INTRODUCTION
Post-weaning diarrhea (PWD) is a multifactorial disease that occurs during the first 14 days after weaning and is caused by factors such as separation from the sow, change to solid feed, and mixing with other piglets [1–3]. The main causative agent of PWD is pathogenic Escherichia coli [4]. Weaning pigs with underdeveloped mucosal immunity and impaired intestinal barrier function are highly susceptible to pathogenic bacteria [5,6]. The PWD causes dehydration, growth retardation and increased mortality and is reported to be one of the largest causes of economic loss in swine production [7,8]. Antibiotics or high doses of zinc oxide are used in weaned pigs’ diets to prevent PWD, but their use is currently restricted worldwide due to antibiotic resistance and environmental pollution problems [8]. Therefore, new strategies are needed to improve the robustness and growth performance of weaned pigs from a nutritional perspective.
Clay minerals (CM) are naturally occurring rock or soil materials, which are mainly particulate aluminosilicate minerals, characterized by high hygroscopicity [9]. Illite and bentonite from the phyllosilicate family are among the best-known CMs, with reported effects such as a lack of primary toxicity and the absorption of feed contaminants through their high binding capacity [10,11]. Phyllosilicate has a conformational structure in which tetrahedral silica and octahedral aluminum layers are repeatedly stacked [9]. When pathogens or enterotoxins and CMs contact the animal’s body, they are adsorbed onto the surface or middle layer of the CM structure through hydrogen bonds, which reduces the number of pathogens in the intestines [12]. When CMs are fed to weaned pigs, the rate at which digesta passes through the intestines is delayed, and the activity of pathogenic bacterial enzymes is reduced, which can alleviate PWD [13,14]. However, the positive effects of adsorption may be influenced by the amount of CM supplemented [15], and since CM cytotoxicity according to the dose has been reported [16], additional research is needed to determine the CM dose.
Intestinal epithelial cells form a physical interface separating the lumen contents from the host environment and serve as an efficient barrier to prevent the invasion of pathogens and toxins [17,18]. The intestinal porcine enterocyte cell lines (IPEC-J2) originating from pig jejunum can easily observe immune and toxic responses, and they are specialized for studying the effects of various substances on intestinal epithelial cells [19,20]. Therefore, this study investigated the toxicity of illite and bentonite using IPEC-J2 cells as a primary screening verification method for the effect of supplementing illite and bentonite and evaluated the antibacterial activity and adsorption capacity of illite and bentonite by infecting IPEC-J2 cells with pathogenic E. coli. This study hypothesizes that appropriate doses of illite and bentonite do not affect cytotoxicity and that the adsorption capacity of illite and bentonite will inhibit the growth of pathogenic E. coli, thereby improving cell viability and wound healing ability.
MATERIALS AND METHODS
The addition levels of illite and bentonite in IPEC-J2 cells medium were set as follows; CON, 0%; I1, illite 1%, I1.5, illite 1.5%; I2, illite 2%; B1, bentonite 1%; B1.5, bentonite 1.5%; B2, bentonite 2%. They were completely dissolved in dimethyl sulfoxide (DMSO; GenDEPOT) according to each addition level. Afterward, they were added in a 1:1 mixture of Dulbecco’s Modified Eagle’s Medium and Ham’s F-12 Nutrient Mixture (DMEM/F-12; Welgene) containing 10% fetal bovine serum (FBS; Corning Cellgro) and 1% penicillin/streptomycin (P/S; Gibco) and used as illite or bentonite medium. The total DMSO volume in the illite or bentonite medium was set not to exceed 1% considering the cytotoxicity of DMSO. Additionally, to confirm the effect of the treatment time of illite and bentonite on IPEC-J2 cells, the treatment time was set to 2 hours, 4 hours, and 6 hours. All experiments were performed with three independent replicates, with six technical replicates per experiment.
The IPEC-J2 cell lines of intestinal porcine enterocytes was isolated from the jejunum of newborn piglets (ACC 701, Leibniz Institute DSMZ). The IPEC-J2 cells were cultured in growing medium (GM) containing 10% FBS and 1% P/S in a DMEM/F-12 containing 15 mM hydroxyethyl piperazine ethane sulfonic acid (HEPES), and cultivated in an incubator at 37℃ and 5% CO2. All experiments used IPEC-J2 cells within seven cell passages (passages 3 to 10). The IPEC-J2 cells were seeded at a density of 3,000 cells/cm2 on 58 cm2 cell plates (SPL Life Sciences) and cultured until more than 90% confluent. The 58 cm2 cell plates were coated with 8 μg/cm2 collagen from calf skin (Sigma-Aldrich). The GM was changed every 2 days.
To infect IPEC-J2 cells with pathogenic E. coli, E. coli KCTC 2571 was supplied from Korean Collection for Type Cultures (KCTC) in a lyophilized state and suspended in sterile distilled water. The 10 μL of the suspended E. coli was added to Luria-Bertani broth (LB broth; KisanBio) and cultured at 37℃ for 18 hours with shaking. After culture was complete, the suspension was vortexed and centrifuged at 4℃, 1,200 rpm, and 10 minutes. The supernatant was removed and washed once with phosphate-buffered saline (PBS). After that, 1 mL of antibiotic-free GM (supplemented with 10% FBS in DMEM/F-12) was added and pipetted. After serially diluting to 10–6 with PBS, 100 μL of the dilution was plated on LB agar (KisanBio) and cultured at 37℃ for 18 hours. The number of E. coli was counted to calculate the colony forming unit (CFU). After recording the CFU of E. coli, the supernatant of each E. coli solution was filtered using a 0.22 μm filter (Sigma-Aldrich) to remove any remaining bacterial cells and stored at –80℃ until use in the experiment as an E. coli challenge medium.
Illite (YonggungIllite) is composed of 67.4% SiO2, 20.3% Al2O3, 5.5% K2O, and other minerals (Fe2O3, Na2O, TiO2, etc.). Bentonite is sodium-bentonite and is mostly composed of montmorillonite. Illite and bentonite were completely dissolved in DMSO according to each supplementing amount and then added to DMEM/F-12.
The IPEC-J2 cells were seeded at 5 × 104 cells/well in a 96-well cell plate and cultured with GM for 24 hours at 37℃ and 5% CO2. After 24 hours, each treatment medium was added and cultured according to each treatment time (2, 4, and 6 hours). Cytotoxicity was analyzed using the Quanti-MAX WST-8 assay (Biomax) according to the manufacturer’s protocol. After incubation, 100 μL of GM and 10 μL of WST-8 assay solution were dispensed into each well and then cultured for 4 hours. Afterward, the cell absorbance of each well was measured at 450 nm using a microplate reader (INNO, LTekc).
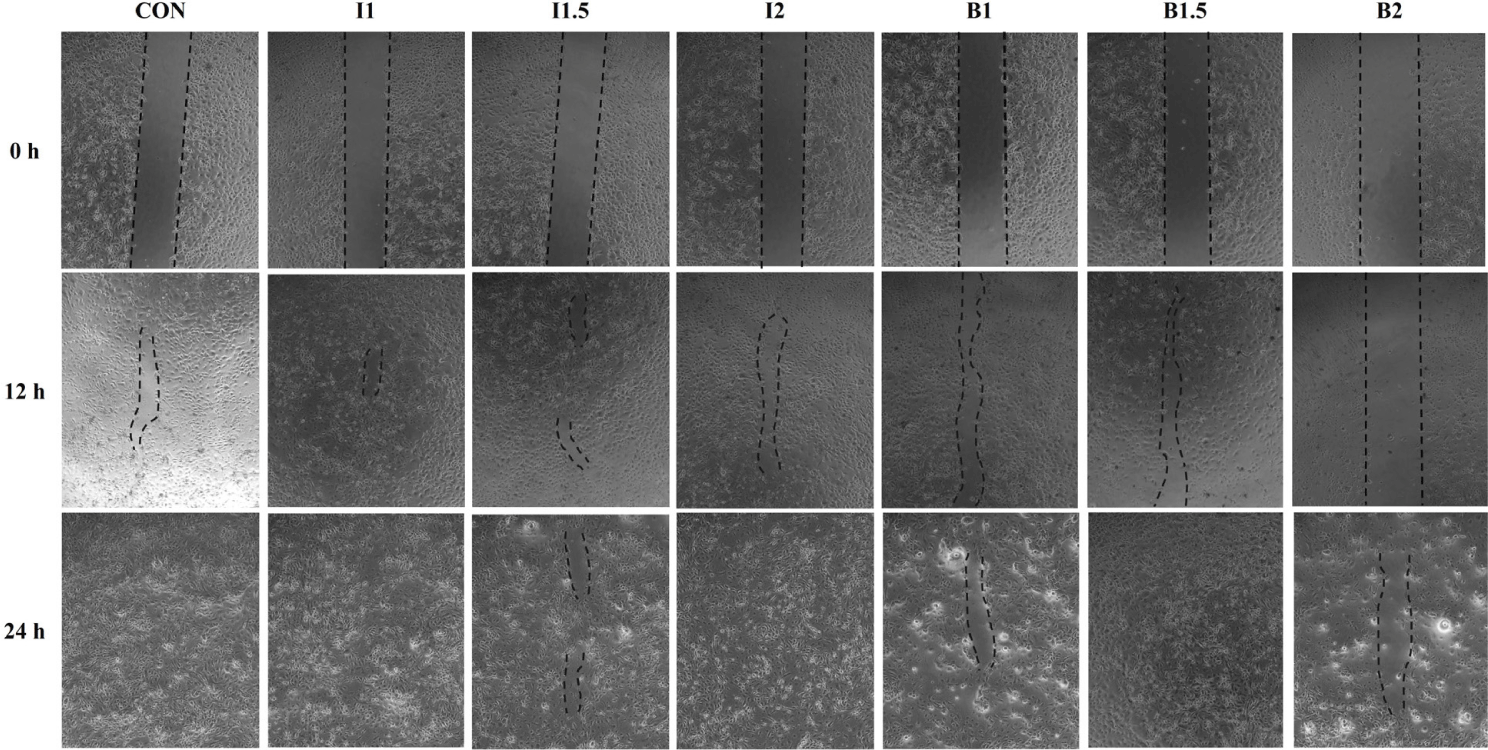
The cells were seeded at 1 × 105 cells/well in a 24-well cell plate and cultured with GM for 24 hours at 37℃ and 5% CO2. After 24 hours, each treatment medium was added and cultured according to each treatment time. After incubation, the cells were washed once with Dulbecco’s Phosphate-Buffered Saline (DPBS; Welgene) and then scratched using a SPLScar Scratcher (SPL Life Sciences). After removing floating cell debris through three DPBS washes, the area of the scratch was measured under an inverted microscope (EVOS M5000 Imaging System, Thermo Fisher Scientific). After 12 and 24 hours, the change in the area of the scratch was measured.
To confirm the effects of illite and bentonite on cell viability during E. coli infection, an E. coli challenge treatment were as followed; No-challenge, no E. coli challenge; E. coli challenge; EI1, E. coli challenge + illite 1%; EI1.5, E. coli challenge + illite 1.5%; EI2, E. coli challenge + illite 2%; EB1, E. coli challenge + bentonite 1%; EB1.5, E. coli challenge + bentonite 1.5%; EB2, E. coli challenge + bentonite 2%. The treatment times were the same at 2, 4, and 6 hours. The cells were seeded at 1 × 105 cells/well in a 24-well cell plate and cultured with GM for 24 hours at 37℃ and 5% CO2. After 24 hours, each treatment medium was added and cultured according to each treatment time. To adapt to antibiotic-free GM before E. coli challenge, all treatment groups were dispensed with antibiotic-free GM and cultured for 30 minutes. After that, the E. coli challenge treatment group was infected with E. coli for 5 hours with E. coli challenge medium having a multiplicity of infection (MoI) of 300. This MoI and culture time were selected through preliminary experiments to allow partial cell damage. The no-challenge group was dispensed with antibiotic-free GM and cultured for 5 hours. After 5 hours, the cells were detached with Trypsin-EDTA (Sigma-Aldrich). To assess cell viability, aliquots of the collected cells were mixed with trypan blue and loaded onto a hemocytometer. The total number of cells and dead cells were counted.
To confirm the E. coli adhesion inhibition ability of illite and bentonite, an E. coli challenge was conducted on all treatment groups. The treatment times were the same at 2, 4, and 6 hours. The cells were seeded at 1 × 105 cells/well in a 24-well cell plate and cultured with GM for 24 hours at 37℃ and 5% CO2. After 24 hours, each treatment medium was added and cultured according to each treatment time. To adapt to antibiotic-free GM before E. coli challenge, all treatment groups were dispensed with antibiotic-free GM and cultured for 30 minutes. After that, the E. coli challenge treatment group was infected with E. coli for 5 hours with E. coli challenge medium having 300 MoI. After the E. coli challenge, the cells were washed once with DPBS to remove any removing E. coli residues, then GM was added and cultured for 24 hours. To remove non-attached E. coli, the cells were washed three times with PBS and then lysed with 1% Triton-X 100 (Sigma-Aldrich). The cell lysate was serially diluted, plated on LB agar, and incubated at 37℃ for 24 hours to measure the number of E. coli.
All experiments were performed with three independent replicates, with six technical replicates per experiment. The mean of the technical replicates was calculated for each experiment. The effects of illite and bentonite’s addition levels, the illite or bentonite’s treatment time, and possible interactions between each of these were analyzed using means of multifactor variance analysis (MANOVA) for repeated measurements of each cell. The measurements were considered repeated over time and were entered as independent variables in the MANOVA. The data on scratch width were analyzed statistically using one-way ANOVA by calculating the change in width according to the illite and bentonite treatment time, and then Tukey’s multiple tests were performed as a post-hoc test. Statistical analyses and visualized graphs were performed using JMP Pro 16 (SAS Institute) and GraphPad Prism (Version 9.1.0; GraphPad Software), respectively. All data are presented as mean ± SE (n = 3). A probability level of p < 0.05 was indicated to be statistically significant.
RESULTS
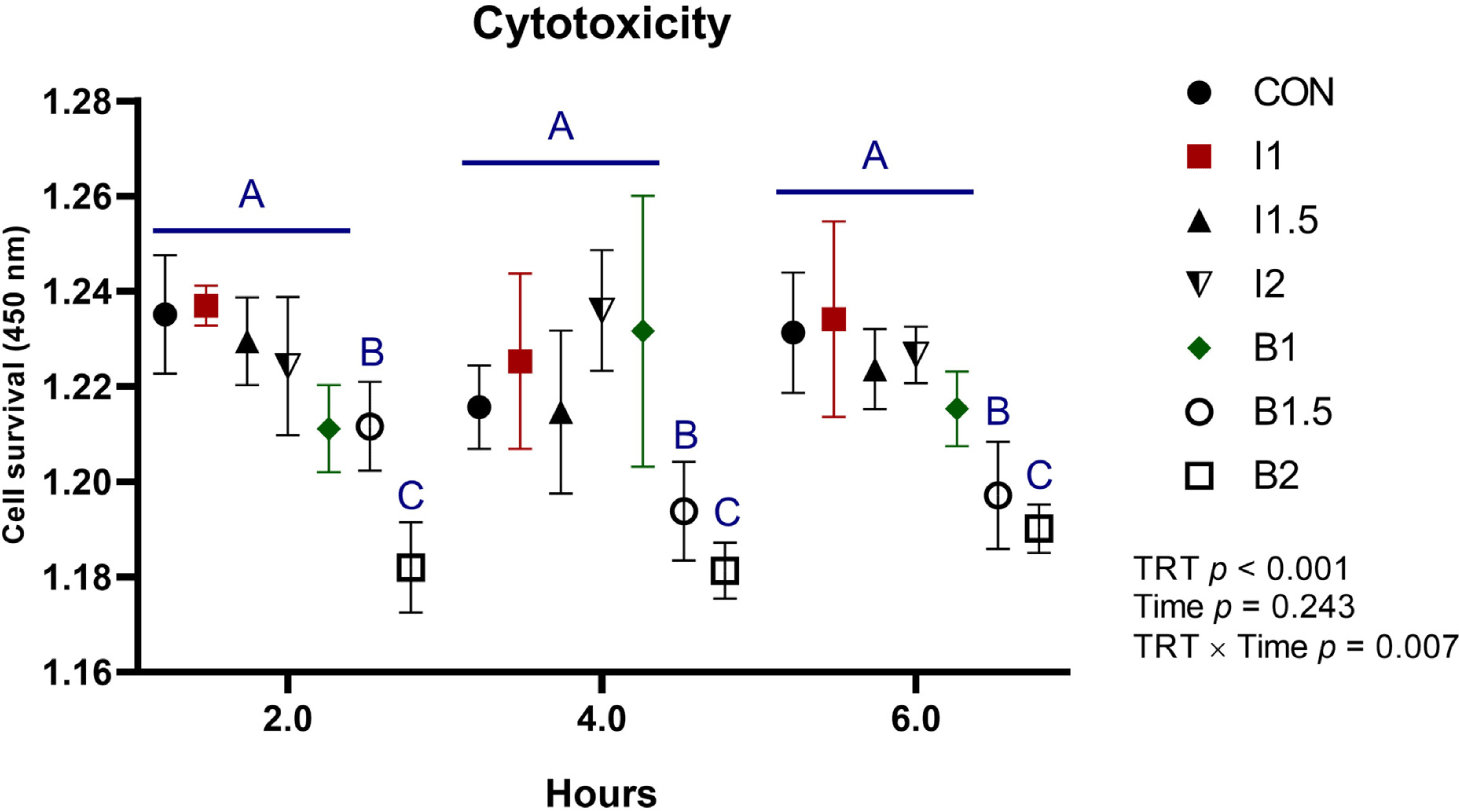
The effects of the addition levels of illite and bentonite and the treatment time on cytotoxicity are shown in Fig. 1. The B1.5 and B2 treatment groups showed higher (TRT, p < 0.05) cytotoxicity than the CON and illite groups. There was no significant difference in cytotoxicity according to the treatment time of illite and bentonite.
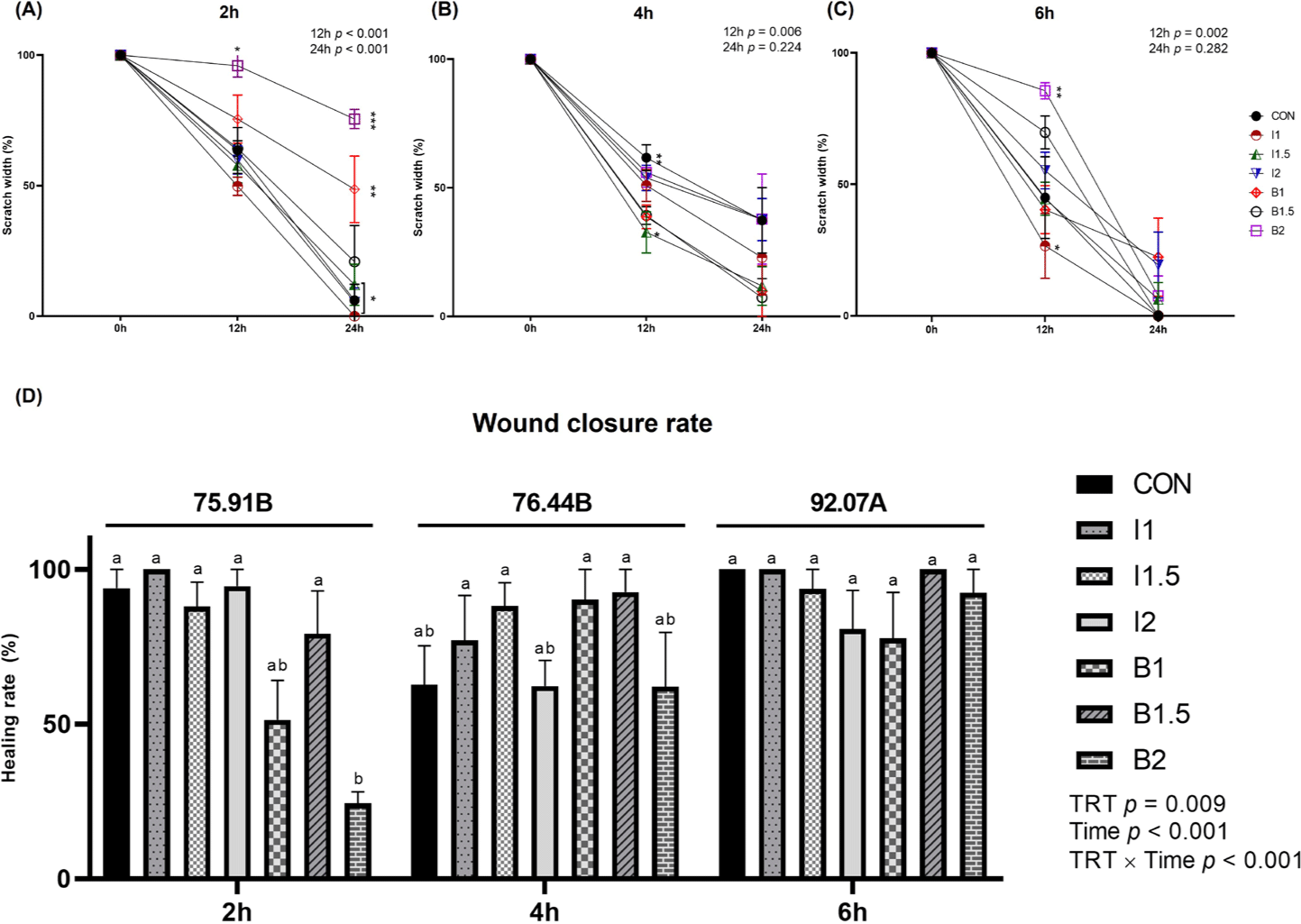
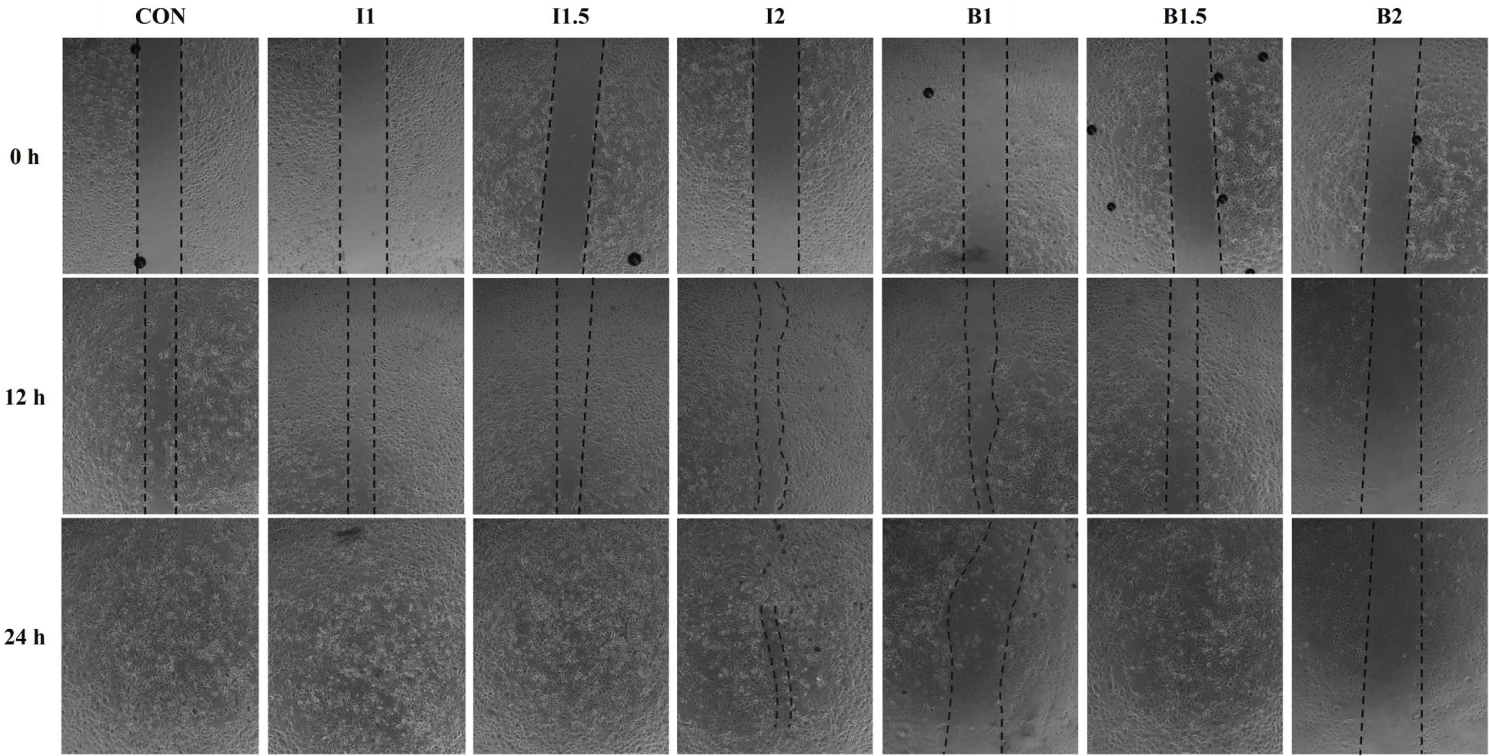
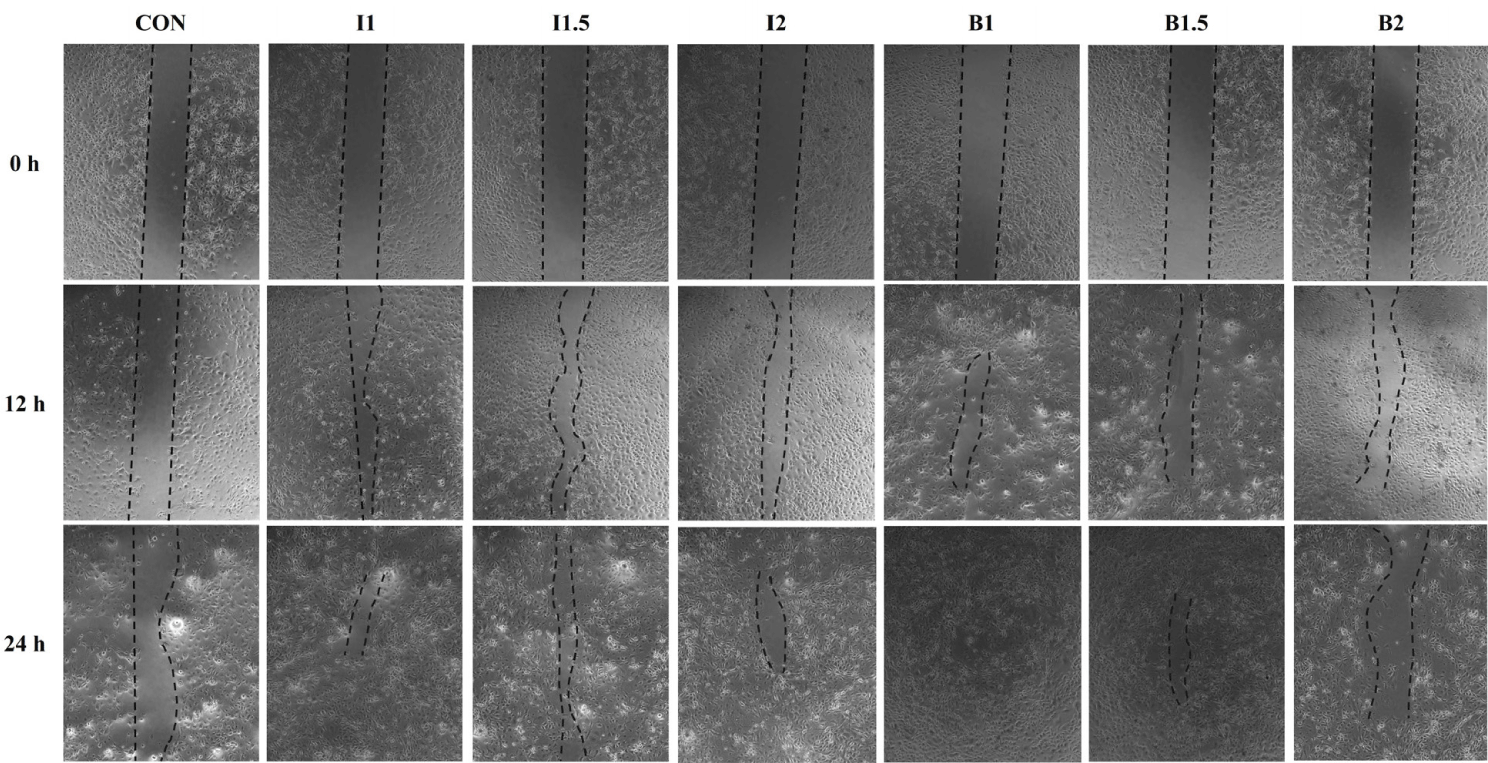
The effects of the addition levels of illite and bentonite and the treatment time on cell healing rate are shown in Figs. 2, 3, 4, and 5. When treated with illite and bentonite for 2 hours, the scratch width of the illite treatment groups and the B1.5 treatment group significantly decreased (p < 0.05) compared to the B2 treatment group and showed a similar width to the CON group after 12 hours (Fig. 2A and 3). After 24 hours, it was confirmed that all scratches in the I1 treatment group had recovered. When treated with illite and bentonite for 4 hours, the scratch width of the I1.5 treatment group significantly decreased (p < 0.05) compared to the CON group after 12 hours, and there was no significant difference among the treatment groups after 24 hours (Fig. 2B and 4). When treated with illite and bentonite for 6 hours, the I1 treatment group showed a significantly lower (p < 0.05) scratch width than the B1.5 and B2 treatment groups after 12 hours (Fig. 2C and 5). For wound closure rate, when illite and bentonite were treated for 6 hours, a significantly higher (Time, p < 0.05) healing rate was shown than that of the 2 and 4-hour treatments (Fig. 2D).
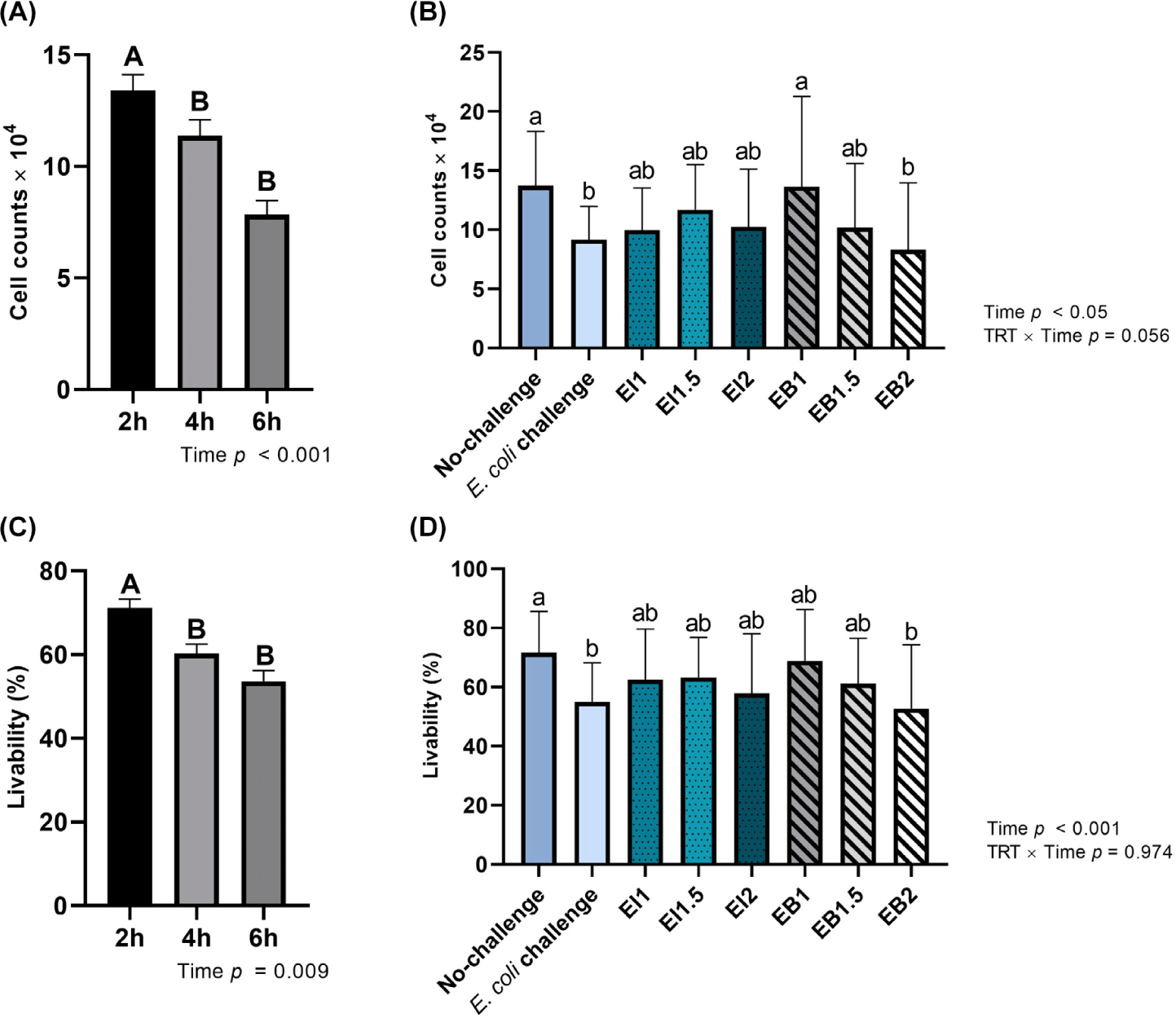
When treated with illite and bentonite for 2 hours, the live cell count and cell viability were significantly higher (Time, p < 0.05) than when treated for 4 and 6 hours (Fig. 6A and 6C). The EB1 treatment group showed a significantly higher (TRT, p < 0.05) live cell count than the E. coli challenge and EB2 treatment groups (Fig. 6B). The E. coli challenge group showed significantly lower (TRT, p < 0.05) cell viability than the no-challenge group (Fig. 6D).
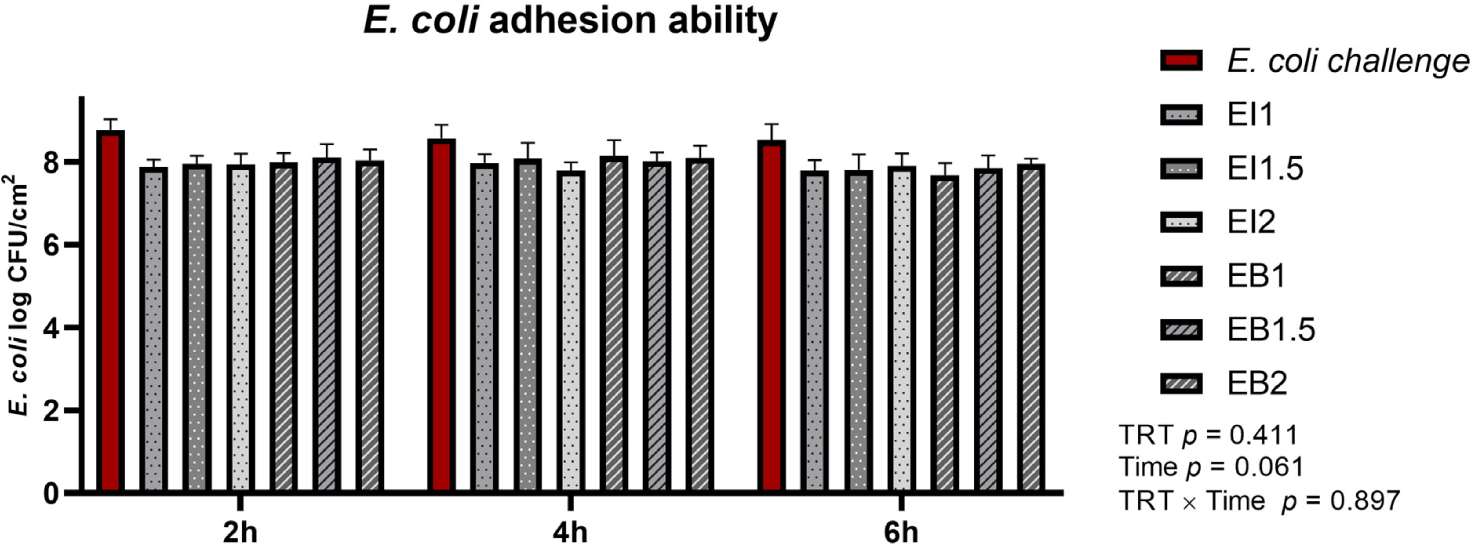
The effects of the addition levels of illite and bentonite and the treatment time on E. coli adhesion ability are shown in Fig. 7. There was no significant difference in E. coli adhesion ability according to the treatment of illite and bentonite.
DISCUSSION
This study aimed to secure basic data for the application of illite and bentonite in weaned pigs’ diets by evaluating the toxicity of illite and bentonite and cell viability through their absorption capacity using IPEC-J2 cells. Illite and bentonite, which are representative examples of phyllosilicates, have attracted much scientific attention due to their wide range of applications [21,22]. In particular, bentonite has been studied as a toxin binder in livestock diets such as broiler and pigs [23,24]. However, bentonites may negatively affect the immune response or growth performance of broilers or pigs [25,26]. Baek et al. [16] reported that montmorillonite, the main component of bentonite, induced oxidative stress and cell membrane damage at 20–1,000 μg/mL in normal human intestinal cells (INT-407), and that long-term exposure resulted in cytotoxicity. In this study, 1.5% and 2% bentonite were observed to be cytotoxic compared to the CON group. Illite showed a survival rate similar to that in the CON group but lower cytotoxicity than bentonite. The bentonite type we used was sodium-bentonite. This refined bentonite has a higher absorption capacity than the original form because the surface area and porosity of the material are larger [26,27]. High absorption capacity can cause cytotoxicity by binding not only toxins but also essential nutrients [26]. In contrast, illite has a structure similar to bentonite but has a weaker absorption capacity than bentonite [28]. Accordingly, it is thought that illite did not show cytotoxicity even at higher addition doses than bentonite. Also, in this study, no cytotoxicity was observed in IPEC-J2 cells according to the treatment time with illite and bentonite. Although exposure to illite and bentonite for up to 6 hours did not appear to affect cytotoxicity, the retention time of CMs in the intestines may vary when ingested by weaned pigs, so further studies in animal models are needed.
Wounds are self-repaired in IPEC-J2 cells through the proliferation and migration of epithelial cells, and the self-repair ability of intestinal epithelial cells plays an important role in maintaining the integrity of the intestinal barrier [29,30]. In this study, we artificially scratched IPEC-J2 cells to evaluate the effects of illite and bentonite on their wound-healing ability. When treated with illite for 2, 4, and 6 hours, the scratch width was reduced compared to the B1.5 and B2 treatment groups, and a fast-healing rate was observed. Cells treated with illite and bentonite for 6 hours showed a higher healing rate than those treated for 2 and 4 hours. This suggests that illite can help alleviate intestinal damage in weaned pigs by accelerating the self-recovery of wound healing in intestinal epithelial cells. The healing rate in the B2 group was significantly slower than in the other treatment groups, and the image showed almost no recovery. This may have been due to the cytotoxicity of the processed bentonite mentioned above.
The PWD caused by pathogenic E. coli in weaned pigs causes systemic inflammation and damage to the intestinal epithelium [4,31]. The intestinal mucosal barrier is the first line of defense against the invasion of pathogenic microorganisms and toxins into the intestine [32]. Pathogenic E. coli induces abnormal intestinal barrier function and affects intestinal health by reducing the expression of tight junction proteins and increasing permeability in intestinal epithelial cells [33–35]. IPEC-J2 cells have typical epithelial cell characteristics and are a permissive host for commensal bacteria and enteric pathogens, making them an excellent model for studying the interaction between bacteria and IPEC-J2 cells [36]. In this study, when IPEC-J2 cells were infected with E. coli at 300 MoI, they showed significantly lower cell viability than the uninfected treatment group. In live cell counts, the EB1 group showed a higher cell count and higher cell viability than the E. coli group. Since bentonite is characterized by high adsorption ability, it was expected to show higher E. coli adhesion than illite. However, this study found no difference between illite and bentonite. Thus, the effects should be further studied through more in-depth cell experiments and animal experiments.
CONCLUSION
Illite and bentonite treatments, except for 1.5% and 2% bentonite, did not induce cytotoxicity regardless of the treatment time in IPEC-J2 cells. When treated with illite for 2, 4, and 6 hours, the scratch width was reduced compared to the B1.5 and B2 treatment groups, and a fast-healing rate was observed. When IPEC-J2 cells were infected with E. coli, the number of live cells was similar to that of the uninfected group when treated with 1% bentonite. In conclusion, this study can be used as basic data on illite and bentonite for the intestinal health of weaned pigs.
