Background
Ghrelin is a 28-amino-acid peptide isolated from the rat stomach in 1999 which is mainly produced by X/A-like cells in the oxyntic mucosa [1]. It was also found in other parts like hypothalamus, pituitary gland, lung, kidney [2–4], duodenum, ileum, colon and pancreas [5]. The ghrelin receptors (GHS-R) have been detected in many central and peripheral tissues and pancreatic α-cells, β-cells, exocrine cells. Ghrelin shows a number of actions at the gastrointestinal tract level. It is well known as a stronger activator of growth hormone (GH) through central nervous system (CNS) and modulation of food intake. And it also exhibits lots of other biological activities including energy expenditure, stimulation of lactotroph and corticotroph secretion, influence on sleep and behavior or modulation of heart rate and blood pressure ([6–9]; Assakawa et al. [10]).
Recent studies demonstrated that ghrelin could modulate exocrine secretions as well as pancreatic endocrine secretions. There were two opposite theories. First, Zhang and coworkers demonstrated that intravenous administration of ghrelin to the rats produced inhibition of enzyme secretion, and that this inhibitory effect of ghrelin on pancreatic exocrine secretion is indirect and may be exerted at the level of intrapancreatic neurons [11]. In contrast, other studies revealed that central as well as peripheral administration of ghrelin significantly increased pancreatic fluid and protein output, though the activation of vagal centers in the brainstem and stimulation of vagal efferent nerves [12, 13]. It is generally believed that, in the intestinal phase, stimulation of pancreatic enzyme secretion depends, in the main part, on the neuronal mechanism involved in the CCK release and activation of cholinergic vago-vagal enteropancreatic reflex. However, these physiological roles of ghrelin in the modulation of exocrine pancreatic functions are still unclear (Gherlaldoni et al. [14]).
Therefore, this study was conducted to determine the effects of exogenous ghrelin, given intraperitoneal (i.p.) on plasma CCK level and α-amylase output in the sprague-dawley rats. Meanwhile, the responses of pancreatic proteins to ghrelin were also analyzed using the 2-DE system.
Methods
Rat ghrelin peptide was obtained from Bachem (Bubendorf, Switzerland). Rat plasma ghrelin and CCK enzyme immunoassay kit were purchased from Phoenix Pharmaceuticals (Mountain View, CA, USA). The α-amylase activity analyzed using an EnzyChrom™ a-Amylase Assay Kit (ECAM-100).
Sprague-Dawley rats (300 g) (Samtaco, Osan, Korea) were used for all experiments. Animals were housed at one animal per cage on a 12/12 h light cycle (lights on at 8 am) and given access to food and water ad libitum. Ghrelin (0.1, 1.0, 10.0 ug/kg, respectively) were injected intraperitoneally in rats. One hour after injection, blood samples (1 ml) were collected from rat caudal vein once time into heparined tube and immediately centrifuged (3,000 rpm/min, 15 min) to obtain the plasma. Aliquots of plasma were stored at -80°C till analyzed. Then rats were anaesthetized via intramuscular injection of zoletil (Vetbutal) at a dose of 15.0 mg/300 g (BW). And the rat pancreas tissues were collected. All experimental procedures were in accordance with the “Guidelines for the Care and Use of Experimental Animals of Pusan National University”.
The plasma ghrelin and CCK concentration were separately determined by enzyme immunoassay kit (Phoenix Pharmaceuticals, Inc., Burlingame, CA, USA). The plasma α-amylase activity analyzed using an EnzyChrom™ a-Amylase Assay Kit (ECAM-100, BioAssay Systems, Hayward, CA, USA). All the operations were done followed the kit manual.
Pancreatic tissues were collected and then pulverized into powder under liquid nitrogen and stored at -80°C until use. Tissues (0.5 g) were homogenized in 1ml lysis solution containing 7 M urea, 2 M thiourea, 4% chaps, 40 mM Tris, 65 mM DTT, 0.5% IPG buffer and 1X protease inhibitor (GE Healthcare, Piscataway, New Jersey, USA). This mixture was stirred every 5 min for 30 min and then centrifuged at 14,000 rpm for 30 min at 20°C. The supernatant was then stored in aliquots at -80°C until analysis.
2-DE was performed of pooling pancreatic tissue samples from three animals in each group. Briefly, protein samples were diluted into isoelectric focusing (IEF) buffer containing 6 M urea, 2 M thiourea, 1% 3-((3-Cholamidopropyl)dimethylammonium)-1-propanesulfonate (CHAPS), 0.002% bromophenol blue, 0.5% phamalyte (pH 3-10NL) and 65 mM dithiothreitol (DTT). Then 100 μg protein smaples of control or treatment was loaded on Immobiline DryStrip gels (pH 3-10NL, 18 cm, GE Healthcare) for rehydration for 12 h at 20°C. The IEF procedures were performed using an IEF electrophoresis unit (GE Healthcare) following the manufacturer’s protocols. The following voltage program was used after the 12 h rehydration: linear ramp from 500 to 1000 V over 2 h, then a constant voltage of 8000 V for 7 h to give a total of 56,000 V h. After focusing, gel strips were equilibrated in a solution containing 50 mM Tris/HCl (pH 8.8), 6 M urea, 2% SDS, 30% glycerol, 0.002% w/v bromophenol blue and DTT for 15 min, followed by incubation in the same solution but replacing DTT with 135 mM iodoacetamide (IAA) for another 15 min. After that the equilibrated strips were inserted into sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) gels (18 cm, 12%). SDS-PAGE was performed using an Ettan DALT 2-D gel system (GE Healthcare). Upon completion, gels were stained using a PluseOne Silver Staining Kit (GE Healthcare). The silverstained gels were scanned using an Umax scanner (PowerLook 2100XL, UMAX Technologies, Inc., Dallas, TX, USA). Scanned gel images were processed by Proteomweaver™ 2-D Analysis Software (Definiens AG, Munich, Germany).
Results and discussion
The i.p. infusion of ghrelin at doses of 10.0 μg/kg body weight (BW) increased the plasma ghrelin concentrations (p = 0.07) and elevated the plasma CCK level significantly (p < 0.05). Although there was no statistically significant, the α-amylase activity tended to increase (Table 1). These data indicated that ghrelin may have role in pancreatic exocrine secretion through the stimulation of CCK release and activation of dorsal vagal nerve. CCK is one of the major gut hormones which released from duodenal mucosa I cells. It can stimulate pancreatic exocrine secretion via activation of CCK1 receptors and entero-pancreatic vago-vagal reflex [15–19]. Whether CCK plays a role in the exocrine secretion response to ghrelin is unknown.
| Items | Ghrelin (ng/ml) | CCK (ng/ml) | α-amylase (U/L) |
|---|---|---|---|
| Control1 | 0.202 ± 0.1522* | 5.745 ± 2.428 | 3791.799 ± 208.308 |
| Treatment1 | 3.223 ± 2.140 | 11.306 ± 3.937 | 4210.847 ± 262.825 |
1Control = vehicle/500 uLSaline, Treatment = Ghrelin (10.0 ug/kg)/500 uLSaline.
2Data are mean ± SEM and *means significantly different, p < 0.05 (Student’s paired t-test).
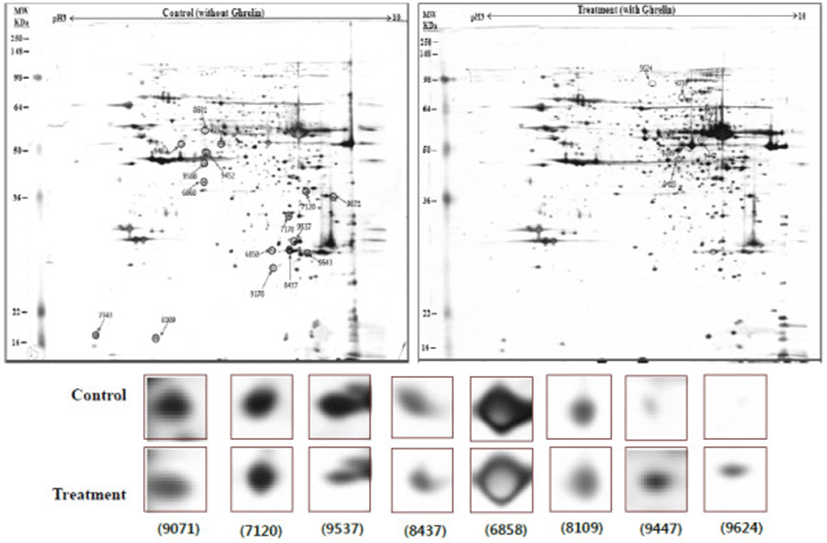
The response of pancreatic proteins to ghrelin administration were analyzed by 2-DE in order to get a better understanding of the mechanisms involved in the ghrelin-exocrine secretion through CCK signal. Eight spots ranging from 6 to 200 kDa were detected differently expressed in the pancreatic protein map (Figure 1). One spot was up-regulated and six were down-regulated compared with control group. One spot was found only expressed in treatment group. These proteins were classified according to their functions (Table 2). Three down-regulated proteins eukaryotic translation initiation factor 5A-1 (eIF5A), peroxiredoxin IV (PRx IV) and ubiquinol-cytochrome c reductase (UQCR) were particular interested. eIF5A is involved in biosynthesis of pancreatic enzyme by increasing plasma CCK level [20]. And this data was different with ours. Judging by our reports, ghrelin may affect on pancreatic exocrine secretion via other factors as well as releasing CCK. Also, Yannick demonstrated that UQCR and PRx IV down-regulated biosynthesis of insulin related to diabetes [21]. Synthetically, when the effects of exogenous ghrelin, given intraperitoneal (i.p.) on pancreatic exocrine secretion, we have to consider other factor like growth hormone related to insulin synthesis as involving the CCK release and activation of cholinergic vago-vagal enteropancreatic reflex.