INTRODUCTION
Salmonella is the most prevalent foodborne pathogen and there are approximately 1.4 million Salmonella infections (salmonellosis), 26,500 hospitalizations, and 420 deaths in the United States every year [1]. Salmonella consists of 2,600 serotypes that are divided into two species including S. enterica and S. bongori. Among these, approximately 99% of Salmonella serotypes cause salmonellosis in humans or other mammals [2]. The general symptoms of salmonellosis are characterized as fever, headache, abdominal pain, diarrhea or constipation, vomiting and nausea [3]. The incubation period for salmonellosis is approximately 12 to 72 h, and the infections dosage is reported to be in the range of 103 to 106 organisms depending on the host, Salmonella strain, and food matrix [4]. Approximately 95% of salmonellosis is caused by the consumption of Salmonella-contaminated food products [1,5]. Its outbreaks have been associated with meat, poultry, eggs, milk, and dairy products as well as fresh produce [3]. Notably, S. Typhimurium and S. Enteritidis have been ranked the most frequent Salmonella serotypes in the world (26.1% in USA, 68.3% in Europe, and 64.8% in Korea) [6,7].
In Korea, pork, poultry, and beef are the second largest consumed food group after grains [5]. The consumption of meat and poultry products has steadily increased almost three fold in 2014 when compared to 1985 [5]. This increase of meat and poultry consumption has provided a greater risk of Salmonella exposures to consumers. Since Salmonella existed in the intestinal tracts of meat and poultry, the highest prevalence of Salmonella is in chicken (18.8%–30.3%), followed by pork (5%–21%), and then beef (1.2%–2.0%) [5]. Thus, the Korean government has implemented the HACCP (Hazard Analysis and Critical Control Point) for the pork industry in 2006, beef industry in 2007 and poultry industry in 2008 for the control of Salmonella [5,8]. Therefore, more rapid, sensitive, specific, simple, and hand-held detection methods are required for on-site applicable Salmonella detection [9].
Due to the necessities of the on-site detection method for Salmonella, a phage-based magnetoelastic (ME) biosensor has been developed as a novel, wireless, and direct detection method for on-site use [10,11]. The phage-based ME biosensor is composed of an ME sensor made from a magnetostrictive material that elongates or contracts (vibration) under an applied magnetic field. This vibration will provide the unique resonant frequency value for each ME sensor, which is measured by a network analyzer. Once the ME sensor is immobilized with a specific phage depending on the target pathogen, the phage-immobilized sensor is placed directly on the surface of food. As long as the specific phage binds with the target pathogen on the food, the mass of the ME sensor will increase so that the vibration of ME sensor decreases due to its heaviness. This reduced vibration changes the original resonant frequency of the sensor so that the resonant frequency shift represents the quantified and qualified numbers of target pathogens bound on the ME sensor. ME biosensors have been successfully implemented to detect S. Typhimurium on fresh produce such as spinaches [11], tomatoes [11], and cantaloupes [10]. In their studies, the E2 phage was employed on the ME biosensor method after the E2 phage was genetically modified from a filamentous fd phage. Further studies also reported that the E2 phage-based ME biosensor method was employed to detect S. Typhimurium in milk [12] and egg shells [13]. Another study [14] reported the use of the C4-22 phage, which was genetically modified from filamentous fd phage, for all Salmonella enterica serovars detection on chicken breast. However, most phage-based ME biosensor methods have been focused on the detection of Salmonella on fresh produce rather than meat and poultry products by using genetically modified filamentous type phages.
For the successful application of ME sensor to poultry products, the selection of an appropriate and optimum bio-receptor is necessary to interact with a specific target pathogen that exist in poultry. Phage (bacteriophage) has been gaining more attentions over several bio-receptors including antibodies, cell, enzyme, and DNA. Phages possess excellent specificity, robustness under harsh conditions (pH, temperature), safety, and relatively cheap and easy production processes [9]. Phages, as a natural predator of bacteria, are viruses that infect the bacteria cell specifically. They consist of DNA or RNA enclosed within a protein coat and can undergo either lytic or lysogenic cycles due to its simple structure. During the lytic cycle, a phage attaches to the host bacteria, replicates, and finally lyses the host bacteria to release the phages. Although a lysogenic phage can also integrate with the host bacteria by introducing phage DNA into the bacterial DNA, the prophage exists in the host bacteria without any replication or lysis of phages from the host bacteria. Among these unique properties, the lytic property and specificity of phage can confer an added advantage for the isolation of a specific phage from co-existing and numerous hosts and phages. In addition, the successful investigation of a phage against its target will guarantee the specificity, reliability, and applicability of the ME biosensor method. Due to these advantages of lytic phages, S. Typhimurium-specific phage was isolated and purified from a poultry processing facility and the feasibility of S. Typhimurium-specific phage as a bio-receptor was then investigated for its employment to ME biosensor method.
MATERIALS AND METHODS
Indicator (S. Typhimurium ATCC 13311) and other bacterial strains used in the specificity study were obtained from the American Type of Culture Collection (ATCC), National Culture Collection for Pathogens (NCCP) and a laboratory in the Department of Plant and Food Sciences (DPFS) at Sangmyung University in Chungnam, Korea. Each bacterial strain was cultivated in 25 mL of tryptic soy broth (TSB, Difco Laboratories, Sparks, MD, USA) for 16 h at 37°C with shaking at 110 rpm. After being washed three times with sterilized phosphate-buffered saline (PBS, pH 7.4, Life Technologies, Paisley, UK) buffer by centrifugation at 7,000×g for 4 min, the bacterial cell was suspended in PBS. The concentration of the indicator strain and each bacterial suspension was adjusted to 108 CFU/mL using pre-constructed standard curves.
An aliquot of 25 mL of washing water obtained from a poultry processing facility (Orpum, Sangju, Korea) was mixed with 225 mL of TSB containing 1 mL of the indicator strain suspension. After incubation for 16 h at 37°C with gentle shaking at 160 rpm, the mixtures were centrifuged at 4,000×g for 10 min at 4°C, followed by filtration of the supernatant using a 0.20-μm cellulose acetate filter (Advantec Toyo Kaisha, Tokyo, Japan). An aliquot of 10 µL of the filtrate was dotted on the surface of pre-solidified TA soft agar (4 g agar, 8 g nutrient broth, 5 g NaCl, 0.2 g MgSO4, 0.05 g MnSO4, and 0.15 g CaCl2 per 1 L) containing 200 µL of the indicator strain suspension. The presence of phages in the filtrate was then confirmed using a dot assay [9]. Once the formation of a clear zone was observed, a plaque assay was performed to isolate single phages from the filtrate. An aliquot of 100 µL of the serially diluted filtrate and 200 μL of the indicator strain suspension were inoculated into 4 mL of TA soft agar and then, poured onto a tryptic soy agar (TSA, Difco Laboratories, Sparks, MD, USA) plate. After incubation for 16 h at 37°C, each plaque was picked with a customized tip and eluted with sodium chloride-magnesium sulphate (SM) buffer (50 mM Tris-HCl, 100 mM NaCl, 10 mM MgSO4, pH 7.5) with vigorous agitation for 1 h at 22°C. All of these procedures were repeated until the single phage was isolated [9]. To increase of the concentration of the single phage, 3 mL of TA broth containing 1% (v/v) indicator strain suspension was incubated for 2.5 h at 37°C. The eluted single phage was then added to the TA broth mixture and incubated for 2.5 h at 37°C After centrifugation at 2,400×g for 10 min at 4°C, the supernatant was filtered through a 0.20-μm cellulose acetate filter. The dot assay was performed to determine the phage concentration. All of these procedures were repeated by increasing the volume of TA broth and indicator strain suspension until phage concentration reached up to 1010 plaque forming unit (PFU)/mL. The propagated filtrate was mixed with 10% (w/v) of polyethylene glycol 6000 (Sigma-Aldrich, St. Louis, MO, USA) and 10 mL of 1 M NaCl, followed by precipitation at 4°C for 16 h. After centrifugation at 7,200×g for 20 min at 4°C, the pellet was suspended in SM buffer and the suspension was purified by performing CsCl gradient ultracentrifugation at 39,000×g for 2 h at 4°C. Finally, the layer of bluish opalescent phage band fraction was separated from the centrifuge tube and dialyzed. The final concentration of the purified phage (indicated as KFS-ST hereafter) was determined using plaque assay and stored at 4°C until further use.
Five microliters of KFS-ST (109 PFU/mL) were adsorbed on the top of a carbon-coated copper grid and negatively stained with 4% phosphotungstic acid (Sigma-Aldrich, St. Louis, MO, USA). The KFS-ST was observed using a TEM (H-7100, Hitachi, Tokyo, Japan) at an accelerating voltage of 100 kV with 50,000 to 200,000 × magnifications.
Dot assay was performed to identify the specificity of KFS-ST against various bacterial strains. Once a clear zone was formed, a plaque assay was performed and EOP value was calculated by dividing the number of plaques on each bacterial strain by the number of plaques on the host bacteria.
The temperature and pH stabilities of KFS-ST were investigated by exposing KFS-ST (106 PFU/mL) at various temperatures (−70°C, −20°C, 4°C, 10°C, 20°C, 30°C, 40°C, 50°C, 60°C, and 70°C) and pHs (1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, and 12) using a plaque assay. An aliquot of 100 μL of KFS-ST was added into 900 μL of TSB and incubated at various temperatures for 1 h and various pHs at 22°C for 1 h, respectively.
One percent of the host suspension was sub-inoculated into fresh TSB medium and incubated until 1.0 of OD640. KFS-ST was added at the multiplicity of infection of 0.01 and incubated for 25 min at 37°C to allow absorption of KFS-ST to the surface of the host cell, which was determined by the previous study. After adsorption, the mixture was centrifuged at 11,400×g for 10 min at 4°C to remove the unabsorbed KFS-ST. The pellet was then resuspended with TSB and incubated at 37°C. At every 5-min interval, 1 mL of the incubated mixture was collected and evenly divided into two portions to add 1% (v/v) of chloroform for the release of intracellular KFS-ST. After 30 s, a plaque assay was performed to determine the eclipse time, latent time, and burst size of KFS-ST.
KFS-ST (109 PFU/mL) in TA broth was stored at −70°C, −20°C, 4°C, and 22°C to investigate the storage stability of phage. In order to prevent any damage in the freezing process, 15% (v/v) glycerol was added into TA broth and stored at −70°C and −20°C, respectively. At every 2-week interval, each sample was collected and plaque assay was performed to measure the stability of KFS-ST during the 12 weeks under various storage temperatures.
The experiment was replicated at least three times and the experimental results were expressed as mean ± SD. All statistical analyses were performed with GraphPad and InStatV.3 programs (GraphPad, San Diego, CA, USA). The student’s paired t-test for two groups and the one-way analysis of variance (ANOVA) among more than two groups were used for the comparison. The significance level was determined at p < 0.05 or p < 0.001.
RESULTS AND DISCUSSION
KFS-ST was isolated and purified from washing water used in a poultry processing facility with a concentration of (11.47 ± 0.25) Log PFU/mL. The isolated and purified single phage was named KFS-ST following the recommendation of Ackermann’s study [15], implying that the ST indicated the host genus and species name of Salmonella. For the morphological characterization of KFS-ST, a TEM image showed that KFS-ST has an icosahedral head and a non-contractile tail (Fig. 1). The width and length of the icosahedral head and tail were 59.90 ± 3.07 nm and 56.91 ± 2.90 nm, and 13.77 ± 1.78 nm and 225.49 ± 2.67 nm, respectively. In addition, it was confirmed that KFS-ST phage had a non-contractile tail because 20 TEM images showed that the tail length was almost the same (Fig. 1B). These results indicate that KFS-ST belongs to the family of Siphoviridae in the order of Caudovirales. To our knowledge, 31 S. Typhimurium-specific phages belonging to the order of Caudovirales have been reported including 15 Siphoviridae, 9 Myoviridae and 7 Podoviridae (Table 1). Among these, 18 phages were isolated and purified from an animal itself or animal-related sewages such as cattle, swine and poultry. The head length of KFS-ST was similar to that of most reported phages of Siphoviridae, such as fmb-p1 [16], φSTIz1 [17] and ES18 [18]. Interestingly, the tail length of our phage was the longest among others reported Siphoviridae phages, which was a very unique morphological characteristic. These data support the novelty of our KFS-ST in regards to the morphological aspect.

| Classification | Phage name | Origin | Morphology | Stability | One-step | Ref. | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Head | Tail | pH | Temp. | Eclipse | Latent | Burst | ||||
| Siphoviridae | EPS7 | Sewage | 65 | 185 | - | - | - | - | - | [32] |
| ES18 | - | 56 | 210 | - | - | - | - | - | [18] | |
| FGCSSa2 | Sewage | 66 | 112 | - | - | - | - | - | [21] | |
| fmb-p1 | Sewage (duck farm) | 57 | 171 | 4–11 | 4–70 | - | 20 | 77 | [16] | |
| P164L1 | Sewage | 73 | 178 | - | - | - | - | - | [33] | |
| PBST10, PBST13, PBST32, PBST35 | - | 45–66 | 94–186 | 4–11 | 30–60 | - | - | - | [19] | |
| phSE-1 | Sewage | 67 | 152 | - | - | 20 | 30 | 28 | [23] | |
| phSE-2 | Sewage | 67 | 177 | - | - | 20 | 40 | 53 | [23] | |
| phSE-5 | Sewage | 67 | 160 | - | - | 15 | 30 | 149 | [23] | |
| SPN3UB | - | - | - | - | - | - | - | - | [34] | |
| SSU5 | Sewage | 70 | 220 | - | - | - | - | - | [35] | |
| STA1, STA2, STA3, STA9 | Sewage (cattle and swine) | - | - | - | - | - | - | - | [22] | |
| Φst1 | Chicken excreta | 67 | 173 | - | - | - | 40 | 22 | [36] | |
| φSTIz1 | Sewage (cattle and poultry farm) | 53 | 190 | 4–11 | 4–60 | - | - | - | [17] | |
| ΦStp1 | Chicken intestinal | 77 | 172 | - | - | - | 30 | 37 | [37] | |
| Myoviridae | Fels2 | - | 55 | 110 | - | - | - | - | - | [38] |
| FGCSSa1 | Sewage | 107 | 123 | - | - | - | 50 | 139 | [21] | |
| SFP10 | Slurry | 69 | 131 | 4–10 | 20–60 | 5 | 25 | 200 | [24] | |
| SPN3US | - | - | - | - | - | - | - | - | [39] | |
| SPT1 | 73 | 113 | - | - | - | - | - | - | [40] | |
| ST64B | - | - | - | - | - | - | - | - | [41] | |
| UAB_Phi87 | Chicken cloacae and pig rectal | 68 | 114 | - | - | - | - | - | [42] | |
| Vi phage E1 | - | 55 | 205 | - | - | - | - | - | [43] | |
| ΦSH19 | Sewage | - | - | - | - | - | - | - | [44] | |
| Podoviridae | F3, F4, F5, F6, F7, F8 | Pig farms | 64 | 16 | - | - | - | - | - | [45] |
| P22 | - | 77 | - | 4–11 | 30–60 | - | - | - | [19] | |
| P22-B1 | - | 55 | - | 4–11 | 30–60 | - | - | - | [19] | |
| SPN1S | - | - | - | - | - | - | - | - | [46] | |
| SPN9CC | Chicken skin | - | - | - | - | 15 | 30 | 220 | [26] | |
| UAB_Phi20 | Chicken cloacae and pig rectal | 60 | 13 | - | - | - | - | - | [42] | |
| UAB_Phi78 | Chicken cloacae and pig rectal | 66 | 14 | - | - | - | - | - | [42] | |
The specificity of the bio-receptor in an analytical biosensor method is defined as the ability to identify the target pathogen among others in a sample and is a very important factor for consideration [9]. Although KFS-ST was purified with great concentration, the specificity needs to be guaranteed for its further use. KFS-ST exhibited clear zones against only S. Typhimurium and S. Enteritidis among the 12 tested Salmonella strains. More interestingly, no clear zones were identified against S. Heidelberg, S. Hartford, S. Mission, S. Montevideo, S. Salamae, and S. Senftenberg, which are considered the most problematic Salmonella stains reported from the CDC [3]. More importantly, KFS-ST did not exhibit any cross-genus specificity against 27 other non-Salmonella pathogens. Therefore, it was concluded that KFS-ST had great specificity against three strains of S. Typhimurium and S. Enteritidis with an EOP of approximately 1 (Table 2).
The specificity of phages is associated with the their capacity to bind to host receptors such as flagella, capsules, lipopolysaccharides and proteins in the outer membrane or slime layer [19]. Although several studies have confirmed the specificity of phage, there is no clear definition of the narrow- and broad-specificity of phages [20]. Carey-Smith et al. [21] reported that FGCSSa2 (S. Typhimurium-specific phage) showed species-level specificity due to its lytic activity against only three strains of S. Typhimurium among eight other strains of Salmonella including S. Typhimurium, S. Infentis, S. Menston S. Saintpaul, S. Entertidis and two Escherichia coli strains. Akhtar et al. [22] demonstrated that STA3, STA9 and STA10 (S. Typhimurium-specific phages) had species-level specificities against only S. Typhimurium and S. Heidelberg among 27 strains of Salmonella, which was very similar to the results of our study. In addition, Wang et al.’s study [16] exhibited more broad species-level specificity by demonstrating that fmb-p1 (S. Typhimurium-specific phage) was specific to 10 of 34 Salmonella strains including S. Typhimurium, S. Enteritidis, S. Agona, S. Anatum, S. Heidelberg, S. Miami, S. Paratyphi-C, and S. Thompson without any specificities against non-Salmonella strains including Listeria monocytogenes, E. coli, Staphylococcus aureus and Bacillus subtilis. Meanwhile, Pereira et al. [23] reported that phSE-1 and phSE-2 (S. Typhimurium-specific phages) showed genus-level specificity against 27 strains of 42 foodborne pathogens, including three strains of S. Typhimurium strains, five strains of S. Enteritidis strains, eleven strains of E. coli strains, two strains of Citrobacter freundii strains, four species of Providencia, Klebsiella pneumonia, and Enterobacter cloacae. In addition, phSE-3 (S. Typhimurium-specific phages) showed genus-level specificity against 25 of 42 strains including three strains of S. Typhimurium, five strains of S. Enteritidis, ten strains of E. coli, two strains of Citrobacter freundii, and four species of Providencia sp. and Klebsiella pneumonia. Furthermore, Park et al. [24] reported the genus-level specificity of SFP10 (S. Typhimurium-specific phages) infecting seven strains of S. Typhimurium and S. Enteritidis, two strains of S. Paratyphi and S. Dublin and four strains of E. coli. Overall, KFS-ST exhibited great narrow specificity against only S. Typhimurium and Enteritidis, which are considered major Salmonella strains in meat and poultry [7]. This excellent specificity will satisfy one of requirements for selecting a bio-receptor for use in the ME biosensor method.
A phage-based ME biosensor may be exposed to various environmental conditions for the detection of S. Typhimurium and S. Enteritidis on poultry products that undergo various harsh conditions during processing and storage. Since major components of phage are nucleic acid and structural proteins, the structural proteins and DNA of phage could be damaged under harsh external conditions, such as extreme temperature and pH [25]. As shown in Fig. 2A, KFS-ST sustained its concentration at a temperature range between 4°C and 50°C. After that, the concentration of KFS-ST significantly decreased at −20°C (17.7% reduction), −70°C (43.5% reduction), and 60°C (30.5% reduction) (p < 0.05). Jonczyk et al. [25] reported that the phages that belonged to the Siphoviridae family were the most resistant to adverse conditions. In terms of temperature stability, φSTIz1 (S. Typhimurium-specific phages) [17] exhibited relatively similar temperature stability (4°C–60°C) with our results. Meanwhile, PBST10, PBST13, PBST32 as S. Typhimurium-specific phages [19] exhibited very narrow temperature ranges of 30°C–60°C. Interestingly, fmb-p1 [16] also represented a further narrow temperature stability of 40°C–75°C.
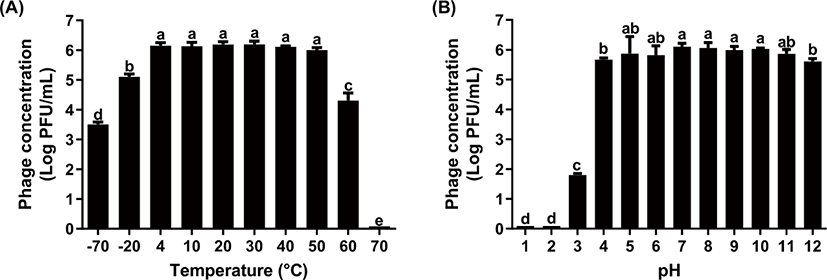
In regards to pH, the stability of phage confirmed by the concentration of KFS-ST phage was sustained at the pH range of 4 to 12 (Fig. 2B). Only approximately 8.1% reduction of phage concentration was observed in this range, although the KFS-ST concentration was significantly decreased at pH 4 and pH 12 (p < 0.05). However, the phage concentration was reduced continuously up to ~29.5% at a pH 3 and finally lost the concentration completely at pH 1 and pH 2 (~99.9% reduction) (p < 0.05). Based on previous studies, φSTIz1 [17], PBST10 [19], PBST13 [19], PBST32 [19], PBST35 [19] and fmb-p1 [16] showed similar pH stability with our studies. Although no clear standards for stability has been established for the optimum phage, our results highlight the relatively broad temperature stability and competitive pH stability of KFS-ST for practical use under various environmental conditions.
Most phages (approximately 90%) that are isolated from the environment are lytic and tailed phages [9]. Although a lytic property is necessary for identifying and purifying of phages from the environment, it is not always beneficial for use as a bio-receptor. Specifically, the ME biosensor is dependent on a mass change on the sensor so that the immobilized phages on the sensor must be sustained without any rupture of bacteria due to its lytic activity. Thus, the eclipse time, latent time and burst size of KFS-ST phage were determined using a one-step growth curve analysis (Fig. 3). The eclipse time of phage indicates the time interval between infection and replication of the phage inside the host cell; it was 10 min for KFS-ST after 25-min adsorption time. The latent time, which is the time interval between the infection and the release of the first phage from the host, was 25 min after adsorption. The burst size of KFS-ST from a single infected bacterial cell was determined as 26 PFU/one infected cell.
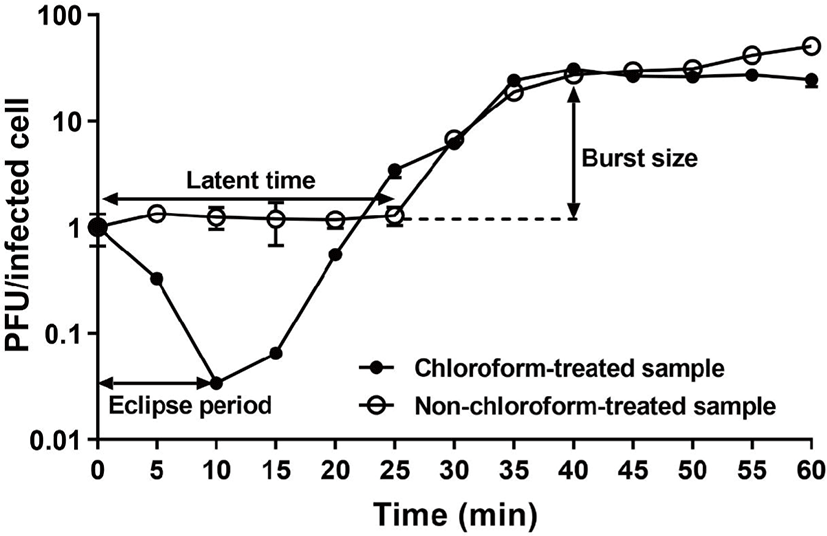
Once phage adsorbed, the latent time and eclipse time were compared with those of other studies. The eclipse time of KFS-ST was longer than that of SFP10 phage, which had an eclipse time of 5 min [24]. On the contrary, our eclipse time was shorter than that of other S. Typhimurium-specific phages including SPN9CC (15 min) [26], phSE-1 (20 min), phSE-2 (20 min), and phSE-5 (15 min) [23]. When employing KFS-ST as a bio-receptor on ME biosensor, the best desirable detection time is within the adsorption time. Just in case the lytic phage goes into the bacterial cell, the overall detection time of the ME biosensor needs to be complete before the combined time of the adsorption time and eclipse time. Our previous studies have confirmed that the total detection time of the ME biosensor including the placement and incubation of phage-immobilized sensor on the surface of the food along with detection by a network analyzer, was 15 min, provided the phage was previously immobilized on the ME sensor. Therefore, the adsorption and eclipse time of KFS-ST provides sufficient time for the phages to bind with Salmonella on the surface of contaminated poultry and meat for its quantification and qualification without any mass loss.
Antibodies are the most popularly employed bio-receptor, however, their most considerable limitations include their need for storage at cold temperatures and their average shelf life of one month [27]. By contrast phage possess excellent stability at relatively broad temperature ranges during long periods [28]. Thus, the influence of storage period and temperature were investigated by measuring the concentration of KFS-ST after exposure to various storage conditions over a period of 12 weeks (Table 3). As long as their storage period increased, their concentration decreased with a similar stability pattern among various temperatures. Although phage concentration decreased significantly, their final decreases during the 12 weeks were only ~0.31 log, ~0.28 log, ~0.25 log and ~0.25 log at temperatures of –70°C, –20°C, 4°C, and 22°C, respectively (p < 0.05). When compared with various temperatures over the same storage period, there were no significant differences among them, except for –70°C at 12 weeks (p < 0.05). Therefore, our phage exhibited a robust and broad storage stability including deep freezing, freezing, cold and room temperatures, suggesting that it has a very competitive factor for use as a novel bio-receptor.
Petsong et al. [29] reported that the concentration of Salmonella-specific phage cocktail including SLP004, SLP005 and SLP050 was reduced by ~1 log after 5 weeks and ~4 log after 12 weeks at 4°C. In the meantime, the phage cocktail lost its concentration completely after 4 weeks at 25°C. The room temperature accelerated the reduction of the phage cocktail concentration more than cold temperature in this study. The other study [28] also confirmed the lower feasibility of Yersiniaenterocolitica-specific phage when exposed to room temperature rather than deep freezing, freezing, and cold temperatures. In fact, most of the several commercial phage cocktails approved by the FDA were sold under cold storage conditions (2°C–8°C) [30]. Even another study [31] has confirmed that the best condition for phage storage is a cold temperature range in most cases. The fact that the stability of KFS-ST at room temperature was maintained at a similar level with cold and freezing temperatures during the whole storage period provides a considerable advantage for the process of carrying and storing KFS-ST for its practical aspect as a novel bio-receptor.
CONCLUSION
This study highlights the feasibility of KFS-ST isolated from a poultry processing facility to search for host-specific phage as a novel bio-receptor that will be used in the ME biosensor method for the detection of S. Typhimurium and S. Enteritidis in poultry products. The phage exhibited morphological characteristics, such as a relatively longer non-contractile tail with an icosahedral head, and was highly specific to S. Typhimurium and S. Enteritidis only. Along with excellent specificity, the phage exhibited relatively broad temperature and competitive pH stabilities. Furthermore, a one-step growth analysis of KFS-ST guaranteed that it provided enough time to bind with Salmonella without any phage-related ruptures for its quantification. Additionally, it possessed excellent storage stability even at room temperature, which was a highly competitive factor for commercial use. In fact, a few studies have employed Salmonella-specific phages on the ME biosensor method for the detection of Salmonella on meat and poultry products. These phages, including E2 phage [12,13] and C4-22 phage [14], were obtained from genetic modification technology for improving their original binding affinity and specificity. Although all of these phages were able to detect Salmonella successfully, the phage typing technology required time- and labor-consuming procedures. Furthermore, their filamentous morphologies induced the aggregation problem when employed to the ME biosensor method. In order to overcome these limitations of previously employed phages, a highly specific tailed-type KFS-ST phage was isolated and purified by using the specific interaction between the phage with a target pathogen existing in poultry. The practical feasibility of this phage was then confirmed in this study. Finally, this study will open the door for further practical developments of tailed-type KFS-ST as a novel bio-receptor by employing it to ME biosensor method for detection of Salmonella on meat and poultry product.

