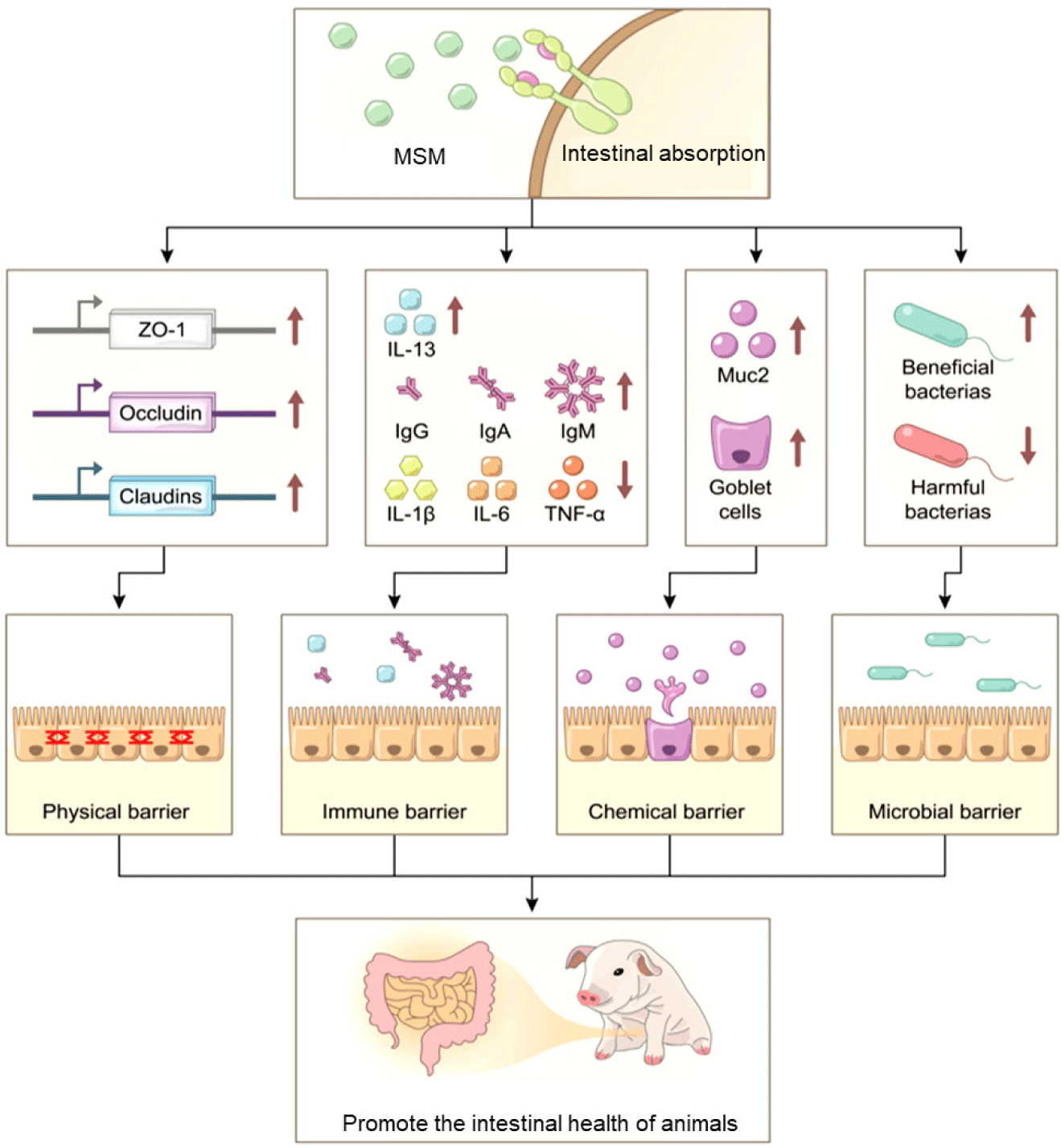
MSM can improve animal intestinal health mainly by enhancing intestinal barrier function (Fig. 1). In vivo studies have shown that MSM can increase the ileal villus height and villus height-to-crypt ratio [10], regulate the gut microbiota by reducing Escherichia coli and increasing Lactobacillus populations [9,49], decrease the severity of ethanol/HCl-induced gastric mucosal injury [23], improve intestinal damage [35], and increase the intestinal transport capacity of animals [17]. In vitro studies have shown that MSM inhibits lipopolysaccharide-induced inflammatory cytokines (TNF- α, IL-1 β, and IL-6) in chicken and porcine intestinal epithelial cells, while regulating cell proliferation and viability [42,43]. Furthermore, MSM can increase the monolayer transepithelial electrical resistance and upregulate the expression of tight junction proteins (occludin, claudin-1, and zonula occludens-1), while reducing the permeability of porcine intestinal epithelial cells [43]. Overall, these findings illustrate the multifactorial mechanism by which MSM maintains intestinal mucosal homeostasis through structural strengthening, microbial balance, and inflammatory pathway regulation.
Due to the beneficial anti-inflammatory and immunomodulatory properties of MSM, it is usually used as a dietary supplement to treat diseases such as arthritis, osteoarthritis, rheumatoid arthritis, and knee joint injuries [46–48,50]. Animal studies have demonstrated that MSM can alleviate knee joint and cartilage degeneration in mice, reduce cartilage damage in rheumatoid arthritis rats, protect the articular cartilage surface in rabbits with osteoarthritis, and prevent cartilage erosion in the meniscus of sheep [51–55]. MSM also affects osteogenic differentiation, bone activity, and bone density of cartilage, and exhibits potential for bone induction and conduction in bone regeneration [56–61]. Furthermore, MSM can promote chondrogenesis and osteogenic differentiation in zebrafish by regulating the SRY-box transcription factor 9/RUNX family transcription factor 2 [56], which are key regulatory genes for chondrogenesis and osteogenic differentiation, respectively [56,58,60]. Additionally, MSM can enhance alkaline phosphatase activity in rabbit radius defect repair and regulate collagen cross-linking and mineralization mediated by key enzymes such as transglutaminase-2 [57,58]. These findings demonstrate that the protective mechanism of MSM on animal bones and joints involves the dual pathways of inflammation inhibition and bone metabolism regulation.
The potential nourishing effects of MSM on skin and hair in animals are hypothesized to be due to its dual mechanisms of supplying sulfur as a critical substrate for the synthesis of structural proteins (methionine, cysteine, and keratin) and exerting antioxidant and anti-inflammatory effects to mitigate tissue damage [3,23,49,62,63]. Experimental evidence demonstrates that MSM can reduce fine lines and wrinkles [64], enhance skin firmness, elasticity, and hydration [65], relieve symptoms of X-linked ichthyosis [66], induce melanin synthesis, and treat skin hypopigmentation diseases such as vitiligo [67]. In studies of animals such as rabbits, guinea pigs, rats, mice, ragdoll kittens, and poodles, MSM has also been shown to have non-irritating effects on the skin and can improve burn skin condition, reduce skin wrinkles caused by ultraviolet radiation B damage, prevent photoaging and reduce the thickness of hair scales [5,49,68].
In vitro studies show that MSM induces cell death by inhibiting signal transducer and activator of transcription 3 and 5B [69], inhibiting Janus kinase/signal transducer and activator of transcription [70], and activating caspase pathways [71], and showed anticancer effects against a variety of cancers (breast, esophagus, stomach, liver, colon, bladder, skin, prostate, lung, and endometrial cancers) [5,69–72]. Furthermore, MSM alleviates exercise-induced pathological conditions (e.g., cardiac dysfunction and muscle injury) via oxidative stress protection [16,73], improves cholesterol metabolism by increasing high-density lipoprotein levels in obesity models [74], and combats Alzheimer’s disease-like neurotoxicity [75]. These biological properties indicate the potential of MSM in improving animal welfare and quality of life.
THE APPLICATION OF METHYLSULFONYLMETHANE IN PRODUCTION
MSM has biological properties such as anti-inflammatory, antioxidant, and immune and intestinal function regulation. These characteristics are crucial for maintaining a balanced gut microbiota, reducing body oxidation, lowering environmental stress, and ultimately improving animal performance [9,10,12,33]. Reports on the effect of MSM on the growth performance of animals were mostly focused on poultry [6], although the research results are not entirely consistent. Some studies have revealed that dietary MSM has no marked effect on poultry growth performance parameters such as body weight (BW), body weight gain (BWG), feed intake, or feed conversion ratio (FCR) [7,18,20,33]. Additionally, it shows no influence on the production performance or egg quality of laying hens [10,20,30]. Conversely, other studies have demonstrated that MSM, either alone or in combination with other additives, generally exerts a positive effect on certain growth performance indicators in poultry, ragdoll cats, and pigs, for example, by decreasing the FCR [11,12,30,34,49]. A small number of studies have indicated that MSM can notably improve the growth performance of broilers and ducks. Jiao et al. [9] found that the supplementation of 0.20% MSM to broiler diets improved BW and BWG and reduced FCR during a 29-d experiment. Zhang et al. [13] found that the administration of MSM at 1 and 2 g/kg resulted in a linear increase in BWG and a reduction in the FCR during the grower phase (days 10–21) in broilers. Yan et al. [8] indicated that the inclusion of MSM (0.3%) resulted in an increase in final BW and BWG during the periods of days 22–42 and days 1–42, and a reduction in FCR during the period of days 22–42 in Pekin ducks. Lim et al. [45] found that MSM (0.1%) produced significant increases in BW, BWG, and feed intake. Overall, the effects of supplementation of animal diets with MSM have not found any related adverse effects on growth performance [6]. The observed differences in the research results may be attributed to various confounding variables, including the form of MSM addition, feed formulation characteristics, species-specific metabolic responses, dose optimization challenges, dietary matrix interactions, and feeding environment and period [6–10,13,49]. This complex interplay of factors underscores the necessity for standardized protocols in future nutraceutical research to elucidate MSM’s precise mechanisms of action.
In terms of carcass yield, breast meat characteristics, and poultry viscera, in most current studies, there is no difference in the relative weight of carcass, abdominal fat, breast meat, thigh and drumstick, liver, gizzard, pancreas, thymus, bursa of Fabricius, or spleen (the amount of MSM added ranges from 0.025% to 0.3%) [6,8,9,13].
The water-holding capacity (WHC), cooking loss, and drip loss play the key role in the production and processing of meat products by affecting the sensory characteristics and nutritional value of meat, thereby affecting meat quality [13]. Current research predominantly demonstrates that MSM supplementation exerts beneficial effects on these parameters, particularly through its modulatory effects on the pH value of meat [6,13,76]. The characteristic pH decline following muscle fiber contraction after slaughter disrupts protein electrostatic interactions, consequently impairing WHC [77]. Notably, MSM administration (0.3%) has been shown to enhance WHC through pH24h elevation and drip loss reduction [8]. Consistent with this result, a study reported that the supplementation of MSM (0.03%) can improve the WHC of ducks [18]. However, Jiao et al. [9] did not observe a positive effect of MSM (0.05%–0.2%) on pH or WHC in broiler chickens, though they observed a linear reduction in drip loss on days 5–7 after slaughter. Similarly, Lee et al. [19] observed a reduction in drip loss in finishing pigs following the addition of MSM (0.03%–0.05%) on day 2 post-mortem. Contrastingly, some studies have failed to detect MSM’s influence on drip loss parameters [13]. In addition, changes in storage temperature and cellular structure can also affect the WHC, cooking loss, and drip loss of meat products [77]. However, as of now, research on the impact of MSM in these areas is still rare, and further exploration is needed in future studies.
The color of raw meat is highly susceptible to alterations in feed and the environment [78]. Current research demonstrates that the supplementation of MSM could increase meat color of redness (CIE a*) in finishing pigs, broilers, and ducks. The underlying mechanism may involve two synergistic pathways: First, the inherent antioxidant capacity of MSM may inhibit myoglobin oxidation [8], thereby delaying the conversion of myoglobin to metmyoglobin during storage, which is the main determinant of meat discoloration [78,79]. Second, MSM appears to modulate heme in myoglobin after animal death. Lee et al. [19] observed an increase in iron deposition in the loin of finishing pigs that were fed a diet supplemented with MSM (0.03%–0.05%.). This iron enrichment may contribute to improved pigment stability, given the established correlation between iron content and meat CIE a* [80]. Notably, while significantly influencing CIE a* parameters, MSM had no significant effect on CIE L* and CIE b* [8,9,18,19]. Zhang et al. [13] found that birds receiving a 2 g/kg MSM diet had higher rates of moderate white striations, suggesting that the fat content of fillets may be increased with a corresponding decrease in protein percentage [81]. This could lead to changes in the nutritional value of the final meat product.
Lipid composition serves as a critical determinant of meat quality, with MDA constituting a principal biomarker for assessing lipid peroxidation intensity in animal-derived products [6]. As we summarized earlier, the comprehensive analysis of multiple species, including laying hens, broiler chickens, ducks, mice, etc., consistently show that supplementing with MSM significantly inhibited MDA levels, confirming its ability to alleviate systemic lipid peroxidation [8,10,12,13,20,31,32,35]. The regulatory mechanisms involved include that MSM may alleviate steatosis by regulating the antioxidant defense system (enhancing superoxide dismutase, TAC, and glutathione peroxidase) and inhibiting mitochondrial energy metabolism related genes [20]. Meanwhile, supplementing MSM can significantly reduce the 2-thiobarbituric acid reactive substance (TBARS) in finishing pigs, broiler chickens, and meat ducks [13,18,19]. The presence of elevated levels of unsaturated fat in meat renders it susceptible to oxidation, which can result in the development of rancidity and a deterioration in both flavour and colour [78,82]. TBARS can alter the concentration of secondary lipid oxidation products, which may cause odors in the meat. The decrease in TBARS levels may also be attributed to the strong antioxidant activity of MSM in scavenging free radicals [13]. It is worth noting that in addition to reducing MDA and TBARS, the antioxidant effects of MSM may also help improve the nutritional properties of meat. For example, dietary MSM supplementation in ducks effectively preserves fatty acid integrity [18], while supplementation in pigs has been shown to alter muscle amino acid profiles [19]. Overall, the antioxidant effect of MSM is the primary reason for the improvement in meat quality and reduction of lipid peroxidation observed in livestock and poultry.
Due to its numerous biological functions, MSM has been shown to enhance animals’ disease resistance and environmental stress resistance through anti-inflammatory, antioxidant, and immune-regulating effects [7,12,13,21,22,44]. Mycoplasma gallisepticum is a common avian pathogen that frequently causes respiratory diseases in poultry, MSM alleviate inflammatory damage and oxidative stress induced by M. gallisepticum by inhibiting the NF-κB and extracellular signal-related kinases/Jun amino terminal kinases-mitogen-activated protein kinases signaling pathways in tracheal tissue and macrophages [44]. Coccidiosis is a parasitic disease in poultry caused by Eimeria, which affects the gastrointestinal tract of chickens, reduces the animal’s ability to digest nutrients, and leads to malabsorption, affecting the growth performance and animal production [12,33]. In coccidiosis of broilers (Eimeria infection), MSM improves growth performance (e.g., elevated BWG), reduces lipid peroxidation (lower TBARS), enhances systemic antioxidant capacity (increased plasma and hepatic TAC), and improves crude ash digestibility, though its direct anti-coccidial efficacy remains limited [12,33]. In addition to disease resistance, as mentioned earlier, MSM has good oxidative stress resistance [7,21,22]. Meanwhile, research has also found that MSM (1 and 2 g/kg) may have a sustained effect on heat-stressed broilers during the finishing period [13]. MSM enhances heat resistance, reduces mortality rate, and improves the oxidative stability of breast meat due to its antioxidant properties [13]. Overall, MSM alleviates pathogen- and stress-induced physiological disorders in poultry primarily by modulating inflammatory signaling and enhancing antioxidant defenses, although its efficacy may vary depending on the disease-specific contexts.
CONCLUSIONS
In summary, MSM is a sulfur-containing organic compound widely existing in nature, which has biological functions, including antioxidant, anti-inflammatory, and immune regulation, etc. MSM participates in various metabolic processes in animals and plays an important role in the efficient production and health maintenance of animals. However, the application of MSM in animal production mainly focuses on the growth performance and the quality of animal products, and its mechanism still needs to be further explored. The functions of MSM in regulating nutrient absorption and metabolism, killing pathogenic microorganisms, and regulating hormone secretion are needed to explore in the future. At the same time, further studies of different forms and dosages of MSM and their different effects on animal production can provide more and more authoritative data for the promotion of MSM in animal diets and its practical application in animal production.
