INTRODUCTION
In the swine industry, stress can be induced by factors such as nutrition, diseases, social environment, and biological changes [1]. In particular, weaning piglets can be exposed to various stresses, including transitioning to solid feed, separation from the sow, and adapting to new environmental changes [2,3]. Such stressors can have negative effects on feed intake, feed efficiency, immunity, and behavior [1]. Hierarchy fights among weaning piglets primarily occur due to the formation of new dominance relationships when piglets are mixed into groups [4]. Reducing the fights is crucial because high levels of aggression can lead to increased injury and stress, compromising pig welfare and growth [4]. In this context, serotonin, a hormone secreted in the central nervous system, can regulate stress [5]. Increasing serotonin expression in the hypothalamus can decrease cortisol levels, making it crucial to induce appropriate serotonin secretion during periods of stress [6,7].
The introduction of multi-suckling systems has led to a continuous increase in litter sizes [8]. With larger litters, there is an elevated risk of maternal stress and heightened nutritional demands to support increased milk production. Therefore, it is crucial to enhance feed intake and provide optimal nutrient levels in the diet of lactating sows to minimize maternal weight loss and improve subsequent reproductive performance. Moreover, weaning mortality factors include diseases, starvation, and crushing, with crushing being a significant contributor [9,10]. Among the factors contributing to pre-weaning mortality, crushing is the leading cause of death [10]. Sows confined within farrowing crates may experience heightened anxiety and agitation during stressful situations, leading to increased frequencies of lying and standing behaviors. This behavioral pattern can elevate the risk of piglet crushing.
Tryptophan is known as a precursor substance of serotonin and melatonin, both of which play crucial roles in regulating emotions and aggressive behavior during stressful situations [11–13]. Serotonin (5-hydroxytryptamine [5-HT]) was found to regulate affective states [14] such as anxiety [15] and aggression [16]. Serotonergic neurons arise from the raphe nuclei located in the brainstem and project throughout different brain regions involved in mood regulation and cognitive functions like stimulus perception, evaluation, and behavioral responses. In both vertebrates and invertebrates, 5-HT systems have been demonstrated to influence cognitive and behavioral functions. Commonly known as the “happiness hormone,” as it participates in stress responses and mood disorders on various levels. In this context, serotonin, a hormone secreted in the central nervous system, plays a crucial role in stress regulation. Increased serotonin expression in the hypothalamus can lower cortisol levels, highlighting its importance in managing stress responses. Additionally, serotonin stimulates the expression of ghrelin, a hormone that enhances appetite, thereby promoting increased food intake and feed consumption [17]. Given its diverse functions, serotonin demand may rise during periods of prolonged stress in animals. In this study, our aim is to assess the effects of different levels of tryptophan supplementation in the diets of weaning pigs and lactating sows on performance parameters, hormone levels, and behavioral traits.
MATERIALS AND METHODS
A total of 144 weaning pigs (Landrace × Yorkshire × Duroc) with initial mean body weight = 6.63 ± 0.21 kg were allotted to three treatment groups with six replicates per group of eight pigs per replicate in a completely randomized design. The study was carried out using a randomized complete block design at the Research Center of Animal Life Sciences, Kangwon National University. The treatments included a control group (CW, basal diet based on National Research Council [NRC] requirements), TW6 (0.06% tryptophan supplementation + basal diet), and TW12 (0.12% tryptophan supplementation + basal diet). The tryptophan used in this study is a 50% tryptophan produced by Daesang Corp. The experimental diets were supplemented for 28 d in two phases: phase 1 (days 0–14) and phase 2 (days 15–28). The pigs were housed in group pens with partially slatted concrete floors measuring 2.80 × 5.00 m each. Each pen was equipped with a self-feeder and nipple drinker to provide ad libitum access to feed and water. Basal diets were composed of a corn-soybean meal formulated as recommended by the NRC to meet or exceed the nutritional requirements of weaning pigs (Table 1).
Supplied per kg of diet: 2,200 IU vitamin A (palmitate), 2.00 mg vitamin B1 (thiamin), 5.50 mg vitamin B2 (riboflavin), 7.00 mg vitamin B6 (pyridoxine), 0.02 mg vitamin B12 (cyanocobalamin), 30.00 mg vitamin B3 (niacin), 10.3 mg vitamin B5 (pantothenic acid), 0.32 mg folic acid, 0.06 mg biotin, 5.00 mg ethoxyquin, 220 IU vitamin D3 (cholecalciferol), 75.0 mg vitamin E (dl-α-tocopheryl acetate), 0.53 mg vitamin K3 (menadione).
A total of 40 lactating sows (Landrace × Yorkshire) were allotted to four treatment groups with ten replicates per group of one pig per replicate in a completely randomized design. The study was carried out using a randomized complete block design at the Research Center of Animal Life Sciences, Kangwon National University. The treatments included a control (CL) group (basal diet based on NRC requirements), TL3 (0.03% tryptophan supplementation + basal diet), TL6 (0.06% tryptophan supplementation + basal diet), and TL9 (0.09% tryptophan supplementation + basal diet). The tryptophan used in this study is a 50% tryptophan produced by Daesang. The experimental diets were supplemented for 21 d in one phase during lactation. The experiment was initiated from 112 days of gestation, and the sows were fed the experimental diets from parturition to weaning. The sows were housed individually in farrowing crates equipped with identical farrowing rails (2,400 mm in length and 800 mm in width) in accordance with their treatment groups. Litter performance was evaluated post-partum. Each farrowing crate was equipped with insulation boxes and heat lamps for the piglets. Basal diets were composed of a corn-soybean meal formulated as recommended by the NRC to meet or exceed the nutritional requirements of lactating sow. The chemical composition of the formulated experimental diets is presented in Table 1 and was prepared in powdered form. The experimental diets were fed twice daily at 07:00 and 16:00. Water was provided ad libitum, and no other additives or medications were used.
Each experimental pig was individually weighed on the first and last day of the experiment. Throughout the entire experiment, feed consumption was monitored. This data was then used to determine the average daily gain (ADG), average daily feed intake (ADFI), and gain-to-feed ratio (G:F) at the end of the experiment.
The weight and backfat thickness of the sows were measured individually immediately post-partum and at the weaning time point. Changes in sow weight and backfat thickness during the lactation period were calculated as the difference between measurements taken at 112 days of gestation and at the weaning time point. Backfat thickness was measured using an ultrasound device (Anyscan BF, SONG KANG GLC) by taking three measurements at 6.5 cm from the 10th rib of each sow and calculating the average value. ADFI was determined by monitoring feed intake during the lactation and estrus recurrence periods. Litter performance was evaluated by measuring sow weight and litter size at farrowing and at weaning for each individual sow.
Before being placed in the experimental pen, each pig underwent a pre-mixing lesion assessment, and a spray mark was applied to differentiate them within the experimental group. Fourteen days after mixing, the lesion score was recorded again, from which the pre-mixing lesion score was subtracted to determine the number of lesions resulting from the first 14 days post-mixing. Twenty-eight days after mixing, the lesion score was recorded once more, and the lesion score from day 14 was subtracted to determine the number of lesions that developed between days 14 and 28.
Skin lesion scores were assessed by the same observer on the first day, as well as on days 14 and 28. The observer classified the overall skin lesion status within the pen using a scoring system ranging from 1 to 3 (score 1: scratches less than 2 cm in length; score 2: scratches between 2 and 5 cm in length; score 3: scratches longer than 5 cm).
In experiment 1, blood samples were collected from a total of 18 piglets, with 3 piglets selected per replicate, corresponding to the average weight at the end of the experiment. Blood (5 mL) was drawn from the jugular vein and immediately transferred to the laboratory. In experiment 2, blood samples were collected from the ear vein, with 5 mL per sow, from a total of 40 sows. The collected blood was centrifuged at 3,000×g and 4°C for 15 minutes to separate the plasma, which was then stored at –20°C until analysis. Growth hormone (#MBS026403, Mybiosource), cortisol (#MBS268128, Mybiosource), and serotonin (#MBS1600878, Mybiosource) levels in the blood were analyzed using ELISA kits following the manufacturer’s instructions.
For hair cortisol analysis, a portion of the dorsal hair was removed from the sows at farrowing, and at the end of the experiment, newly grown hair from the same area of each individual was collected for cortisol analysis. Hair samples were washed and cortisol was extracted according to the method described by Ataallahi et al. [18]. The extracted samples were measured using a Cortisol ELISA kit (ADI-900-071, Enzo Life Sciences).
Behavioral traits of the weanling pigs (experiment 1) and lactating sows (experiment 2) were meticulously observed using high-definition cameras (FIX extreme action cam pro 4K UHD, XAC-502, Fix) that were strategically installed at the center of the ceiling for weanling pigs or at the center of the ceiling for lactating sows. This setup ensured comprehensive coverage of the pen, eliminating any blind spots and thereby providing an unobstructed view of all the pigs. The behavior of the pigs was systematically recorded on video twice a day, specifically during two three-hour periods from 06:00 to 09:00 and from 16:00 to 19:00. These observations were conducted at 1–7, 14, 21, and 27 days old age for piglets and 1, 7, 14, and 20 days post-lactation for lactating sows. Unlike studies that rely on automated software to extract behavioral data, this study employed a more hands-on approach. A single dedicated observer meticulously watched all the recorded videos, ensuring consistency and accuracy in the data collection process. For each observed behavior, the number of weaning pigs engaged in that behavior was counted. Additionally, the observer recorded the duration of time that the pigs spent on each specific behavior. The analysis of these behaviors was conducted in accordance with the method outlined by Lee et al. [19]. For piglets, we defined standing, standing on all four legs; lying, lying on the side and sternum; belly nosing, a repeated rhythmic up-and-down massage of the snout on a pen-mate’s mid-section; tail biting, taking the tail of another pig into the mouth; ear biting, taking the ear of another pig into the mouth; aggressive interaction, engaging in agonistic interaction - pushing, biting, and head-knocking with another pig. For lactating sows, we defined standing, standing on all four legs; lying, lying down, either on the belly or on one side; sitting, dog-sitting with rump on the floor and shoulders raised up with front legs extended; feeding, lactating time for suckling pig; stereotype, sham-chewing, bar biting and head-knocking with another pig.
In experiment 1, data generated in the present study were subjected to statistical analysis using the one-way ANOVA method in SAS (SAS Institute) with a completely randomized design. Statistical significance between treatment groups was assessed using Tukey’s Honestly Significant Difference test. Replicate from each pen was implemented as the experimental unit for all measurements. Differences were deemed significant when the p-value was below 0.05. In experiment 2, orthogonal polynomials were employed to analyze the linear and quadratic impacts of varying dietary tryptophan levels and analyze the p-value of the one-way ANOVA method for cortisol and serotonin (SAS Institute). Replicate from each pen was implemented as the experimental unit for all measurements. A p-value of less than 0.05 was considered indicative of significant differences.
RESULTS
There was no significant difference in the final BW based on the level of tryptophan supplementation, there was a tendency (p = 0.092) for higher final BW in the TW12 compared with the CW (Table 2). In phase 1, average daily gain and feed efficiency significantly increased (p < 0.05) at TW12 compared with CW. Additionally, in phase 2, average daily gain (p = 0.089), overall average daily gain (p = 0.069), and feed efficiency (p = 0.068) showed a tendency to tryptophan supplementation.
Although there was no significant improvement in the severity of skin lesions with increasing levels of tryptophan supplementation in phase 1, the total skin lesion counts were significantly lower (p < 0.05) in the TW12 group compared to the CW (Table 3). In phase 2, there was no significant improvement in the severity of skin lesions and the total skin lesion count with increasing levels of tryptophan supplementation.
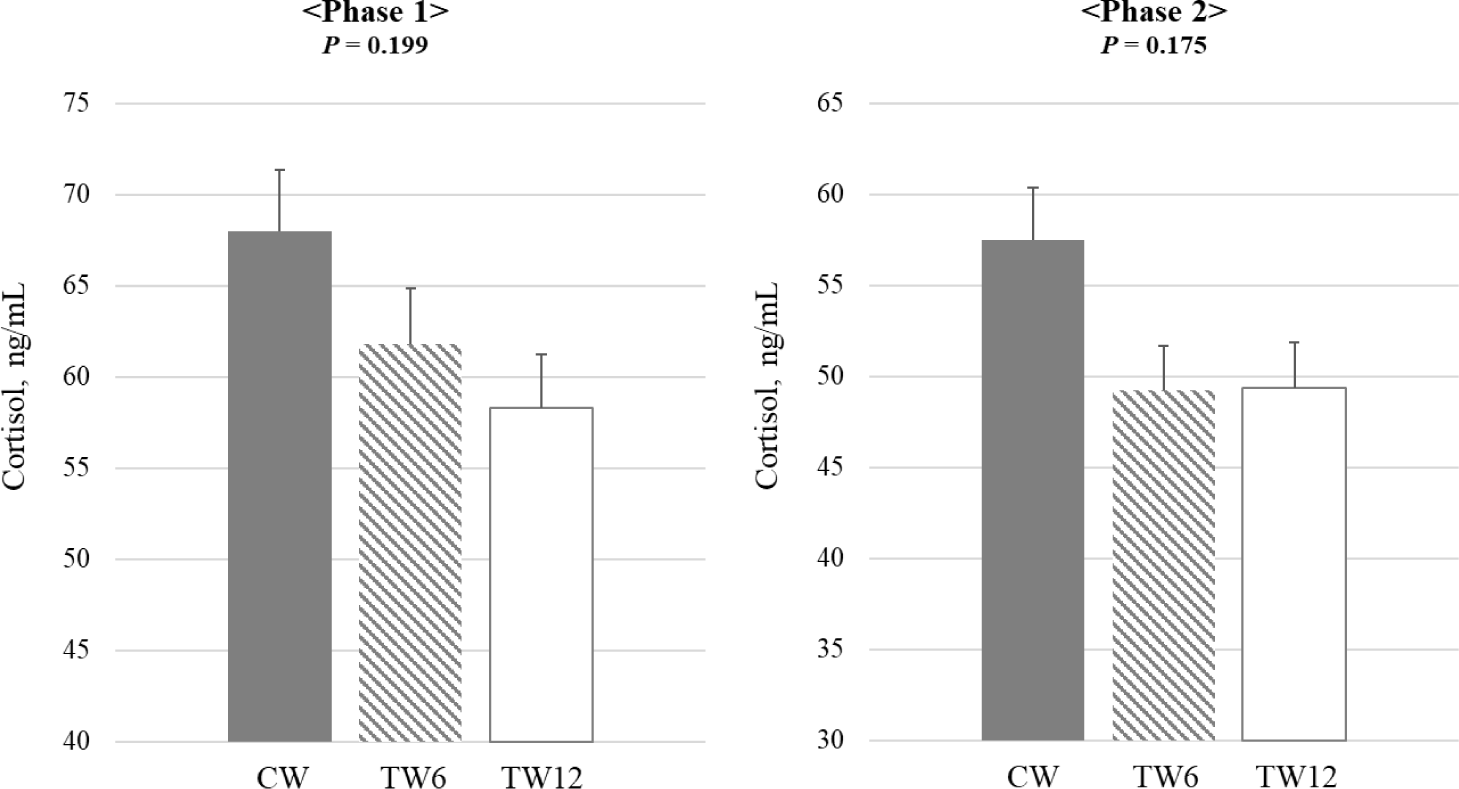
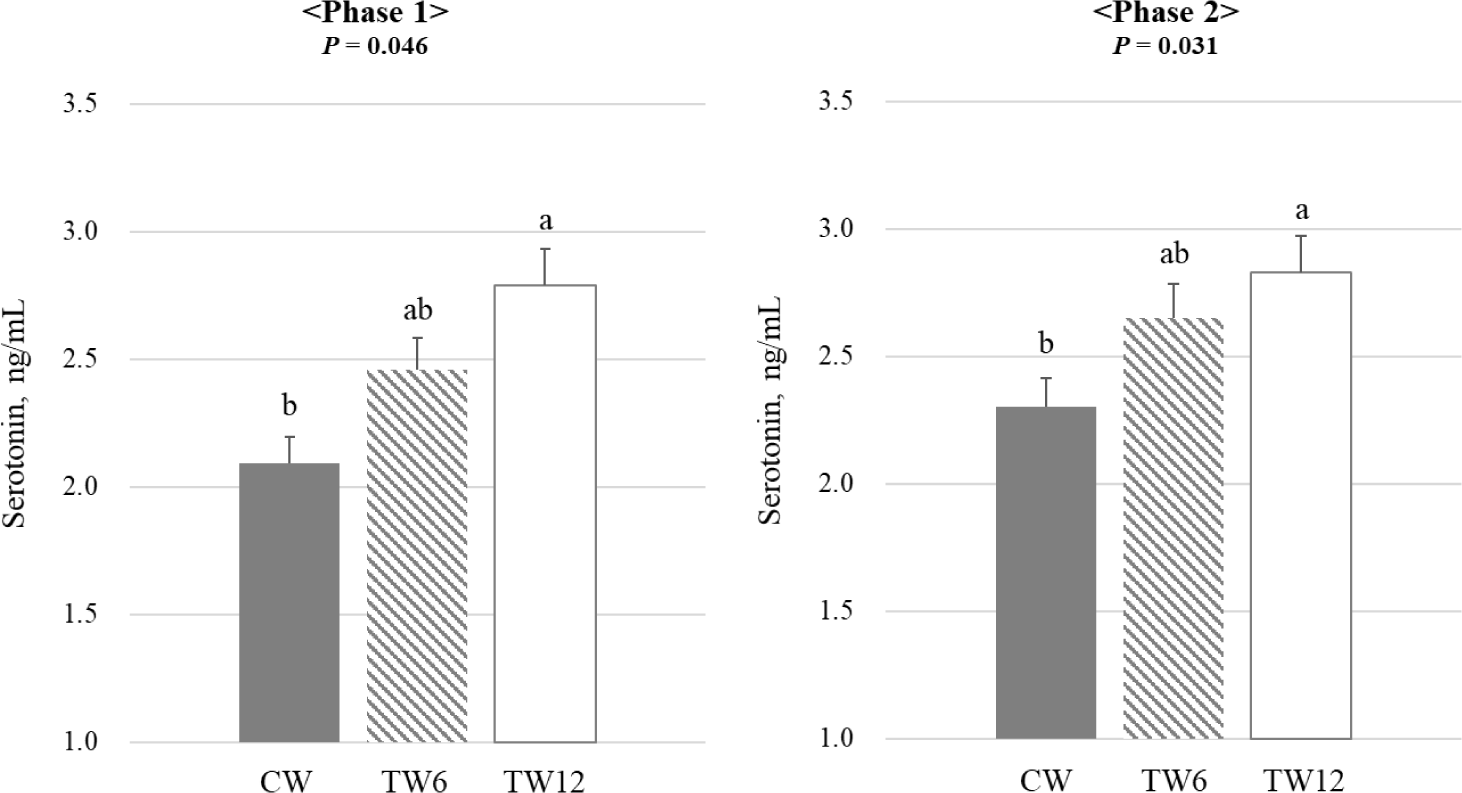
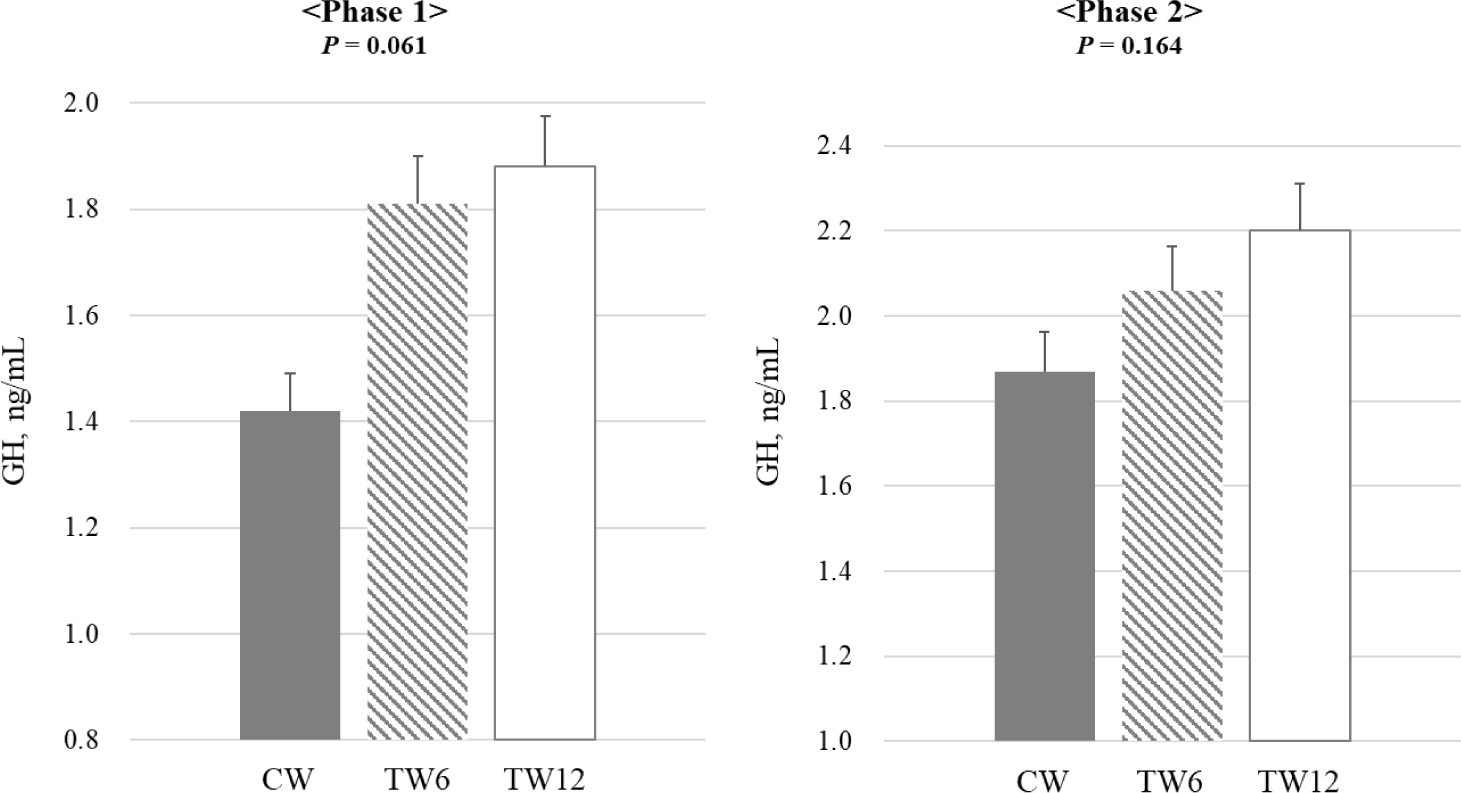
Fig. 1 illustrates the impact of tryptophan supplementation on the blood cortisol levels of weaning pigs. The study found no statistically significant difference in the blood cortisol levels between phases 1 and 2. Fig. 2 illustrates the impact of tryptophan supplementation on serotonin levels in weaning pigs. During both phases 1 and 2 of the study, serotonin levels were significantly higher (p < 0.05) in the TW12 group compared to the CW. This indicates that tryptophan supplementation led to increased serotonin levels in the weaning pigs across both phases of the study. Fig. 3 illustrates the effect of tryptophan supplementation on growth hormone levels in weaning pigs. In phase 1 of the study, there was a trend (p = 0.061) towards higher growth hormone levels when tryptophan was supplemented compared to the CW. However, this difference did not reach statistical significance. In phase 2, there was no significant difference observed in growth hormone levels between the tryptophan-supplemented group and the CW.
With the addition of tryptophan, the lying behavior of weaning piglets was significantly higher (p < 0.05) in the TW6 group and TW12 group compared to the CW (Table 4), while ear biting tended to decrease (p = 0.05) in the TW12. There was a tendency (p = 0.067) for a decrease in aggressive interactions when tryptophan was supplemented.
There were no significant differences in weight changes of lactating sows based on the level of tryptophan supplementation in the feed (Table 5). However, during the lactation period, there were linear and quadratic decreases (p < 0.05) of backfat loss. Additionally, average daily feed intake was linearly increased (p < 0.05) with supplementation of tryptophan.
The results regarding the effects of tryptophan supplementation levels in the diet of lactating sows on litter performance is presented in Table 6. There were no significant differences in the number of piglets born alive, piglet birth weight, and litter weight based on the level of tryptophan supplementation in the feed.
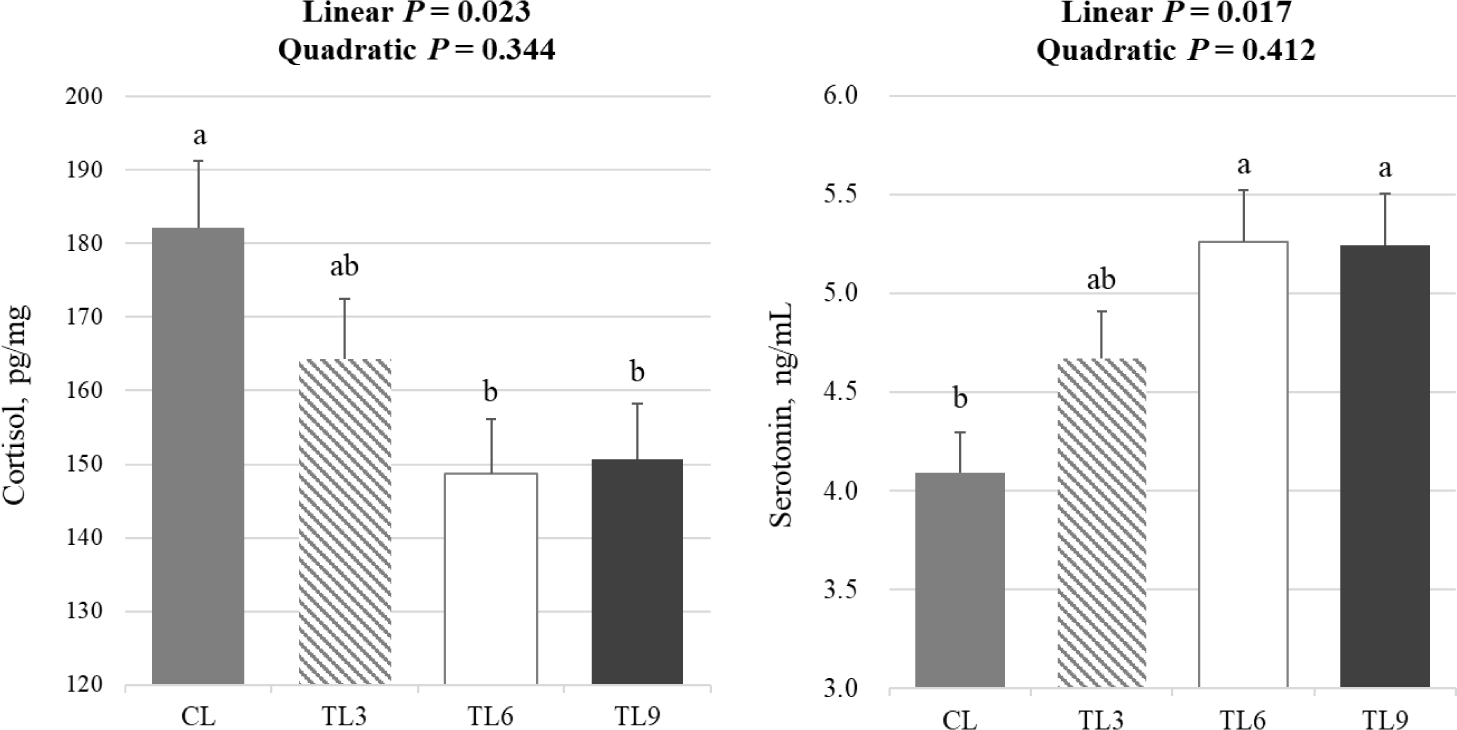
The concentration of hair cortisol linearly decreased (p < 0.05) with the addition of tryptophan, while the concentration of serum serotonin linearly increased (p < 0.05) with the supplementation of tryptophan (Fig. 4). Also, Cortisol levels were significantly lower (p < 0.05) in the TL06 and TL09 group compared to the CL group. Serotonin levels were significantly higher (p < 0.05) in the TL06 and TL09 groups compared to the CL group.
The results regarding the effects of tryptophan supplementation levels in the diet of lactating sows on behavioral traits are presented in Table 7. With increasing levels of tryptophan, the lying behavior of lactating sows linearly increased (p < 0.05), while stereotypic behavior linearly decreased (p < 0.05).
DISCUSSION
In the swine industry, stress from nutrition, disease, and social environment negatively impacts feed intake, growth, immunity, behavior, and welfare [1,4,20]. The improvement in growth performance with the addition of tryptophan in feed as shown in our study may be due to tryptophan serving as a precursor to serotonin. Serotonin is known to regulate functions such as immune response, stress hormone secretion, and reduction of aggressive behavior [13,16]. Particularly, serotonin role in emotional regulation may decrease social competition in pigs, thus positively impacting growth performance. Increasing the dietary tryptophan level from 0.18% to 0.28% linearly increases the concentration of serotonin in growing pigs [12]. Rao et al. [21] demonstrated significant improvements in final body weight, daily weight gain, daily feed intake, and feed efficiency in pigs with tryptophan supplementation in feed. The lower total skin lesion in pigs fed tryptophan is consistent with Henry et al. [22] who reported a significant improvement in skin lesions with the addition of tryptophan in growing pig feed. Moreover, the increase in lying behavior with tryptophan supplementation is in line with Koopmans et al. [11] who reported the supplementation of 0.5% in weaned pigs increased the lying behavior and decreased the standing behavior. Oglodek [23] observed that exposure to stress results in a decrease in tryptophan and serotonin levels and an increase in cortisol compared to normal conditions. Supplementing tryptophan to piglets can regulate cognitive and emotional functions, potentially reducing cortisol responses to stress, as evidenced by decreased cortisol levels in growing pigs with tryptophan supplementation [24]. Furthermore, the concentration of blood growth hormone in pigs tended to be higher in tryptophan-supplemented treatments. Activation of serotonin receptors in the pituitary gland can increase the release of growth hormone-releasing hormone and subsequently increase growth hormone levels [25]. When tryptophan was administered to cattle, leading to an increase in serotonin receptors in the cerebrospinal fluid, there was a significant increase in plasma growth hormone [7,25,26], suggesting that tryptophan supplementation in feed can increase growth hormone levels in weaning pigs, as they exhibit similar metabolic actions as mammals. Although an increase in serotonin in the hypothalamus has been shown to decrease cortisol levels [5–7], this study blood analysis revealed that while serotonin levels increased, there was no significant difference in cortisol levels. These findings are consistent with the improvement in aggressive behavior observed with tryptophan supplementation in feed. The ADG, G:F, total skin lesion, and serotonin level were improved in the first 2 weeks (phase 1), which highlights the supplementation of Trp in the diet in phase 1, where the weaning level is much higher, rather than weeks 3 to 4 (phase 2).
The introduction of multi-suckling systems has increased litter sizes, leading to higher maternal stress and nutritional demands [8,27]. Enhancing feed intake and optimizing nutrient levels in lactating sows’ diets is crucial to reducing maternal weight loss and improving reproductive performance. A previous study showed that feed intake and piglet preweaning mortality were decreased when the SID Trp level in multiparous sow diets increased to 0.23% [9]. This study evaluated the effects of tryptophan supplementation level in swine feed on the sow and litter performance, hormones, and behavioral traits of lactating sows. The improvement in feed intake and reduction of stereotypic behavior with the addition of tryptophan in feed as shown in our study appears to be due to tryptophan serving as a precursor to serotonin. This serotonin, in turn, stimulates the expression of the appetite-stimulating hormone ghrelin, thereby increasing feed intake in pigs [17]. Tryptophan and serotonin can promote the expression of the appetite-stimulating hormone ghrelin, which is known to increase appetite in animals [28]. In the case of pigs, it has been reported that injection of ghrelin can lead to increased feed intake and body weight [17]. The findings of this study suggest that the enhanced backfat thickness can be linked to feed intake during the lactation period. Also, the addition of tryptophan at different levels increases serotonin in the body through the serotonin pathway [29]. Tryptophan is metabolized through the kynurenine pathway, serotonin pathway, and indole pathway [30]. The conversion of tryptophan into serotonin and subsequently into melatonin shows its role in modulating circadian rhythms, stress responses, and behavioral patterns in pigs [11,31]. Among these, the serotonin pathway can lead to increased serotonin levels in the body with tryptophan supplementation [29]. Serotonin is known to regulate affective states such as anxiety and aggression [14–16]. By supplementing tryptophan, the metabolic flux towards serotonin and melatonin pathways is enhanced, improving neurochemical balance and mitigating the adverse effects of stress on behavior and feed intake [11]. Furthermore, in stressful situations, increase in serotonin in the hypothalamus when different levels of tryptophan were administered to growing pigs [32], which is consistent with the findings of this study. Among pre-weaning mortality factors, crushing is the leading cause of death [10]. It was predicted that an increase in the sow’s lying behavior would reduce pre-weaning mortality, but there was no significant difference in mortality rates observed. The addition of tryptophan was found to decrease stereotypic behaviors in lactating sows. Li et al. [33] reported improvements in aggressive interactions, specifically head-to-head knocking and head-to-body knocking, when adding 0.20% and 0.27% tryptophan during the gestation and lactation periods of sows. This finding is consistent with the results of this study.
CONCLUSION
The improvement in growth performance with the addition of tryptophan in feed as shown in our study appears to be due to tryptophan serving as a precursor to serotonin. Based on the findings of this study, it can be inferred that supplementing feed with tryptophan may improve performance metrics and mitigate skin lesions during the early weaning period. Additionally, the inclusion of tryptophan could potentially decrease aggressive behaviors observed during the weaning period. Also, the addition of tryptophan in swine feed can increase feed intake and reduce stereotypic behaviors during lactation.
