INTRODUCTION
Time is a crucial factor in all food production systems, as technology efficiency is measured based on production yield over time. High yield within a short period is usually favored in a fast-paced system. In other words, quantity is prioritized over quality, as different processes and additives can be used to later improve the quality to meet consumer acceptance.
In the field of meat production, fast growth rate and high feed conversion efficiency are favored to meet the demand for meat, whether for direct consumption or further processing [1]. Consequently, innovations related to meat production are aimed toward faster growth and higher muscle mass through improved breeding programs, development of high-quality feeds and additives, genetic modification of animals (e.g., disease resistance, double muscling, fast growth), improved housing conditions, and improvements in overall animal health [2–4]. Moreover, growth promoters have been developed that mainly target the improvement of feed utilization and accelerated growth rates [1]. Despite improvements in practices for livestock meat production, there remains a demand for slaughter-free products and sustainable food sources [5].
Cultivated meat production is a technology aimed at addressing welfare and sustainability issues in animal-based food production [6]. The demand for alternative protein sources has been increasing as environmental and ethical concerns over conventional meat production continue to be magnified. This promising technology involves the mass-production of animal cells (e.g., embryonic stem cells [ESCs], induced pluripotent stem cells [iPSCs], mesenchymal stem cells [MSCs], adipocytes, muscle satellite cells), which are grown in vitro, to provide raw materials essential in simulating conventional meat or meat products [7,8]. One of the promises of cultivated meat technology is the large-scale production of meat precursors in a shorter period than traditional meat production, which entails months to years of production time depending on the livestock species [9]. However, the status of cultivated meat production remains at pre-commercialization level due to the relatively high production costs associated with scale-up and commercialization [10]. Consequently, academic researchers and industry stakeholders are collaborating to address the high production costs by developing low-cost media components, three-dimensional (3D) biomaterials, and cell engineering, for example [11].
Ultimately, traditional and cultivated meat production both aim to ensure food security for future generations. Considering the effects of a continuously increasing global population and global warming, strategies and innovations must be developed to mitigate the impacts of these global issues on food security, specifically animal-based products. Thus, this review focuses on identifying growth promoters for traditional and cultivated meat production.
COMPARISON BETWEEN TRADITIONAL AND CULTURED MEAT PRODUCTION
Meat, a popular animal-based food product, is an important source of nutrients (e.g., proteins, essential amino acids, vitamins, minerals) in human diets [12]. Traditional meat production encompasses several stages, such as animal rearing, slaughtering, processing, and packaging and distribution [13]. Conversely, cultivated meat production involves cell selection, mass cultivation, 3D formation/assembly, processing, and packaging and distribution [6]. Meat from both production systems consists of animal cells and their metabolic products, based on the processes of myogenesis in vivo or in vitro [14]. However, it is reasonable to conclude that traditional meat is more cell diverse, considering the presence of connective tissues and other cellular components from adjacent organs [15]. Meanwhile, cultivated meat is currently a product of monocultured myogenic cells due to the current limitations in media formulations, scaffolding, bioreactors, and other variables that support in vitro cultivation [8,16]. Despite the technology for industrializing cultivated meat being in its nascent stage and a mass production process not yet established, cultivated meat holds promise as an alternative meat source for human consumption, provided it meets acceptable production efficiency and regulatory standards [17]. This section reviews the differences between traditional and cultivated meat production in terms of time or production duration. It also explores previous and current growth promoters used as accelerators in either of these meat production systems.
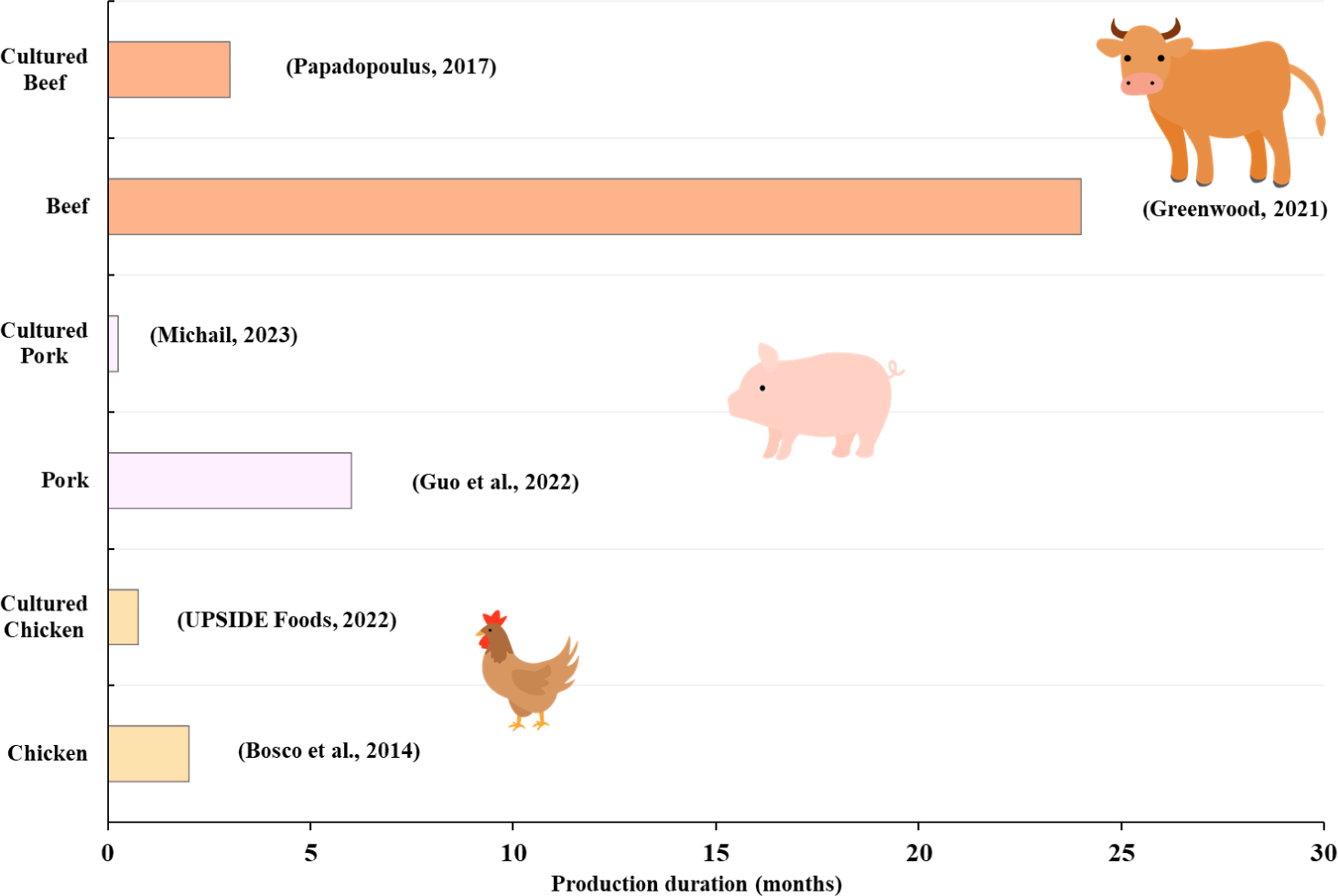
Major meat types, including chicken, pork, and beef, undergo several processing steps before consumption. The largest difference among these meat types lies in the production process, particularly the rearing of animals until they reach physical maturity as a meat source [2–4]. Commonly, the acceptability for slaughter is based on weight and age [18–20]. For practical reasons, underperforming animals, in terms of meat production-relevant growth parameters, are culled at an earlier age to prevent wastage of animal feed, supplements, and other cost-incurring practices [21]. Underperformance is typically measured by the feed conversion ratio (FCR), which is a metric that assesses feed intake and weight gain. Given the high cost of feeds, FCR has become an important measure and guide for livestock raisers, especially for meat production [22]. However, it should be noted that improved rearing and production time is just one consideration among others in meat production. Chickens have a significantly shorter meat production period compared to swine and beef cattle (Fig. 1). Their short rearing time and high feed conversion efficiency make them a popular meat type due to the protein content and affordability [23,24]. Meanwhile, pork production takes less than 6 months to reach slaughter weight (Fig. 1) [25,26]. This is significantly shorter than beef production, which takes approximately four times the duration of pork production [26,27]. Both chicken and swine are monogastric animals, thus having a more direct digestion of feed as a source for muscle build-up [28] compared to beef cattle, which rely heavily on their gut microbiome for feed digestion, as cellulose and hemicellulose are indigestible without microbial enzymes [29]. Differences in the digestive system, as well as the type of feed consumed, result in variations in the duration of meat production among these animals. Therefore, optimized feeding strategies are needed to expedite the production process and meet the increasing demand for meat.
The majority of the production cost in livestock raising is for feed/nutrition [30]. This is because the building blocks for the growth and development of animals come from their diet. For this reason, strategies to improve livestock production are directed toward optimizing their diets to provide optimal nutritional benefits, resulting in improved health, increased weight gain, and superior product quality [31]. Given the importance of nutrition, it is important to understand how nutrients are allocated after digestion.
There are generally five physiological functions in animals—maintenance, immune response, growth, reproduction, and fat storage, which differ based on factors such as species, age, health, genetic potential, and environmental conditions [32]. Maintenance involves functions such as organ function and thermoregulation; thus, it lies at the top of the hierarchy for nutrient allocation [33]. Depending on the animal’s health and well-being, the immune response utilizes nutrients to provide active immunity and prevent damage from existing health issues, which explains why health management and disease prevention are important strategies for improving livestock production [34]. After satisfying maintenance needs, the growth of organs, bones, and muscles is then prioritized, which is the reason basal metabolic needs are considered in the computation for feed formulation to provide more than just the maintenance needs of an animal [35]. Reproduction, particularly gamete production and gestation, also utilizes nutrients [35]. Any excess from the previously mentioned functions is converted into fat, which is stored as an energy source for the animal [36].
Decades of livestock research and development have led to numerous scientific breakthroughs enabling highly efficient animal production systems. Improvement in animal production is often attributed to a holistic approach, which involves considering multiple factors that the animals need to promote high growth and performance [3]. For example, in terms of genetic improvement, the use of genetic markers to identify genetically superior individuals can fast-track the pooling of desirable production traits. This can be supported by improved management practices that ensure the health and welfare of animals, thereby allowing the favorable expression of desired traits. However, among production requirements, nutritional optimization takes the lead as arguably the biggest factor affecting animal production [37].
Although optimized nutrition can lead to improved muscle gain, a shorter production period is still desired. As a solution, growth promoters have been introduced to improve animal growth rates, whether intentionally or unintentionally (i.e., as a side effect) [38]. In this review, five categories of growth promoters were identified, namely, antibiotic growth promoters (AGPs), hormonal growth promoters (HGPs), microbiome-based promoters (MPs), enzyme additives (EAs), and phytogenic additives (PAs). Table 1 lists the various categories of growth promoters along with representative products and the mechanism behind their growth-promoting effects [39–56].
| Category | Product examples | General mechanism | References |
|---|---|---|---|
| Antibiotic growth promoters (AGPs) | Tylosin, Bacitracin, Virginiamycin | Improve growth rates and feed efficiency by modifying gut flora. | [39–41] |
| Hormonal growth promoters (HGPs) | Trenbolone acetate, Zeranol, Estradiol (active ingredient, ractopamine) (Paylean®, Optaflexx®), Zilpaterol | Increase muscle deposition and feed conversion rates. | [42–46] |
| Microbiome-based growth promoters (e.g., probiotics, prebiotics, postbiotics, parabiotics) | Lactobacillus, Bifidobacterium, Inulin, Fructooligosaccharides | Promote beneficial gut microflora and enhance immunity and nutrient absorption. | [47–49] |
| Enzyme additives | Phytase, Xylanase, Protease | Improve nutrient availability by breaking down complex feed components such as non-starch polysaccharides. | [50–52] |
| Phytobiotics/Phytogenics | Plant oils, Spices, Oleoresins, Plant extracts | Have antimicrobial activity, anti-inflammatory activity, and enzyme-enhancing activity, which can improve animal health and productivity. | [53–56] |
AGPs tend to benefit the animals by mainly enhancing their growth and efficiency, and preventing disease occurrence [39,40,57]. Generally, antibiotics have diverse mechanisms of action against bacteria, but mainly include the inhibition of cell wall, protein, and DNA synthesis, as well as the disintegration of the cytoplasmic membrane [58]. In the study of Plata et al. [41], it was determined that narasin can influence the nitrogen utilization of chicken gut microbiomes, leading to improved performance [41]. However, a study using AGPs (e.g., virginiamycin, chlortetracycline, bacitracin methyl disalicylate, lincomycin, tylosin) in broiler production showed no significant effects on growth or feed efficiency, thereby calling for the reduction of AGP use as a precautionary measure toward antimicrobial resistance [59]. Meanwhile, the use of tylosin in pigs did not yield a significant difference in weight gain but resulted in earlier maturation of swine gut microbiota [60]. Similarly, pigs fed with a combination of chlortetracycline, sulfathiazole, and penicillin did not show growth promotion but inhibited the growth of potential pathogenic bacteria in weaned piglets [61]. In beef production, Bretschneider et al. [62] reviewed and concluded that there is an increasing trend in the average daily gain and feed conversion of forage-fed beef cattle treated with increasing ionophore (e.g., lasalocid, tetronasin, lysocellin) dosage [62]. Based on the reviewed reports, no single AGP can be concluded to be effective across species given the usual combination of multiple AGPs as treatments. This suggests that the use of AGPs differ among species. Although certain combination of AGPs have been found to improve animal gut health and performance, the disadvantages of using AGPs must also be taken into account. The most crucial consideration in the use of AGPs is the development of antimicrobial resistance among pathogenic microorganisms, which poses a threat to both animal and human health [63,64].
HGPs are natural or synthetic hormones primarily used to influence muscle deposition and improve feed conversion rates [65,66]. In broilers, Islam et al. [67] found that dexamethasone supplementation negatively affected the feed intake, feed efficiency, and weight gain [67]. The study also warned of the potential accumulation of dexamethasone in the meat. Meanwhile, after reviewing numerous studies that involved the use of HGPs in beef and pork production, Aroeira et al. [65] noted that the administration of the correct dosage of HGPs in livestock animals can effectively meet production demands for animal protein. For example, Athayde et al. [68] concluded that a 5 mg/kg ractopamine supplementation can improve the performance of pigs without affecting pork quality parameters (e.g., color, drip loss, marbling score, intramuscular fat content) [68]. Meanwhile, the use of zilpaterol hydrochloride improved live growth performance, carcass yield, and characteristics of beef steers [69]. Given the advantages of HGPs in livestock production, countries such as the United States, Canada, and Australia still permit their use, albeit under strict regulations [70]. The ban in Europe is due to a report that found six HGPs to have deleterious effects (e.g., endocrine-disrupting, genotoxic, immunotoxic, carcinogenic) on consumers’ health [70,71]. The recognition of risks associated with the use of HGPs has triggered numerous studies aimed at developing analytical methods for detecting residues in animal meat and other products [72,73]
Instead of AGPs and HGPs, alternatives focusing on promoting beneficial gut microflora to enhance immunity and nutrient absorption are being promoted. These MPs can be classified into probiotics (e.g., Lactobacillus, Bifidobacterium), prebiotics (e.g., non-digestible oligosaccharides, non-starch polysaccharides), and postbiotics (e.g., exopolysaccharides, short-chain fatty acids, enzymes, cell-free supernatants) [47,48]. Altogether, these MPs promote the proliferation of beneficial microorganisms while decreasing the population of potential pathogenic ones, thereby improving overall gut health, nutrient absorption and utilization, and immunity [74,75].
EAs, conversely, improve nutrient availability by catalyzing the digestion of feed components [50,52]. By facilitating feed digestion, the energy typically needed to digest feed components is reduced, allowing for energy allocation toward other physiological functions such as maintenance and growth. Some commonly used enzymes include phytase, xylanase, and protease [50–52]. These enzymes not only increase digestibility but also reduce feed cost. Additionally, phytase has been used in pig diets to reduce phosphate pollution in pigs and poultry, thereby mitigating the environmental impact of pig and poultry production [50].
Meanwhile, PAs are plant-derived feed additives that possess immunoregulatory, antimicrobial, antimutagenic, and/or antioxidative properties [53]. These beneficial properties can improve animal health, similar to the mechanism of MPs and AGPs. In broiler production, the use of Achyranthes japonica extract has been shown to improve growth efficiency, nutrient utilization, daily feed intake, and dry matter digestibility [76,77]. Similarly, the extract from Silybum marianum was shown to enhance growth performance and nutrient absorption in both weaned piglets and fattening pigs [78,79]. In beef production, Digestarom®, a commercial phytogenic feed additive, improved the dry matter intake and carcass weight of beef steers [54]. The increasing familiarity with AGP and HGP alternatives shows increasing awareness of antimicrobial resistance and its consequences, as well as considerations for animal welfare in commercial animal production systems.
Overall, shorter production periods were never mentioned in any of the reports reviewed, mainly because production duration was kept constant, and differences in growth performance were primarily observed. From a different perspective, the increase or enhancement in average daily gain can be related to time: the higher the average daily gain, the faster the ideal slaughter weight is reached. This means that growth promoters can shorten production strictly based on the time it takes to reach the ideal slaughter weight. Thus, the increased growth performance and nutrient utilization efficiency could result in lower production costs due to shortened production duration and feed provision.
As a developing technology geared toward sustainable animal-based food production, cultured meat represents a promising alternative protein source for human diets. Unlike traditional meat production, which uses feed components/stuff as a source of nutrients for growth and maintenance, cultured meat production relies on cell culture media as a direct source of essential nutrients for cell proliferation and maintenance. Analogous to FCR, media conversion ratio (MCR) can be used to determine nutrient utilization efficiency, which is calculated as the total mass of cultured meat produced divided by the total volume/mass of culture media used [80]. This allows researchers and developers to assess cost efficiency, environmental sustainability, and scalability of cultured meat [80,81]. In example, Santander et al. [82] theoretically computed the conversion ratio of cultivated chicken using the current cultured meat production system estimating to be at 1.8 which is comparable to the FCR of traditional chicken production of 1.6–17 [82]. However, the cost of cultured meat is still significantly higher compared to traditional meat.
Currently, pioneering cultured meat companies (e.g., Aleph Farms, GOOD Meat/Eat Just, UPSIDE Foods, Mosa Meat, Believer Meats, Meatable) have provided their approximate production times, offering insight into the typical duration of cultured meat production. Production duration depends on the time it takes to complete the main production stages, namely, cell expansion, differentiation/maturation, and harvest and postharvest processing [83]. Production scale can also affect production duration, with small-scale production estimated to be about four times longer than pilot-scale or early commercial production, assuming optimized processes [84]. Some leading cultured meat companies provided estimates of production duration (Fig. 1). Mosa Meat is estimated to produce beef within approximately 3 months, while Meatable claims to be able to produce hybrid sausages containing cultured pork as quickly as 4 days [85,86]. Meanwhile, UPSIDE Foods, the first to commercialize cultured chicken, claims to produce structured chicken filets in about 3 weeks [87]. The relatively fast production reports of cultured meat companies already provide insight into the potential of this production system to provide sustainable and resource-efficient animal-based products. However, numerous factors should be considered before making direct comparisons based on production efficiency among companies. For example, companies would differ in the cells (e.g., primary myogenic, immortalized, engineered, fibroblasts, ESCs, iPSCs) or method (e.g., suspension, 3D-printing, scaffold-based) used, all depending on production strategy and goals. Thus, careful considerations must be taken prior to making conclusions regarding production efficiency.
The culture medium contains all the essential building blocks for cells to grow and develop. In traditional animal production, culture media are synonymous with feeds, which function as the source of all essential and non-essential nutrients to maintain, promote, or influence physiological functions. For this reason, it is necessary to understand how nutrients are allocated in vitro in relation to cell proliferation and differentiation for cultured meat production.
Unlike live animals, which require a digestive system for digesting feed and a circulatory system for transporting nutrients, cell cultures have immediate access to nutrients available in the media [88,89]. The processes involved in feed conversion alone use energy and involve several body systems, which entail additional nutrient and energy expenditures for maintenance and growth [88]. Thus, the direct utilization of nutrients by the cells in vitro is arguably more nutrient- and energy-efficient. Generally, the culture media must be able to provide amino acids for protein synthesis, fatty acids for cell membrane development, carbohydrates (e.g., glucose, galactose, sucrose) as an energy source, and vitamins and minerals to support cell functioning [90,91].
Cells in vitro differ from cells in vivo in their nutrient uptake, based on their growth phases. These phases are regulated by the activity of adenosine monophosphate (AMP)-activated protein kinase (AMPK) and mammalian target of rapamycin complex 1 (mTORC1), which are involved in cellular metabolism [92]. For example, proliferating cells need more nutrients to increase in mass and number, while quiescent cells are less metabolically active, resulting in lower nutrient and energy requirements [93]. The difference in the commitment of cells to either proliferate, differentiate, or become quiescent affects the overall utilization of nutrients in the culture media. This highlights the importance of growth factors, which regulate cell commitment, in optimizing cell culture efficiency. Generally, growth factors dictate cell fate by binding to cell-surface receptors that triggers a cascade of events such as receptor activation, signal transduction, and alteration of gene and protein expression leading to cell commitment [94,95]. Without these growth factors, production goals such as proliferation and differentiation will not be achieved. Thus, the optimization of culture media composition, with high considerations towards growth factor supplementation, has become important in cost-reduction efforts in cultured meat production [96,97].
Arguably, the most important component in cultured meat production is the cell type/line used. Each cell type/line used as the progenitor in cultured meat differs in metabolism, nutrient requirements, and growth patterns, thus making it an important consideration prior to large-scale cultivation [15,80]. The usual monoculture of cells in cultured meat production makes non-secretory progenitor cells rely on the growth factors and cytokines available from the growth medium. O’Neill et al. [80] demonstrated that multiple cell types used for cultured meat exhibit significant differences in their culture media nutrient utilization [80]. This suggests that specialized media formulation is needed depending on the cell type used. Additionally, cell passage numbers should also be considered, as differences in growth and metabolic rates, as well as cell functioning, have been observed [98,99]. These changes are most likely due to repeated passaging of cells which can lead to genetic instability and phenotypic variability [100,101]. Consequently, media conversion efficiency of the progenitor cells will dictate the cost efficiency and profitability of cultured meat. For example, bovine primary myoblasts require 20% fetal bovine serum (FBS) supplementation during proliferation, but fibroblast cells need only 10% FBS to proliferate. The difference in the amount of serum or growth factors used can directly impact the production cost [102]. Thus, it can be inferred that the production efficiency in cultured meat production relies heavily on the cells used. Considering these factors, MCR of cultured meat production cannot be used as a sole determinant of production efficiency.
As a direct impact of the progenitor cells, optimization of media composition ensues. It is estimated that the majority of the production cost is attributed to the culture media [17]. There are two types of culture media used in production: proliferation/growth media and differentiation media. The culture media includes basal media, which comprises the bulk of the total volume and contains the essential macro- and micronutrients for cell growth. It is typically supplemented with serum, which contains growth factors and cytokines that regulate cell metabolism and growth [7]. However, animal sera have undefined chemical composition and are targeted for replacement or removal due to ethical issues, performance variability, and most importantly, high cost [103]. The development of serum-free media can greatly reduce the costs of cultured meat production by eliminating animal serum in the media composition [103,104]. Numerous reports provide promising results towards the development of serum-free media for cultured meat, with words such as simple, effective, and defined being common among recent reports [105–107]. Good Meat has reportedly produced cultured chicken meat using serum-free media [108]. Meanwhile, companies such as CellMeat and Multus Biotechnology continue to dedicate their efforts on serum-free media development [109,110].
To meet the demand for cultured meat, time- and cost-efficient production systems are necessary. Time and resources are important considerations in assessing the efficiency and sustainability of cultured meat production, parallel to those in traditional meat production. In other words, a faster and less expensive production system is desirable. In response to this need, academic institutions and industry players have reported innovations focused on improving cell proliferation and differentiation in cultured meat production, which are vital for achieving high yields and similar physicochemical properties to traditional meat.
Cell proliferation is characterized by an increase in cell number, which is dependent on the ability of cells to produce clones/daughter cells as part of active mitotic cell division. This process is guided by cell signaling through growth factors and cytokines that mediate the expression and suppression of related genes. In vivo, growth factors and cytokines are released from secretory organs and cells [111]. Conversely, cell cultures rely on external sources such as animal sera, which remain undefined in terms of specific composition levels of growth factors and cytokines that promote cell proliferation [103,112]. Although animal sera have been regularly used, food-safe, ethically sourced, and inexpensive growth-promoting media components remain a priority in the cultured meat industry [88].
Table 2 presents recent reports from the past 5 years that have focused on increasing the proliferation and differentiation potential of cultured meat progenitor cells [112–131]. Numerous reports have focused on developing media components for cultured beef production compared to those for cultured porcine and chicken production. Generally, the beneficial effects of media additives used are based on the expression of myogenesis-related genes and proteins, such as myoblast determination protein 1 (MYOD), myogenin (MYOG), myosin heavy chain (MYHC), and desmin, determined via polymerase chain reaction (PCR), liquid chromatography-mass spectroscopy (LC-MS), western blotting, and enzyme-linked immunosorbent assay (ELISA; Table 2). These biomarkers provide insight into the status of cell proliferation and differentiation in vitro. The changes in their expression could provide clues on the effect and potential mechanisms/metabolic pathways involved.
| Target species | Component | Mechanism | References |
|---|---|---|---|
| Bovine | Insulin |
Increased proliferation marker expressions (e.g., Ki67, PCNA); Differentiation marker expression (e.g., MYOD, MYOG, MYH) related to myogenesis. |
[113] |
| Parthenolide (0.5 and 1 µM) and rutin (100 and 200 µM) for proliferation; Adenosine for differentiation | Reduced H2O2-induced cytotoxicity; Increased expressions of myogenic proteins (e.g., MyoD, MyoG, MHC RNA); prevention of muscle atrophy | [114] | |
| Engineered autocrine signaling | Increased FGF-2 and RasG12V production, leading to myogenic stability but reduced myotube formation | [115] | |
| Crude polysaccharides from brown seaweed (Ecklonia cava) hydrolysate |
Increased cell migration activity related to the HGF/MET pathway and FAK pathway; Improved differentiation marker expressions (e.g., MYH2, MYH7, MYOG) |
[116] | |
| Vitamin C and N-acetylcysteine |
Decreased oxidative stress and senescence; Activated Akt/mTOR/P70S6K signaling pathway |
[117] | |
| Citrus sunki peel extract | Increased gene expression (CEBPA, CEBPB, PPARG) and protein expression (FASN, CEBPB, PPARG) of fibro-adipogenic progenitor cells during culture | [118] | |
| Drone pupae extract | Increased differentiation and myotube formation based on increased differentiation markers (e.g., MYH2, MYOG, DES) | [119] | |
| Porcine | Quercetin, Icariin, 3,2′-Dihydroxyflavone |
Quercetin (50 nM) improved differentiation based on increased MyHC expression; 3,2′-dihydroxyflavone (10 μM) increased the expression of PAX7 related to proliferation and stemness |
[120] |
| Vitamin C | Activation of PI3K/AKT/mTOR pathway through the IGF-1 signaling | [121] | |
| Recombinant Porcine Fibroblast Growth Factor 1 | Activation of ERK-dependent phosphorylation of DRP1 at Ser616, resulting in improved mitochondrial function and proliferation | [122] | |
| Naringenin | Upregulation of IGF-1 signaling | [123] | |
| N-acetylcysteine | Upregulation of PI3K-AKT pathway | [124] | |
| Diarylheptanoid derivative | Regulation of Wnt/β-catenin signaling | [125] | |
| YAP and lysophosphatidic acid (LPA) |
Activation of YAP proteins using LPA increased proliferation; Regulation of Hippo-YAP signaling involved in cell contact inhibition |
[126] | |
| Fermented soybean meal and edible insect hydrolysates |
Promoted cell proliferation (0.01%–5% FAB-H and FB-H, 0.01%–1% TM-H, or 0.01%–0.1% GB-H); 50% FBS replacementmaintained cell proliferation and differentiation capacity (0.01% and 0.1% of FAB-H, FB-H, and TM-H) |
[127] | |
| Chicken | Glycyrrhiza uralensis crude water extract and licochalcone A and B |
Antioxidant activity during differentiation; Promoted cell proliferation and adhesion in spheroid culture |
[128] |
| Black soldier fly hydrolysates |
Upregulated myogenic markers (e.g., PAX7, NCAM1, MYF5, MYOD1); BLH50 promoted cell proliferation but decreased differentiation capacity with increasing BLH concentration |
[129] | |
| Bovine-Porcine-Chicken | Quercetin |
Upregulated myogenic gene and protein expressions (e.g., MYOD, MYOG, MYH); Inhibited MSTN, ACVR2B, and SMAD2/3, resulting in improved differentiation and myotube formation |
[130] |
| Laxogenin (LAX), Laxogenin-5-alpha-hydroxy-laxogenin (5HLAX) |
MSTN inhibition using 10 nM LAX and 5HLAX; Promoted myogenesis, and myotube formation and maturation; Antioxidant activity by serving as agonists for ROS-downregulating factors |
[131] |
PCNA, proliferating cell nuclear antigen; MYOD, myogenic differentiation protein 1; MYOG, myogenin; MYH, myosin heavy chain; MHC, myosin heavy chain; RNA, ribonucleic acid; FGF-2, fibroblast growth factor 2; HGF, hepatocyte growth factor; MET, MET proto-oncogene, receptor tyrosine kinase; FAK, focal adhesion kinase; MYH2, myosin heavy chain 2; MYH7, myosin heavy chain 7; mTOR, mechanistic target of rapamycin; CEBPA, CCAAT/enhancer binding protein alpha; CEBPB, CCAAT/enhancer binding protein beta; PPARG, peroxisome proliferator-activated receptor gamma; FASN, fatty acid synthase; DES, desmin; MyHC, myosin heavy chain; IGF-1, insulin growth factor 1.
Some commercial media components have shown potential in increasing the proliferation and differentiation capacity of bovine muscle cells. The use of insulin improved the expression of both proliferation (e.g., Ki67, proliferating cell nuclear antigen [PCNA]) and differentiation markers (e.g., MYOD, MYOG, MYHC) [111]. Insulin is commonly used for managing cellular glucose uptake in vivo and in vitro. Given the importance of glucose as an energy source for cellular functions, improved glucose uptake can assist in enhancing proliferation and differentiation in vitro [113,132]. Meanwhile, Yu et al. [114] reported that parthenolide and rutin can improve proliferation, while adenosine can be used to improve differentiation [114]. Using antioxidants, such as rutin, can reduce hydrogen peroxide (H2O2)-induced cytotoxicity and help prevent muscle atrophy [114]. This can also be observed with vitamin C and N-acetylcysteine, which have been reported to reduce oxidative stress and senescence of bovine cells [117]. Food-grade and food-derived media additives have also been reported for cultured beef production. A common denominator among these media additives is their potential to improve differentiation and myotube formation, which are important considerations for the eventual buildup of muscle mass, rather than undifferentiated cell mass [116,118,119]. Among these additives, Citrus sunki peel extract was used on fibro-adipogenic progenitor cells, unlike other additives used in studies focused only on muscle progenitor cells [116].
In cultured pork production, similar media additives used for cultured beef can be observed, such as vitamin C and N-acetylcysteine, which confirms their effectiveness in culturing muscle cells [121,124]. A potent antioxidant, quercetin, has also been used in combination with icariin and 3,2′-dihydroxyflavone, with quercetin improving differentiation and 3,2′ dihydroxyflavone improving proliferation and stemness [120]. In cultured meat production, both cell proliferation and stemness are important for providing an ample number of cells for 3D culture (e.g., scaffolds, 3D printing, microcarriers) to achieve traditional meat-like characteristics or to increase cell mass in suspension cultures for hybrid cultured meat products. Lysophosphatidic acid has been used to regulate the Hippo-Yes-associated protein (YAP) signaling pathway, which is involved in cell contact inhibition, allowing increased proliferation of porcine muscle cells [126]. Some natural products, such as naringenin and diarylheptanoid derivatives, have been used to regulate insulin growth factor 1 (IGF-1) signaling and Wnt/β-catenin signaling, respectively [123,125]. Both signaling pathways are closely related to cell proliferation and differentiation and are commonly used as targets for improving cultured meat production.
There are limited studies on improving cultured chicken production through the use of media additives. Lee et al. [128] utilized Glycyrrhiza uralensis crude water extract and licochalcone A and B in culturing chicken muscle progenitor cells, which resulted in improved cell proliferation and adhesion in spheroid cultures, as well as enhanced antioxidant activity during proliferation [128]. Another study determined that black soldier fly hydrolysates can upregulate myogenic proliferation and differentiation markers [129]. However, it was concluded that there is an inverse effect on the differentiation capacity of cells cultured with increasing concentrations of black soldier fly hydrolysates.
Some studies used multiple livestock cells to prove their applicability in cultured meat production. Similar to the results of Guo et al. [120] on quercetin use, Ahmad et al. [130] demonstrated the ability of quercetin to inhibit myostatin (MSTN), activin type-2B (ACVR2B), and SMAD2/3, leading to improved differentiation and myotube formation of bovine, porcine, and chicken muscle cells [130]. Laxogenin (LAX) and 5-alpha-hydroxy-laxogenin (5HLAX) can also inhibit MSTN expression, effects similar to those of quercetin [131]. Considering all the reviewed reports, the use of antioxidants (e.g., quercetin, LAX, 5HLAX, G. uralensis crude water extract, licochalcone A and B, naringenin, vitamin C) in cultured meat production generally favors cell proliferation, differentiation, myotube formation, and prevention of muscle degradation (Table 2). This highlights the importance of antioxidants as components of media formulation for cultured meat production. It also recognizes the general considerations for improving mitochondrial functions, given that mitochondria are directly involved in the generation and accumulation of reactive oxygen species (ROS).
CRITICAL PERSPECTIVES ON SHORTENING THE DURATION OF TRADITIONAL AND CULTURED MEAT PRODUCTION
Reducing the time required for conventional meat production through improved breeding methods, better feeding schedules, and faster growth technology has a number of effects on the economy, the environment, ethics, and human health. Livestock raisers can increase turnover rates, cut production costs, and increase profitability by shortening the time it takes for animals to reach market weight. This allows for more meat to be produced in the same amount of time, which could stabilize prices and increase customer availability. The effects on the ecosystem, however, should still be monitored and considered. Another benefit of shorter production cycles is that they can result in lower total resource inputs per unit of meat produced, including feed, water, and land. However, the use of antibiotics, growth hormones, and concentrates, which are frequently linked to faster meat production, can result in increased greenhouse gas emissions per animal, water pollution, as well as increased risk of antibiotic resistance [33,40,53]. Ronquillo & Hernandez [64] extensively reviewed the potential environmental impacts of AGPs and HGPS citing numerous reports that support how excessive and unmetabolized growth promoters can lead to environmental pollution and antibiotic resistance [64]. Furthermore, the change in gut microbiome due to AGP use could result in increased methane emissions [133].
Rapid meat production generates serious ethical questions regarding the well-being of animals [134]. The quality of life of live animals is sometimes compromised by intensive farming methods intended to enhance development, which result in cramped living quarters, restricted mobility, and increased disease susceptibility [135]. Animals raised for fast growth may also experience biological stress, which can result in persistent pain, organ failure, and physical abnormalities. The misuse and overuse of antibiotics to accelerate animal growth and prevent disease, even in healthy animals, have contributed to antimicrobial resistance, which poses major health hazards to both animals and humans [39,40]. Furthermore, the nutritional composition of meat from animals bred under such rigorous settings may differ from that of meat from animals raised more slowly and naturally [136,137]. Therefore, reducing the time needed to produce meat may have some logistical and financial benefits, but it also presents challenges pertaining to environmental sustainability, animal welfare, and public health [39,40].
Similar to traditional meat production, reducing production time in cultured meat production can improve the ecological efficiency of cultured meat compared to both traditional and slower cellular agriculture methods by lowering the energy, water, and nutritional inputs needed per unit of meat [138,139]. This can hasten its potential to lower water use, land use, and greenhouse gas emissions, making it a more practical approach to addressing the environmental issues associated with traditional meat production. Moreover, shorter production cycles translate into faster scaling, lower operational costs per batch, and increased market competitiveness. These factors may help cultured meat overcome one of its main obstacles, which is high production costs. As a result, consumers may find it easier to obtain, which might promote broad adoption and acceptability.
Although these developments can increase productivity, they also raise concerns about the long-term safety and nutritional value of the finished product. For instance, tissue structure, flavor development, or nutrient composition may be impacted by the components used for cell culture, necessitating further processing or formulation to satisfy consumer demands. However, the need for accelerated growth must also consider consumer perception, regulatory supervision, and biosecurity, particularly when using synthetic chemicals or novel growth factors [140]. Cutting the time needed to produce cultured meat could greatly forward the goal of developing scalable, sustainable, and ethical meat substitutes. However, in order to achieve long-term success, quality, safety, and public opinion must be carefully considered.
CONCLUSION
The benefits and drawbacks of reducing the time it takes to produce both traditional and cultured meat are complicated and represent larger concerns about public health, ethics, and food sustainability. Accelerated timelines in traditional meat production may increase profitability and efficiency, but they frequently come at the expense of increased health hazards, environmental degradation, and animal welfare. Conversely, reducing the time required to produce cultured meat has the potential to lower resource consumption and improve scalability, but it still confronts obstacles related to technology, regulations, and consumer acceptance. The desire to produce meat more quickly—whether through traditional or cultured meat technology—must be directed by a well-rounded strategy that places equal emphasis on efficiency and speed as well as long-term sustainability and the integrity of the food chain.
