INTRODUCTION
In modern poultry production, optimizing the health and productivity of laying hens is important to achieving sustainable and profitable outcomes [1]. Amino acids are essential components of protein synthesis and play a crucial role in the growth, maintenance, and overall well-being of poultry [2–4]. Among these, tryptophan (Trp) is an essential amino acid, which is a precursor of serotonin [5,6], a neurotransmitter that regulates mood, behavior, and stress responses [7,8]. It also contributes to the synthesis of melatonin and regulates circadian rhythms and immune responses [9]. The importance of Trp becomes evident in commercial poultry systems, where laying hens are often exposed to stressors such as high stocking densities in battery cages, limited mobility, and the physiological demands of sustained egg production [2,10,11]. These stressors, exacerbated in late production stages as hens age, can lead to heightened physiological strain, fatigue, and adverse behavioral and physiological responses [12]. Such responses include elevated corticosterone levels, impaired immune function, and reductions in egg quality [1–3,10]. The Trp role in serotonin production suggests it may help modulate stress responses and improve welfare. Addressing these challenges is critical for ensuring the welfare and productivity of hens, as well as maintaining egg quality and food safety. Given its role in serotonin synthesis, Trp supplementation may offer a nutritional strategy to mitigate stress and enhance overall hen welfare [6].
Although the stress-reducing properties of Trp have been acknowledged in poultry research, its effects on both stress biomarkers and key production parameters under high-stocking-density conditions remain incompletely understood. This study aims to address these gaps by investigating the effects of dietary Trp supplementation on laying hens at late production stages. The research focuses on egg production, yolk color, and shell thickness, alongside physiological and biochemical indicators such as lymphocyte count, triglyceride levels, lactate dehydrogenase activity, and corticosterone concentrations.
MATERIALS AND METHODS
The experiment utilized 700 Hy-line laying hens, acquired at 68 weeks of age, with an average body weight of 2.01 ± 0.16 kg. The study lasted for four weeks, following a two-week acclimatization period during which baseline data on egg production, egg weight, egg quality, and general health were recorded. At 70 weeks, the hens were randomly allocated to one of four dietary treatments: 0%, 0.25%, 1%, and 2% Trp supplementation, with each treatment replicated five times and each replicate consisting of 35 hens. The diets were based on a corn-SBM mixture (Table 1) and prepared at the university’s facility using a horizontal feed mixer (1,200 kg capacity, 1 hp motor, KH super 15. H.P). To avoid cross-contamination, the control diet was mixed first, followed by the addition of Trp for the other groups. All feed was provided in mash form and was labelled with the preparation date, treatment code, and net weight. No coccidiostats, growth promoters, or antibiotics were included. The diet met the nutritional requirements specified by the Hy-line brown breeding company. Feed and water were provided ad libitum. The hens were housed in a temperature-controlled, windowless environment maintained at 20°C–22°C, with a 16-hour light/8-hour dark photoperiod. Enrichments, including perches and nesting areas, were provided in accordance with EU regulations. Each pen was equipped with 14 nipple drinkers (Big Dutchman AG), a feeding trough providing 15 cm of space per hen, and a claw-shortening device, all housed within a 6.19 m² area (2.25 × 2.75 m) providing 1,767 cm² per hen. The flock and facilities were monitored three times daily, at 9 am, 4 pm, and 8 pm.
Provides per kilogram of diet: vitamin A, 10,000 IU; cholecalciferol, 2000 IU; vitamin E, 0.25 IU; vitamin K3, 2 mg; vitamin B12, 10 mg; choline, 250 mg; folacin, 1 mg; niacin, 30 mg; pantothenic acid, 10 mg; pyridoxine, 3 mg; riboflavin, 6 mg; thiamine, 2 mg; ethoxyquin, 125 mg; Co, 0.3 mg; Cu, 10 mg; Fe, 60 mg; I, 0.5 mg; Mn, 40 mg; Se, 0.2 mg; Zn, 50 mg.
Performance metrics, including feed intake (FI) and body weight, were recorded for 4 weeks. Hen-day egg production (HDEP) was calculated by dividing the total number of eggs produced by the number of hens alive during the period, then multiplying by 100. Egg quality assessments, including Haugh units, yolk and albumin weights, yolk color, average egg weight (AEW), and albumin height, were conducted using an egg multi-tester (Tohoku Rhythm). Egg mass was determined as the product of HDEP percentage and AEW. The feed conversion ratio (FCR) was calculated by dividing the average daily FI by AEW. Albumin and yolk percentages were calculated as albumin weight or yolk weight divided by AEW, multiplied by 100. Eggshell strength was measured using a type II eggshell force gauge (Robotmation), while shell thickness was measured with a dial pipe gauge (Ozaki MFG), focusing on the sharp and round edges, and the midsection, excluding the membrane. The eggshell color was assessed with a Chroma Meter CR-400 (Minolta) using the CIE color system for CIE L*, CIE a*, and CIE b*.
At the end of the experiment, blood samples (10 mL each) were collected from six hens per replicate via the wing vein [13]. Blood was drawn into non-treated vacuum tubes, left at 25°C for serum separation, and then centrifuged at 3,000×g for 15 min at 4°C. The serum was stored at –20°C until analysis. Serum metabolites, including total cholesterol, triglycerides, glucose, total protein, aspartate aminotransferase, alanine transaminase, creatinine, albumin, immunoprecipitation, lactate dehydrogenase, and insulin, were analyzed using commercial kits (Fujifilm) on an automated chemistry analyzer (Fuji Dri-chem 3500i, Fujifilm). Calcium levels were measured colorimetrically using a biochemical analyzer (Hitachi modular system, Hitachi). Corticosterone levels were determined using an ELISA kit (Enzo Life Sciences). On day 28, additional blood samples were taken to analyze white blood cell (WBC) and red blood cell (RBC) counts using Natt-Herrick solution. Hemolysis-free serum was stored at –80°C for further analysis. WBC, heterophils, lymphocytes, monocytes, neutrophils, eosinophils, and basophils counts were conducted using the Hemavet® Hematology System (CDC Technologies). Hemoglobin concentration was measured using the cyanmethemoglobin method, and hematocrit was assessed via the microhematocrit method. Mean corpuscular volume, mean corpuscular hemoglobin, and mean corpuscular hemoglobin concentration were analyzed using a hematology analyzer (Fuji Dri-chem 3500i, Fujifilm).
Data analysis was performed using the Statistical Analysis System (SAS Institute, 2012). The pen, containing layer hens per treatment, served as the experimental unit for production performance and egg quality, and individual layer was considered as experimental unit for blood parameters and hormones. Tukey’s multiple range test was used to detect differences in blood corticosterone and serotonin levels. Linear and quadratic orthogonal polynomial contrasts were applied to analyze performance parameters, blood metabolites, and egg quality data. Results were reported as means and standard deviations, with significance thresholds set at p < 0.01 and p < 0.05.
RESULTS
In the period from 0 to 4 weeks of experiment, the HDEP showed an increasing quadratic response (p < 0.01) up to a maximum level of 84.7% with 1% Trp supplementation and decreased to 75.6 thereafter with 2% Trp supplementation (Table 2). An increasing quadratic response (p < 0.01) was observed up to the level of 1% Trp inclusion for egg mass up to the level of 48.7% and decreased thereafter. The FCR showed a decreasing quadratic response (p < 0.01) up to a minimum ratio of 2.7 with 0.25% and 1% Trp supplementation and increased to 3 thereafter with 2% Trp supplementation. The effect of the levels of Trp on FI and mortality was not significant.
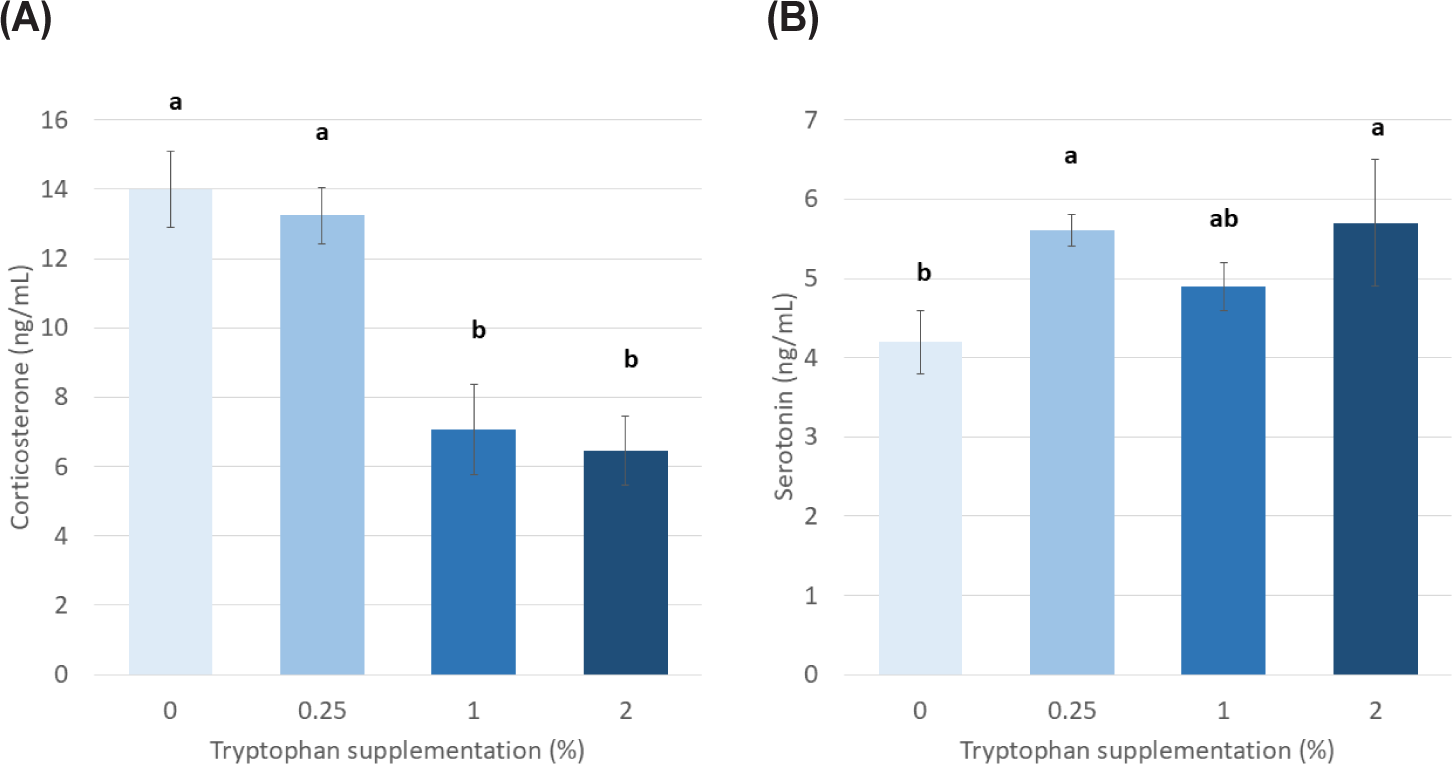
The blood content of WBC, heterophile (HE), lymphocyte (LY), and monocyte was linearly reduced by supplementation of Trp. However, the effect of the levels of Trp on HE/LY, eosinophil, and basophil was not significant (Table 3). A linear decrease in the content of blood RBC, hemoglobin, and hematocrit was observed with the supplementation of Trp, however, the content of mean corpuscular volume, mean corpuscular hemoglobin, and mean corpuscular hemoglobin concentration was unaffected. The concentration of triglyceride was linearly decreased, and an increasing quadratic response was observed up to the level of 1% Trp inclusion and decreased thereafter (Table 4). The blood content of glucose was linearly increased by supplementation of Trp. The effect of the levels of Trp on total protein, aspartate aminotransferase, alanine transaminase, calcium, creatine, and albumin was not significant, however, the concentration of immunoprecipitation and lactate dehydrogenase was linearly decreased with supplementation of Trp. The concentration of blood corticosterone was higher in laying hens fed 0% and 0.25% of Trp compared with 1% and 2% supplementation (Fig. 1A). The concentration of blood serotonin (Fig. 1B) was higher in laying hens fed 0.25% and 2% of Trp compared with 0% supplementation.
At week 2, the haugh unit showed a decreasing quadratic response (p < 0.05) up to a minimum of 79.2 with 0.25% Trp supplementation and increased thereafter with 1% and 2% Trp supplementation (Table 5). A decreasing quadratic response (p < 0.01) was observed up to the level of 0.25% and 1% Trp inclusion for yolk color and increased thereafter. The shell strength was linearly increased with supplementation of Trp. The effect of the levels of Trp on albumin height and shell thickness was not significant. The CIE a* of shell was linearly reduced and the CIE b* of shell was linearly increased with the supplementation of Trp, however, the shell CIE L* was unaffected. At week 4, the yolk color, shell strength, and shell thickness showed a linear increase (p < 0.05) with Trp supplementation, however, there was no difference in albumin height and haugh unit. The shell CIE L* showed an increasing quadratic response (p < 0.05) up to a maximum of 56 with 0.25% Trp supplementation and decreased to 55.2 thereafter with 1% and 2% Trp supplementation. The shell CIE a* and CIE L* were unaffected.
DISCUSSION
The observed quadratic response in HDEP and egg mass, peaking at 1% Trp supplementation, is consistent with the role of Trp in modulating physiological processes related to stress and serotonin synthesis [3,6]. Serotonin is a neurotransmitter that can be synthesized from Trp with significant impact on mood regulation and stress response [5,12,14]. In poultry, serotonin influences a range of behaviors, including feeding, aggression, and social interaction [6,8,9], which can directly affect feed intake and overall productivity. The peak in HDEP and egg mass at 1% Trp supplementation suggests that this level optimizes the physiological benefits of serotonin without introducing metabolic imbalances. Beyond this threshold, the decline in HDEP and egg mass observed at 2% Trp supplementation could be indicative of a potential feedback inhibition mechanism or an imbalance in amino acid ratios. Excess Trp leads to the accumulation of metabolites that disrupt normal physiological functions [15], thereby decreasing nutrient absorption and utilization efficiency. These findings align with earlier research, which highlighted the importance of maintaining amino acid balance in poultry diets [4]. The reduction in FCR at the optimal level of Trp further supports the hypothesis that appropriate Trp supplementation enhances nutrient utilization efficiency. This improvement in FCR may be due to reduced stress, which conserves energy that would otherwise be expended in coping mechanisms.
The observed linear reduction in WBC count, lymphocytes, and monocytes, alongside decreased levels of triglycerides and corticosterone, indicates that Trp supplementation exerts a systemic anti-inflammatory and anti-stress effect in laying hens. This effect is likely facilitated by the conversion of Trp into serotonin, a neurotransmitter that not only modulates mood and behavior but also plays a crucial role in regulating immune responses and reducing stress-induced hormonal secretion [7]. Corticosterone is a glucocorticoid hormone that can suppress immune function, disrupt metabolic processes, and reduce metabolic efficiency [12,13,16]. The reduction in corticosterone levels with Trp supplementation suggests a mitigated stress response. The decrease in triglyceride levels points to improved lipid metabolism, which is linked to more efficient hepatic function [17]. This metabolic improvement could result from a combination of reduced stress and improved nutrient assimilation. These findings align with previous research by Kwon et al. [10], which also reported the immunomodulatory benefits of dietary Trp in laying hens by improving immune homeostasis under stress conditions. This impact on immune and metabolic parameters highlights the role of Trp in enhancing poultry health.
The enhancements in egg yolk color and shell strength observed at optimal Trp supplementation levels can be attributed to reduced oxidative stress and improved mineral metabolism by serotonin. Serotonin role in modulating oxidative stress is well-documented [6,8,14], and it likely contributes to the protection of egg components from oxidative damage and maintaining yolk integrity and shell robustness [6]. The observed decline in haugh unit and shell strength at lower Trp levels (0.25%) suggests that insufficient Trp may hinder protein synthesis and the processes involved in shell formation. Furthermore, the increase in yolk pigmentation and yellowness with higher Trp supplementation could be linked to more efficient absorption and deposition of carotenoids. This is supported by the quadratic response pattern observed, indicating an optimal range for Trp effects on pigmentation. Conversely, the linear decrease in shell redness and consistent CIE L* across Trp levels might be attributed to changes in calcium metabolism and pigment deposition, which are influenced by the overall dietary composition and the metabolic state of the hens [8]. These findings collectively suggest that Trp plays a crucial role in modulating factors that affect egg quality, highlighting the importance of precise Trp supplementation in optimizing both the nutritional and egg quality.
Overall, the results of this study underscore the importance of optimal Trp supplementation in laying hen diets, not only for enhancing performance and egg quality but also for reducing stress. The findings suggest a 1% Trp supplementation is required to maximize these benefits, with implications for feed formulation strategies aimed at improving both animal welfare and product quality in the poultry industry.
