Lactococcus lactis is a homo-fermentative, Gram-positive cocci and is traditionally used around the world in the fermentation process of dairy products, kimchi and silage [1]. Recent research show that L. lactis suppressed methane production in ruminant because these bacteria had the ability to inhibit rumen methanogens [2].
Among the antibacterial compounds produced by lactic acid bacteria, bacteriocin is a natural proteinaceous substance that inhibits the growth of pathogenic bacteria present in food and whose production can be controlled through genetic manipulation [3]. When ingested, it is decomposed by proteolytic enzymes in the digestive system and is harmless to the human body. Therefore, bacteriocin is attracting attention as a natural, non-toxic preservative produced by microorganisms with potential applications in the food industry [4]. Many bacteriocins have been identified and a few under the same Lactobacillus genus are known to produce different types with different activity ranges [5]. Nisin, a representative bacteriocin produced by Lactococcus lactis subsp. lactis, is a polypeptide composed of 34 amino acids that is non-toxic to humans. Owing to its characteristics, its application as a food preservative has been expanded, and it is currently permitted as a food additive in several countries [6].
In the present study, L. lactis JNU 534 producing bacteriocin (previously identified as Pediococcus damnosus) was isolated from kimchi [7]. This strain was cultured in de Man, Rogosa, and Sharpe (MRS) broth (Difco, Sparks, MD, USA) at 37°C for 24 h. Total genomic DNA of L. lactis JNU 534 was extracted using the PureHelixTM Genomic DNA prep kit (Nanohelix, Daejeon, Korea) according to the instructions of the manufacturer. The concentration of extracted DNA and quality were measured using the Qubit 2.0 Fluorometer (Invitrogen, Carlsbad, CA, USA) and the SpectraMax ABS Plus microplate reader (Molecular devices, San Jose, CA, USA), respectively.
The complete genome of L. lactis JNU 534 was sequenced using the PacBio Sequel system (Pacific Biosciences, Menlo Park, CA, USA) and the Illumina HiSeq (151 × 2 base pair [bp] paired-end sequencing) platforms at Macrogen (Macrogen, Seoul, Korea). The preparation of sequencing libraries for PacBio sequencing was performed using the SMRTbellTM template prep kit 1.0 (Pacific Biosciences) and Illumina sequencing was performed using the NEBNext® UltraTM DNA library prep kit for Illumina® (NEB, Ipswich, MA, USA), according to the instructions of the manufacturers. A total number of 131,035 reads with a mean subread length of 9,196 bases (N50, 10,823 bases) were obtained with PacBio sequencing, and 33,461,064 paired-end reads totaling 5,052,620,664 bp were obtained with Illumina sequencing. De novo assembly was carried out using microbial assembly application within the PacBio SMRT analysis pipeline 8.0 with default options (http://www.pacb.com/devnet/). The Microbial Assembly Application can determine whether contigs are circular and automatically rotate each contig start site as the origin of replication. Assembly error correction was performed using Pilon version 1.21 [8]. The completeness of the genome assembly was assessed using the Benchmarking Universal Single-Copy Orthologous suite (BUSCO, v5.1.3) [9]. The chromosome, plasmid, and contig annotation were performed using rapid prokaryotic genome annotation (Prokka, v1.13) [10].
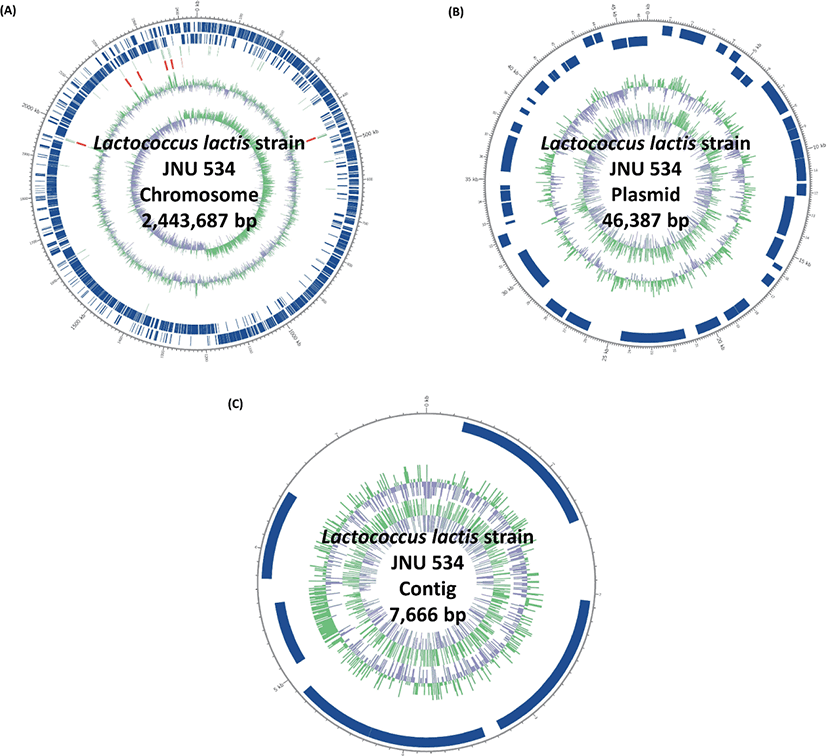
The complete genome sequence of L. lactis JNU 534 consists of one circular chromosome (2,443,687 bp) with an overall GC (guanine-cytosine) content of 35.2%, one circular plasmid sequence (46,387 bp) with a GC content of 34.5%, and one contig sequence (7,666 bp) with a GC content of 36.2% (Table 1). A total of 2511 predicted genes, including 19 rRNA and 62 tRNA, were identified on the genome, of which 2455, 49, and 7 coding DNA sequences were located on the chromosome, plasmid, and contig, respectively. (Fig. 1) The genomic information of L. lactis JNU 534 may guide future research on the characteristics and stability of this strain and confirm its applicability as a preservative of foods of animal origin.
| Name | Length (bp) | GC (%) | Depth | CDSs | tRNA | rRNA |
|---|---|---|---|---|---|---|
| Chromosome | 2,443,687 | 35.2 | 421.4 | 2,455 | 62 | 19 |
| Plasmid | 46,387 | 34.5 | 1,427.1 | 49 | 0 | 0 |
| Contig | 7,666 | 36.2 | 94.6 | 7 | 0 | 0 |
| Total | 2,497,740 | 35.2 | 439.1 | 2,511 | 62 | 19 |
NUCLEOTIDE SEQUENCE ACCESSION NUMBER
The complete chromosome, plasmid, and contig sequences of L. lactis JNU 534 have been deposited in GenBank under the accession numbers CP095737.1, CP095738.1, and CP095739.1, respectively. The BioProject and BioSample accession numbers are PRJNA826729 and SAMN27578898.
















