INTRODUCTION
The number of oocyte-containing follicles is the key to successful assisted reproductive technologies (ARTs) in domestic animals [1,2]. Despite a worldwide increase in the application of ARTs, the amount of healthy follicle reserves on the ovaries remains a limiting factor to ARTs success in domestic animals [3]. Antral follicle count (AFC), obtained using high resolution transrectal ultrasonography, refers to the total amount of antral follicles (AF; follicle population) present in an ovary at a specific time [4,5]. During the natural ovarian cycle, AFC in mono-ovulatory large ruminants is consistent throughout their estrous cycle, and cattle with a greater AFC have improved pregnancy outcomes [6]. As compared to mono-ovulatory cattle, poly-ovulatory ruminant species such as sheep and goats can potentially ovulate more than one follicle per ovarian cycle. In poly-ovulatory small ruminants, fertility (prolificacy) is intimately correlated to the condition of the follicle population in ovaries, ovarian follicular development, and ovulation rate [7]. In goat production, producers have an intense interest in increased productive efficiency that affects their farm profit; therefore, it is very important to attend goat fertility (an economically important trait) [8]. Due to the utilization of an economically important trait for genetic improvement of livestock production, more research is needed to considerably investigate the association between the ovarian follicular reserve (remaining oocyte supply) and reproductive potential in species with low ovulation performance, including sheep and cattle [9,10]. To investigate this point, information regarding the involvement of AFC available for selection into the ovulatory pool in poly-ovulatory species is needed. Despite the wide use of AFC as a biomarker for identifying fertility potential in mono-ovulatory animals, there is so little information on the association between AFC and fertility potential in poly-ovulatory small ruminants, including goats. Until now, the influence of different AFC at the onset of synchronization (exogenous hormonal trigger) on ovarian response to stimulation and fertility potential has not been explored in goats. Thus, understanding ovarian biology in sheep and goats is an important component in manipulating ovarian functions in poly-ovulatory small ruminants, and a better body of knowledge about follicular development is crucial to increasing used ARTs in small ruminant herds [11,12]. Taking all of these observations into consideration, we hypothesized that different numbers of AF and presence of corpora lutea (CL) at the onset of synchronization would lead to different follicular responses to stimulation and fertility potential in goats following the fixed-time artificial insemination (fixed-time AI) program. The present research was planned with the objective of evaluating the effects of different numbers of AF and the presence of CL at the onset of hormonal synchronization for fixed-time AI on ovarian response to stimulation and fertility potential in primiparous Boer does.
MATERIALS AND METHODS
The Animal Care and Use for Science and Technology Research of Maejo University (MACUC019A/2564) approved experiment protocol.
The research was conducted using 128 primiparous, non-pregnant crossbred does (local × Boer) with an average age of 19.3 ± 3.4 months (mean ± SD) and body condition score (BCS) of 2.5 ± 0.8 (mean ± SD). Does were reared in a semi-intensive management and fed a diet consisting fresh-cut ruzi grass (Brachiaria ruziziensis) and commercial concentrate (18% crude protein). Fresh drinking water and mineral licks were provided to goats throughout the study period. The study was carried out at goat farms in Ching Mai province, Thailand (latitude 18°36′36′′N, longitude 98°53′7′′E, and altitude 300 m), which was conducted over the summer season of March to May 2022.
At the initiation of hormonal synchronization for ovulation and fixed-time AI (on Day 0; Fig. 1), 128 does were evaluated by high-frequency (7.5 MHz) transrectal ultrasound with a linear-array transducer (HS-1600V, Honda Electronics) to detect all visible AF (≥ 2 mm in diameter) [9,13] and corpora lutea (CL) on both ovaries.
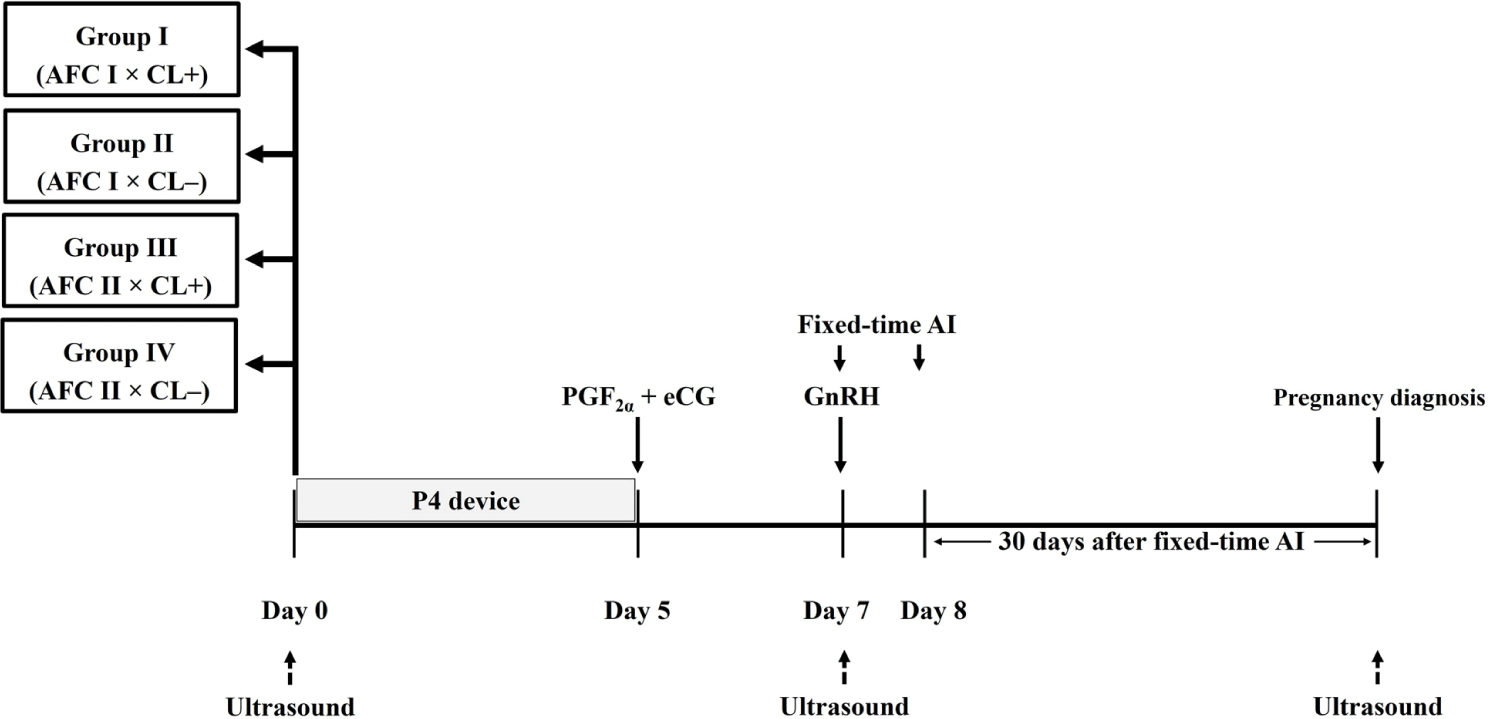
Antral follicles on both ovaries were counted to generate AFC. The reproductive conditions of the does are in luteal status (presence of CL; n = 21) and follicular status (absence of CL; n = 107). Based on two factors (AFC [AFC I and AFC II] and CL [with CL and without CL]), 128 does were divided into four groups in a 2 × 2 factorial arrangement. Group I (AFC I × CL+; n = 10) included does having AFC ≤ 3 follicles (1–3 follicles; AFC I) and with CL (CL+). Group II (AFC I × CL–; n = 61) included does having AFC ≤ 3 follicles (1–3 follicles; AFC I) and without CL (CL–). Group III (AFC II × CL+; n = 11) comprised does having AFC > 3 follicles (4–9 follicles; AFC II) and with CL (CL+). Group IV (AFC II × CL–; n = 46) comprised does having AFC > 3 follicles (4–9 follicles; AFC II) and without CL (CL–). At each examination, the relative location and follicular characteristics (number and diameter) of detected ovarian AF in both ovaries were recorded and sketched on ovarian charts. Based on the follicular diameter, the AF were classed as small-sized (2–4 mm) or large-sized (> 4 mm) [14].
At the beginning of synchronization protocol (on Day 0; Fig. 1), does were inserted with progesterone (P4)-releasing an intravaginal device (CIDR; 300 mg of P4, Eazi-Breed®, Zoetis). At P4-device withdrawal on Day 5, all does received intramuscular administrations of prostaglandin F2 alpha (PGF2α; 0.25 mg of cloprostenol, Estrumate®, MSD Animal Health) and equine chorionic gonadotrophin (eCG; 400 IU, Folligon®, MSD Animal Health). On Day 7, does were administered with gonadotropin-releasing hormone (GnRH; 0.01 mg of Buserelin acetate, Receptal®, MSD Animal Health) and were inseminated with cervical AI using the first dose of frozen thawed semen. All does were inseminated a second time 24 h later (on Day 8). The straw semen (0.25 mL) contained 200 × 106 spermatozoa/0.25 mL straw.
On Day 7 (Fig. 1), all does were scanned by a transrectal ultrasound to detect all visible AF (≥ 2 mm) on both ovaries. Antral follicles were classified, based on diameter, as small AF (2–4 mm) or large AF (> 4 mm) [14]. Ovarian response to successful hormonal induction in the does was indicated by the emergence of the large preovulatory follicles (POFs) (> 4 mm) on their ovaries after the end of the hormonal synchronization period [14,15]. Responsive rate (%) computed as the percentage of does that emerged large POFs (> 4 mm) on Day 7 divided by the number of experimental does. In addition, the 128 does were sub-classified, based on ovarian response to hormonal stimulation, into two groups: ovarian responsive does (n = 107) and ovarian non-responsive does (n = 21).
All does were evaluated by transrectal ultrasonography to diagnose their pregnancy status by scanning the uterine contents at 30 days after fixed-time AI. Pregnancy was identified by the presence of an amniotic vesicle containing an embryo.
The pregnancy rate was computed as the percentage of animals pregnant divided by the total number of experimental animals. The kidding rate was computed as the percentage of females having birth divided by the number of pregnant females. Single, twin, triplet, and multiple kidding rates were determined as the percentage of does having a single kid, twin, triplet, or multiple kids divided by the number of does having birth. In addition, fertility (prolificacy) was the number of kids born per does that kidded [16].
Analysis of all data was performed in SAS OnDemand for Academics (SAS Institute). The class variables of the statistical model were the different number of AF and the different status of CL on Day 0 and the emergence of large POFs on Day 7. The covariates of the model were BCS and age; however, BCS and age prior to start the study had no effect (p > 0.0500) on number and diameter of AF and fertility. A 2 × 2 factorial analysis was used to consider the effect of AFC types, CL presence, and their interaction on number and diameter of AF on Days 0 and 7 and fertility. Regardless of AFC and CL groups, the differences in number and diameter of AF on Days 0 and 7 and fertility between ovarian responsive and non-responsive does were estimated using Student’s t-test. Continuous values (number and diameter of AF and litter size) were represented mean ± SEM. The differences in ovarian responsive, pregnancy, kidding, single kidding, twin kidding, triplet kidding, and multiple kidding rates among groups were estimated using Chi-square test. Logistic regression methodology, which generated estimates of odds ratios (OR) and 95% confidence intervals (CI), was used to assess the ovarian-important factors (number and diameter of AF and CL appearance) at the onset of the hormonal synchronization for ovulation and fixed-time AI (on Day 0) and the likelihoods of ovarian response to hormonal stimulation and multiple kidding occurrences. Significance was stated when p ≤ 0.0500.
RESULTS
At the time of synchronization (on Day 0), no effect of AFC × CL interaction (p > 0.0500) was observed for all parameters of ovarian AF (Table 1). In the main factor, does having AFC > 3 follicles (AFC II) had a greater number of small AF (p = 0.0001) and a total number of AF (p = 0.0001) on Day 0 than those in does having AFC ≤ 3 follicles (AFC I) on their ovaries (Table 1). Does in AFC II (AFC > 3 follicles) had, on average, the larger size of the largest AF (p = 0.0206) than does with AFC ≤ 3 follicles (AFC I) on their ovaries (Table 1). The number of large AF and diameter of AF were similar (p = 0.3689 and p = 0.1181, respectively) between AFC I and AFC II does (Table 1). Besides, does with CL (CL+) on Day 0 had a larger (p = 0.0422) population of small AF compared with does without CL (CL–) on their ovaries (Table 1). Does in CL– had, on average, larger diameters of AF (p = 0.0005) and the largest AF (p = 0.0001) than does having CL (CL+) (Table 1). The number of large AF (p = 0.6741) and the total number of AF (p = 0.9949) were unaffected by CL status (Table 1).
1) The data (mean ± SEM) of the numbers of small AF and large AF, total number of AF, and diameters of AF and the largest AF at the time of exogenous hormonal trigger (synchronization) and at the time of fixed-time AI in does having AFC ≤3 follicles (AFC I) and with CL (CL+) (Group I), AFC ≤3 follicles (AFC II) and without CL (CL–) (Group II), AFC >3 follicles (AFC II) and with CL (CL+) (Group III), and AFC >3 follicles (AFC II) and without CL (CL–) (Group IV) on their ovaries (n = 128).
At the time of fixed-time AI (on Day 7), no effect of AFC × CL interaction (p > 0.0500) was observed for all parameters of ovarian AF (Table 1). In the main factor, does having AFC > 3 follicles (AFC II) on Day 0 showed a significantly increased (p = 0.0001) population of small AF on Day 7 (Table 1). The large AF and the total population of AF on the day of fixed-time AI were greater (p = 0.0217 and p = 0.0001, respectively) in does having AFC > 3 follicles (group II) than in does having AFC ≤ 3 follicles (group I) on Day 0 (Table 1). On Day 7, there were no differences in the diameters of AF (p = 0.0639) and the largest AF (p = 0.7973) between does in AFC I (AFC ≤ 3 follicles) and AFC II (AFC > 3 follicles) (Table 1). Moreover, no CL status on Day 0 was affected (p > 0.0500) on all parameters of ovarian AF at the time of fixed-time AI (Table 1).
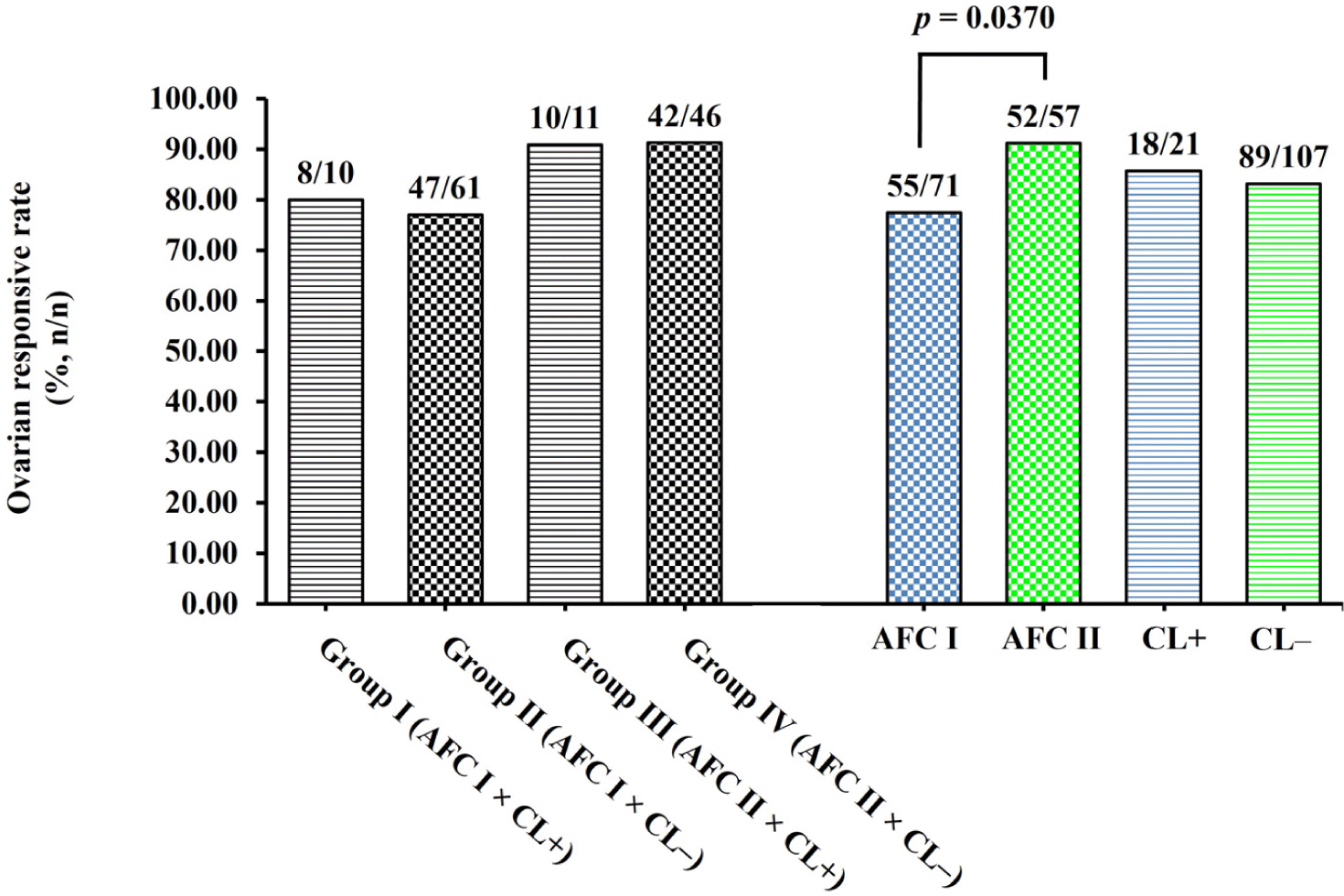
Based on the emergence of the large POFs (> 4 mm) on Day 7, the responsive rate was higher (p = 0.0370) in does having AFC > 3 follicles (AFC II) at the time of synchronization than in does having AFC ≤ 3 follicles (AFC I) on their ovaries (91.23% vs. 77.46%; Fig. 2). In the CL group, the ovarian responsive rate did not differ (p = 0.7750) between CL+ (85.71%) and CL– (83.18%) does (Fig. 2). Moreover, a comparison of the ovarian responsive rate among does in group I (AFC I × CL+; 80.00%), II (AFC I × CL–; 77.05%), III (AFC II × CL+; 90.91%), and IV (AFC II × CL–; 91.30%) did not statistically significant difference (p > 0.0500; Fig. 2).
Regardless of AFC and CL groups, ovarian responsive does had a greater number of small AF (p = 0.0078) and a total number of AF (p = 0.0009) at the time of synchronization (on Day 0) than those in non-responsive does (Table 2). The number of large AF (p = 0.6729) and sizes of AF (p = 0.4161) and the largest AF (p = 0.8491) on Day 0 did not differ between responsive and non-responsive groups (Table 2).
Regardless of AFC and CL groups, responsive does had a greater total number of AF (p = 0.0001), and a greater size of AF (p = 0.0001) and the largest AF (p = 0.0001) on Day 7 than non-responsive does (Table 2). On Day 7, compared with the responsive group, non-responsive does showed a greater (p = 0.0087) number of small AF (Table 2).
In the AFC group, the pregnancy rate of AFC I group (30.99%) was similar to that of the AFC II group (33.33%) (p = 0.7780; Fig. 3A). In the CL group, the pregnancy rate of CL+ does (33.33%) was similar to that of CL– does (31.78%) (p = 0.8890; Fig. 3A). Moreover, the pregnancy rate was also similar to that of does in group I (AFC I × CL+; 30.00%), II (AFC I × CL–; 31.15%), III (AFC II × CL+; 36.36%), and IV (AFC II × CL–; 32.61%) (p > 0.0500; Fig. 3A).
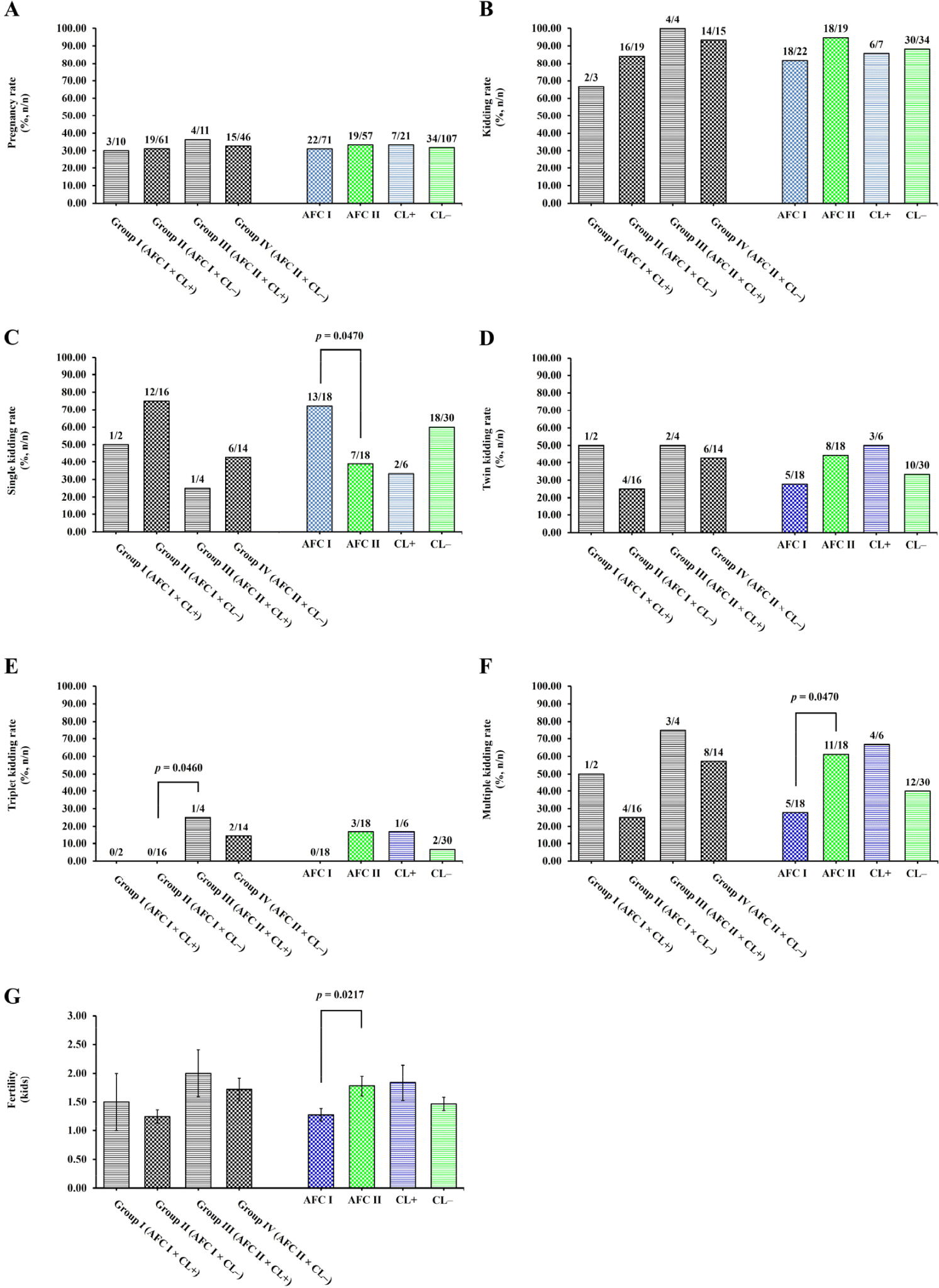
In the AFC group, the kidding rate did not differ (p = 0.2130) between the AFC I (81.82%) and AFC II (94.74%) groups (Fig. 3B). In the CL group, the kidding rate did not differ (p = 0.8550) between CL+ (85.71%) and CL– (88.24%) does (Fig. 3B). Moreover, the kidding rate was similar (p > 0.0500) among does in group I (AFC I × CL+; 66.67%), II (AFC I × CL–; 84.21%), III (AFC II × CL+; 100.00%), and IV (AFC II × CL–; 93.33%) (Fig. 3B).
Interestingly, does with AFC ≤ 3 follicles (AFC I) at the time of synchronization (on Day 0) had a higher single kidding rate (p = 0.0470; Fig. 2C) than does with AFC > 3 follicles (AFC II) on their ovaries (72.22% vs. 38.89%; Fig. 3C). In the CL group, the single kidding rate did not differ (p = 0.2370) between CL+ (33.33%) and CL– (60.00%) does (Fig. 3C). Moreover, the does in group I (AFC I × CL+; 50.00%), II (AFC I × CL–; 75.00%), III (AFC II × CL+; 25.00%), and IV (AFC II × CL–; 42.86%) (Fig. 3C) had a similar single kidding rate (p > 0.0500).
In the AFC group, the twin kidding rate did not differ (p = 0.3050) between the AFC I (27.78%) and AFC II (44.44%) groups (Fig. 3D). In the CL group, the twin kidding rate did not differ (p = 0.4440) between CL+ (50.00%) and CL– (33.33%) does (Fig. 3D). Moreover, the twin kidding rate (p > 0.0500) was similar among does in group I (AFC I × CL+; 50.00%), II (AFC I × CL–; 25.00%), III (AFC II × CL+; 50.00%), and IV (AFC II × CL–; 42.86%) (Fig. 3D).
In the AFC group, the triplet kidding rate did not differ (p = 0.0740) between the AFC I (0.00%) and AFC II (16.67%) groups (Fig. 3E). In the CL group, CL presence (CL+) and CL absence (CL–) did not significantly affect (p = 0.4250) the triplet kidding rate (16.67% vs. 6.67%, respectively; Fig. 3E). Likewise, we found no effect (p > 0.0500) of factor combination on the triplet kidding rate of does in group I (AFC I × CL+; 0.00%) and IV (AFC II × CL–; 14.29%) (Fig. 3E). However, does in group III (AFC II × CL+; 25.00%) had a higher triplet kidding rate (p = 0.0460) than does in group II (AFC I × CL–; 0.00%) (Fig. 3E).
Interestingly, compared to does with AFC ≤ 3 follicles (AFC I), does with AFC > 3 follicles (AFC II) at the time of synchronization (on Day 0) showed significantly (p = 0.0470) increased multiple kidding rate (61.11% vs. 27.78%; Fig. 3F). In the CL group, the multiple kidding rate did not differ (p = 0.2370) between CL+ (66.67%) and CL– (40.00%) does (Fig. 3F). Moreover, no difference in the multiple kidding rate (p > 0.0500) was detected among does in group I (AFC I × CL+; 50.00%), II (AFC I × CL–; 25.00%), III (AFC II × CL+; 75.00%), and IV (AFC II × CL–; 57.14%) (Fig. 3F).
Additionally, does with AFC > 3 follicles (AFC II) at the time of synchronization (on Day 0) had a greater fertility (p = 0.0217) than does with AFC ≤ 3 follicles (AFC I) on their ovaries (1.78 ± 0.17 kids vs. 1.28 ± 0.11 kids; Fig. 3G). In the CL group, fertility did not differ (p = 0.1964) between CL+ (1.83 ± 0.31 kids) and CL– (1.47 ± 0.11 kids) does (Fig. 3G). Fertility was not different (p > 0.0500) among does in group I (AFC I × CL+; 1.50 ± 0.50 kids), II (AFC I × CL–; 1.25 ± 0.11 kids), III (AFC II × CL+; 2.00 ± 0.41 kids), and IV (AFC II × CL–; 1.71 ± 0.19 kids) (Fig. 3G).
Interestingly, the likelihood of follicular response to hormonal synchronization in does was higher (OR = 3.03, p = 0.0370) with greater AFC at the time of synchronization (on Day 0; Table 3). The presence of CL (OR = 0.82, p = 0.7750), numbers of small AF (OR = 2.55, p = 0.0670) and large AF (OR = 0.86, p = 0.8060), and diameters of AF (OR = 0.54, p = 0.2050) and the largest AF (OR = 1.36, p = 0.5250) at the time of synchronization were not associated with ovarian response to hormonal synchronization (Table 3).
Moreover, the multiple kidding rate in does was higher (OR = 4.09, p = 0.0470) among does with greater AFC on Day 0 (Table 4). The presence of CL (OR = 0.33, p = 0.2370), numbers of small AF (OR = 1.86, p = 0.3710) and large AF (OR = 4.09, p = 0.1140), and diameters of AF (OR = 0.54, p = 0.3710) and the largest AF (OR = 1.11, p = 0.8800) at the time of synchronization stimulation were not associated with multiple kidding rate (Table 4).
DISCUSSION
In the current study, the impacts of AFC and CL presence on ovarian response to hormonal stimulation and fertility potential were discovered in inseminated does. To the best of our ability, the present research is the first to explore whether the different number of AF (≥ 2 mm) at the time of synchronization reflects the oocyte-containing follicle supply related to production of multiple large-sized follicles after hormonal synchronization, and the subsequent enhancement of fertility (litter size) in primiparous does. The likelihood of ovarian response to synchronization increased by 3.03 times in does with a greater total number of AF (AFC > 3 follicles) at the time of synchronization. However, it should be noted that the presence (luteal status) or absence (follicular status) of ovarian CL at the time of synchronization did not affect the results of ovarian follicular response to hormonal stimulation. In ruminants, increased ovarian reserve due to genetic selection has been reported to contribute to increased reproductive capacity, which AFC (direct evaluation) and blood level of anti-Müllerian hormone (indirect evaluation) have been extensively investigated as phenotypic biomarkers of ovarian reserve [17,18]. Although the evaluation of AFC has been offered as a tool for indicating better ovarian reserve in mono-ovulatory large ruminants, studies regarding the application of AFC for evaluating ovarian response to hormonal synchronization and fertility potential in does are limited. In the current study, the assessment of ovarian AF population and counting number of ovarian AF as AFC at the time of synchronization are valuable as an alternative indicator for the prediction of ovarian response to stimulation and fertility in inseminated does. Responsive does to hormonal stimulation also had a greater population of ovarian AF at the time of synchronization than non-responsive does. Similar to our findings, other studies emphasize that high AFC is an important indicator to select the sheep with high genetic merit for predictable potential of high ovarian response to hormonal stimulation [19]. The numerically greater population of AF at the onset of the hormonal synchronization and subsequent higher population of large AF at the onset of fixed-time AI were as expected. To explore the possible importance of oocyte-containing follicles in identifying the potential of high responder donor goats, a cohort of small AF was synchronized, and it became clear that the population of small AF was positively associated with the superovulatory response [14]. With respect to ewes, a greater number of ovarian AF at the beginning of hormonal administrations can influence directly in the response to multiple ovulation stimulations [20]. Together, these data emphasize the importance of synchronizing a pool of emerging AF (≥ 2 mm) in does and ewes when performing multiple ovulation stimulations [14,20]. Under the exogenous hormonal control of preovulatory wave emergence and AI in goats, the follicular reserve status prior to starting synthetic P4 trigger is also very important [21,22]. On the day of exogenous hormonal administration (synchronization), the use of synthetic P4 can promote the destruction of previous dominant follicles (DFs) [22] and subsequently a cohort of AF (2–3 mm) emerges that continues directly to grow and differentiate to become a single or multiple POF [13,22–23]. This suggests AF emerging or growing from a pool of growing AF on ovaries, which highlights the importance of AFC (≥ 2 mm) at the time of synchronization. In the present study, compared with does having AFC ≤ 3 follicles (1.69 ± 0.09 follicles), does having AFC > 3 follicles (≥ 2 mm) with 4.02 ± 0.19 follicles of small AF (2–4 mm) at the time of synchronization produced greater large AF (> 4 mm) (2.06 ± 0.13 follicles) on Day 7. Supporting the current study, previous research has revealed that the appearance of a greater population of co-DFs (the presence of two or more large AF in each follicular wave) in poly-ovulatory goats resulted in the population of small AF being counted, as more gonadotrophin-responsive AF within a cohort of small AF tended to proceed to large sizes [24]. In fact, the population of co-DFs in the ovulatory follicular wave is usually associated with the number of ovulations in poly-ovulatory goats [24]. Synchronized ovulatory does had increased the number of co-DFs at the time of finishing the hormonal stimulation compared with non-synchronized does [25]. Moreover, the number of small AF is a mechanism in regulating the number of ovulated oocyte-containing follicles and in contributing the ovulation rate and timing of ovulation in does [26]. Thus, it is quite possible that AFC at the time of synchronization is closely related to the population of future large AF and subsequently increased the number of ovulations in poly-ovulatory goats.
Interestingly, does having AFC > 3 follicles (≥ 2 mm) at the time of synchronization produced greater large AF and greater fertility (1.78 ± 0.17 kids) as compared to does having AFC ≤ 3 follicles (1.28 ± 0.11 kids) submitted to fixed-time AI. The likelihood of multiple kidding increased by 4.09 times in does with a greater total number of AF (AFC > 3 follicles) at the time of synchronization. This implies that AFC at the time of synchronization is closely related to the fertility potential in poly-ovulatory goats. Typically, a greater number of ovulations results in an increase in the litter size (fertility) in sheep and goats [27]. Although the ovulation of large AF was not assessed in the current trial, we suppose, based on earlier findings, that the incidence of high-ovulation rate in high-fecundity sheep is a raised dynamic reserve, resulting in a greater population of AF usable for selection into the ovulatory pool [28–30]. As stated above, our results support the results of previous investigators who have indicated that greater ovulation numbers and fertility (litter size) in poly-ovulatory ewes are demonstrated by the previous appearance of more massive AF on their ovaries [31]. In goat models, the presence of more AF per ovarian tissue and differential expression of intra-ovarian factors may be potential regulators of greater fertility in does [32]. In order to understand the underlying importance population of AF prior to hormonal trigger, melatonin was implanted into goats prior to the onset of the P4-eCG protocol, and it was found that a rise in the populations of AF (2 to < 5 mm) tended to be maximum numbers at the time of exogenous P4 synchronization, which resulted in an increase in fertility [33]. Together, our findings imply that does having AFC > 3 follicles at the time of synchronization develop a greater population of larger AF, suggesting an increase in the development of multiple POFs after completion of the hormonal stimulation period, and promotion of an increased litter size when performing hormonal synchronization for fixed-time AI.
CONCLUSION
A greater number of AF (AFC > 3 follicles) at the time of synchronization can promote not only ovarian response to hormonal stimulation but also fertility in primiparous does following the fixed-time AI program. In the end, ultrasonographic evaluation of AFC is an easy-to-achieve procedure and AFC at the time of synchronization had the potential to be used as an alternative indicator for the prediction of ovarian response to hormonal synchronization and fertility in inseminated does.

