INTRODUCTION
The gastrointestinal (GI) tract of humans harbors diverse bacteria, which have mutually evolved with the host [1–4]. These bacteria aid the host in digesting carbohydrates, resisting infections, and regulating the immune system [5,6]. Lactic acid bacteria (LAB), which produce lactic acid as the sole or main product of carbohydrate metabolism, are well-known probiotics. They are found in the GI tract, oral cavity, saliva, and breast milk, ranging from humans to animals. They are also abundant in some plants and fermented foods. One species of LAB, Lactobacillus reuteri (L. reuteri), first isolated in 1962, helps in the prevention and/or amelioration of multiple disorders in the host [7].
Since the effects of probiotics depend on the genera, species, and strain, it is essential to verify their identity, characterization, and safety. The main criteria for selecting a probiotic strain is its safety, which includes the verification of hemolytic activity, biogenic amine production, and antibiotic resistance, as well as its adhesion to the intestinal mucosa [8–10].
Probiotics are generally recognized as safe (GRAS) because of their long-term use in fermented foods and dairy products [11]. Although the efficacy of probiotics has been studied extensively, the adverse effects of probiotics have been documented. These include sepsis in cardio-surgical patients, anaphylactic reactions, systemic fungemia, and the risk of antibiotic resistance gene transfer to pathogenic bacteria [8]. Hence, the United Nations Food and Agriculture Organization (FAO) and World Health Organization (WHO) have suggested that it is important to perform a minimum safety assessment for potential hemolysis, toxin production, and antibiotic resistance before its use in humans [12].
The mucus layer of the GI tract provides the first contact point between the intestinal microbiota and the host, creating a fluid environment. The GI tract is classified into the lower and upper GI tract, consisting of intestinal epithelial cells (IEC), including paneth cells and goblet cells [13]. Probiotics can regulate IEC function by various ways, including improving microbial colonization, enhancing tight junction (TJ) and mucin production, promoting antimicrobial peptide production, and modulating the immune response of the host. To confer health benefits, probiotics must interact sufficiently with the host by binding to the mucus layer of the GI tract [14–18].
Inflammatory bowel disease (IBD) is a chronic disorder of the GI tract and can be classified into two clinical entities: Crohn’s disease (CD) or ulcerative colitis (UC) [19,20]. The pathogenic mechanisms of IBD mainly involve immune cells, including activated dendritic cells (DCs) and macrophages, via the secretion of cytokines, such as IL-1β and TNF-α [21]. IL-1β is a proinflammatory cytokine that plays a critical role in a wide range of systemic and local effects. Notably, increased IL-1β expression in patients with CD and UC is significantly associated with the severity of intestinal inflammation, leading to a defective intestinal epithelial TJ barrier [19,20,22]. Some studies have shown that lipopolysaccharide (LPS)-induced HT-29, with the epithelial morphology of colonic adenocarcinoma cells, secrete Th2 cytokines, including IL-1β, IL-6, TNF-α, and IL-4, mediated by the NF-κB and/or MAP kinase pathway [23–25]. However, the inflammatory response in IL-1β-induced HT-29 via the MAP kinase pathway has yet to be elucidated.
In this study, we evaluated the potential risk of RR-LM1071 isolated from breast milk by testing its hemolytic activity, biogenic amine production, and antibiotic resistance. To propose RR-LM1071 as an effective probiotic for regulating intestinal inflammation, we tested its adhesive properties and anti-inflammatory effects on HT-29 cells.
MATERIALS AND METHODS
RR-LM1071 (KCCM 12650P) used in this study was isolated from human breast milk of theree voluteer mothers, who are living in rural areas in South Korea. The mothers were between 25 and 40 years old, and samples were donated after obtaining their written consent. After isolation, the strain was stored at ‒80°C in a cryoprotectant mixture of 15% (w/v) skim milk and 10% glycerol until further use. RR-LM1071 was identified using 16S rDNA sequencing (Macrogen, Korea). Finally, the sequencing results were compared against reference sequences from the GenBank database (http://www.ncbi.nlm.nih.gov/Blastn/).
For the hemolysis test, RR-LM1071 was grown in MRS broth at 37°C for 18 h, and then streaked on agar plates containing 5% sheep blood. The plates were incubated at 37°C for 24 h. The formation of clear zones around colonies was determined as β-hemolysis. Escherichia coli ATCC 25922 was used as a positive control.
According to Bover-Cid and Holzapfel, RR-LM1071 was cultured in MRS at 37°C for 24 h. Biogenic amine production was determined using a special agar medium with lysine, tyrosine, histamine, and ornithine as the precursors for the synthesis of cadaverine, tyramine, histamine, and putrescine, respectively, at 37°C for 48 h. E. coli ATCC 25922 was used as a positive control.
For the antibiotic susceptibility test, RR-LM1071 was cultured at 37°C for 48 h in a broth containing agar IST (90%) and MRS (10%), according to the Clinical and Laboratory Standards Institute (CLSI) recommendations. The minimal inhibitory concentration (MIC) was determined using the agar dilution method. Briefly, a single colony was suspended in phosphate buffered saline (PBS) at an optical density (OD) of 0.01, and inoculated on plates using a multipin-inoculator at 37°C for 24 h. The lowest concentration of antibiotics that suppressed visible growth was denoted as the MIC value, as per European Food Safety Authority (EFSA) recommendations.
HT-29 cells, human colon adenocarcinoma, were purchased from American Type Culture Collection (ATCC). The cells were cultured in RPMI 1640 medium supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 100 mg/mL streptomycin at 37°C in an atmosphere of 5% CO2. To evaluate bacterial adhesion, HT-29 monolayers were prepared on 24-well tissue culture plates and seeded with 1 × 105 cells per well. The culture medium was replaced with fresh medium after 24 h.
The cultured bacterial cells were diluted in peptone water at 0.1 OD, and the diluted cells (1 mL) were treated on HT-29 cells for 2 h at 37°C in an atmosphere of 5% CO2. After incubation, the HT-29 cells were washed by PBS to remove unadhered bacteria and treated with 0.25% trypsin-EDTA solution. A bacterial cell count was performed using serially diluted samples via the pour plate method. The diluted samples (1 mL) were placed on a plate before the MRS agar was poured. The results are expressed as a relative percentage of adhesion (CFU adhered bacterial / CFU added bacteria × 100).
Microbial adhesion to solvents (MATS) was measured according to Kos et al. [26] with some modifications. Briefly, the bacterial cells were cultured in MRS broth for 24 h and harvested by centrifugation at 10,000×g for 2 min. The cell pellets were washed by PBS and diluted approximately to 1.0 × 108 CFU/mL at a final OD of 1.0 ± 0.05 at 600 nm (A0). The cell suspension was mixed well by vortexing with the same volume of solvent, and then allowed to stand for 30 min at room temperature. The absorbance of the aqueous layer was evaluated at 600 nm (A1). The percentage of bacterial adhesion to the solvent was calculated as (1 – A1 / A0) × 100.
To determine the expression of cytokines at the mRNA level, HT-29 cells were seeded at 1 × 106 cells per well in 6-well plates for 24 h. The next day, LGG and RR-LM1071 were incubated at a non-toxic dose (1 × 108 CFU/mL) in the presence of IL-1β (10 ng/mL) for 24 h. Total RNA was extracted using RNAiso plus (TaKaRa, Shiga, Japan) solution, followed by the reverse transcription of 2 µg of total RNA into complementary DNA (cDNA) using PrimeScriptTM 1st strand cDNA synthesis kit (TaKaRa). To amplify cDNA, we used SYBR Green PCRmaster mix (Applied Biosystems, Foster City, CA, USA). The primers used in this study were as follows: IL-6-specific primer pairs consisted of 5’-TCAATGAGGGAGCTTGCCTG-3’ (forward), 5’-GATGAGTTGTCATGTCCTGC-3’ (reverse); TNF-α-specific primer pairs consisted of 5’-ACAAGCCTGTAGCCCATGTT-3’ (forward), 5’-AAAGTAGACCTGCCCAGACT-3’ (reverse); IL-4-specific primer pairs consisted of 5’-CCCCTCTGTTCTTCCTGCT-3’ (forward), 5’-TCGTCTTTAGCCTTTCCAAG-3’ (reverse); the internal control β-actin-specific primer pairs consisted of 5’-ATTGCCGACAGGATGCAGAA-3’ (forward), 5’-AAGCATTTGCGGTGGACGAT-3’ (reverse). The amplication conditions were as follows: 50°C for 2 min and 95°C for 10 min as the hold stage, followed by 95°C for 15 s and 55°C for 1 min as the PCR stage with 40 cycles. To analyze the amplified data, the ΔΔCt method was employed.
HT-29 cells were seeded at 1 × 106 cells per well in 6-well plates for 24 h. After 24 h of incubation, LGG and RR-LM1071 were pre-treated for 1 h followed by IL-1β (10 ng/mL) stimulation for 1 h. The levels of p44/42 MAP kinase were evaluated using an automated JESS-ProteinSimple instrument, according to the manufacturer’s protocol (Minneapolis, MN, USA). Briefly, equal amounts of proteins were mixed with a fluorescence master mix at a ratio of 4:1, and then denatured at 95°C for 10 min. The prepared protein samples, rabbit anti-p44/42 MAP kinase antibody (Cell Signaling Technology, Danvers, MA, USA), and rabbit anti-α-tublin antibody (Cell Signaling Technology), as a loading control, were loaded into the prefilled microplates, and then incubated with HRP-conjugated secondary antibodies. The chemiluminescent signal was determined using a charge-coupled device camera with the luminol-S/peroxide substrate reacted.
RESULTS AND DISCUSSION
We isolated various lactic acid bacteria from human breast milk and denoted them as Bifidobacterium bifidum LM1108, Bifidobacterium bifidum LM1109, Lactobacillus gasseri LM1065, and Lactobacillus reuteri LM1071, respectively. Since L. reuteri is known for playing a critical role in the production of antimicrobial molecules, the modulation of the host immune system, and the reinforcement of the intestinal barrier [1,7], the L. reuteri strain was selected for further analysis. 16S rDNA gene sequence alignment revealed that RR-LM1071 was most closely related to Lactobacillus reuteri DSM 20016 (99.87% similarity).
Hemolysis is a process by which red blood cells (RBCs) are destroyed. α-Hemolysis is considered as a partial breakdown of the hemoglobin in RBCs, while β-hemolysis and γ-hemolysis are considered as complete or no breakdown, respectively. Pathogenic bacteria or fungi that cause hemolysis can produce hemolysins that damage the cell membrane of RBCs, leading to sepsis [12]. Hence, it is necessary to evaluate whether probiotics have hemolytic activity. As shown in Table 1, RR-LM1071 showed no hemolytic activity compared to the positive control, E. coli ATCC 25922, which showed β-hemolysis.
| Strain | Hemolysis activity | Biogenic amine | |||
|---|---|---|---|---|---|
| Histamine | Cadaverine | Tyramine | Putrescine | ||
| RR-LM1071 | Gamma | − | − | − | − |
| E. coli ATCC 25922 | Beta | + | + | + | + |
Biogenic amines are low molecular weight organic nitrogenous compounds formed by the removal of α-carboxyl groups from amino acids, which could induce toxicological events, including headache, hypertension, pyrexia, or heart disease. Thus, determining whether probiotics produce these biogenic amines is one of the critical points in safety [27]. The key biogenic amines are histamine, cadaverine, tyramine, and putrescine, which are synthesized by histidine, lysine, tyrosine, and ornithine, respectively [28,29]. As shown in Table 1, RR-LM1071 was found to be negative for biogenic amine production.
Some studies have indicated that commensal bacteria may store antibiotic resistance genes, as seen in pathogenic bacteria [11]. These bacteria can transfer antibiotic resistance genes to pathogenic bacteria. Therefore, it is necessary to evaluate the antibiotic susceptibility of probiotics [30]. As shown in Table 2, RR-LM1071 was found to be susceptible to ampicillin, erythromycin, gentamicin, tetracycline, streptomycin, vancomycin, chloramphenicol, kanamycin, and clindamycin, as per the EFSA criteria. Therefore, RR-LM1071 does not appear to pose a notable threat that it transfers antibiotic resistance genes to other intestinal bacteria, especially pathogenic bacteria. However, additional studies would be needed to verify whether it has resistance genes, such as through genomic analyses.
The GI tract of humans harbors ~14 Log bacteria, consisting of over 1,000 species. Bacterial adhesion to the mucus layer of the GI tract is a critical prerequisite for modulating immune responses and stabilizing the mucosal barrier of the gut. As previously reported, Lactobacillus rhamnosus GG (LGG) possesses mucus-binding properties [31,32]. For this reason, LGG is the desired reference for evaluating the adhesive properties of other strains. When we evaluated the adhesive properties of HT-29 cells in vitro, the adhesive properties of RR-LM1071 were found to be higher than those of LGG (Fig. 1A). In a previous study, genomic analysis showed that LGG has pilus gene clusters (spaCBA and spaFED), which encode diverse pili, wherein SpaA and SpaD form the pilus shaft, SpaB and SpaE adorn the pilus backbone, and SpaC and SpaF serve as adhesive components [31,33]. One limitation of this study is the cell-based analysis used to demonstrate the adhesive properties of RR-LM1071. Future studies should explore whether this is attributed to gene cluster and/or gene expression using genomic, transcriptomic, and proteomic analyses.
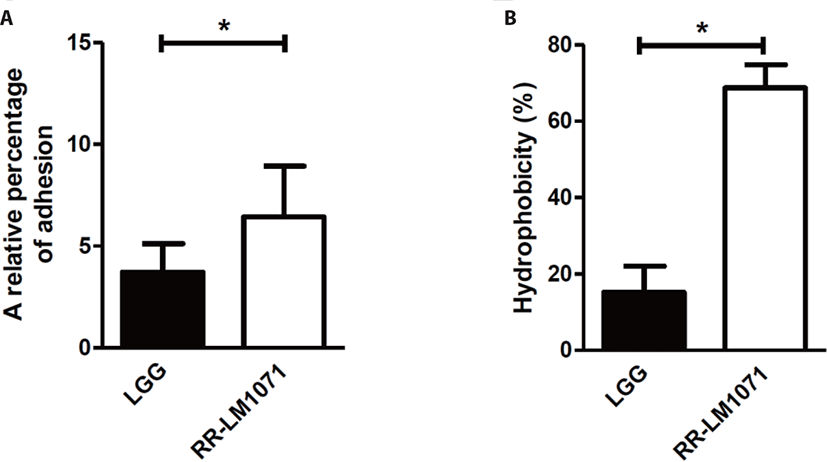
The inhibition of pathogens is attributed to the physicochemical properties of the bacterial cell wall, which has adhesive effects. The adhesive properties are affected by various interactions, such as van der Waals forces, Lewis acid-base interactions, and hydrophobic and electrostatic interactions [34,35]. Hydrophobicity is the key to ensuring the first contact between a bacterial cell and mucous or epithelial cells [35,36]. As a result of MATS, RR-LM1071 showed a stronger hydrophobicity than LGG in a hexadecane solvent (Fig. 1B), while there was no differences between the hydrophobicity of the strains in a xylene solvent (data not shown).
The bacterial cell wall includes a thick layer of peptidoglycan, teichoic acid, lipoteichoic acid, surface-exposed proteins, and polysaccharides [26]. The hydrophobicity of probiotics is affected by proteins expressed on the cell surface, such as the Pilin SpaC subunit and MabA. The Pilin SpaC subunit promotes binding to human mucin and IEC. In addition, MabA, an LPXTG cell wall-anchoring protein, helps bacteria adhere to epithelial cells, as well as in biofilm formation [37]. Although the results (Fig. 1) may be ascribed to the rate and amount of cell surface protein expression, further studies will be needed to fully understand the correlation between adhesive properties and hydrophobicity.
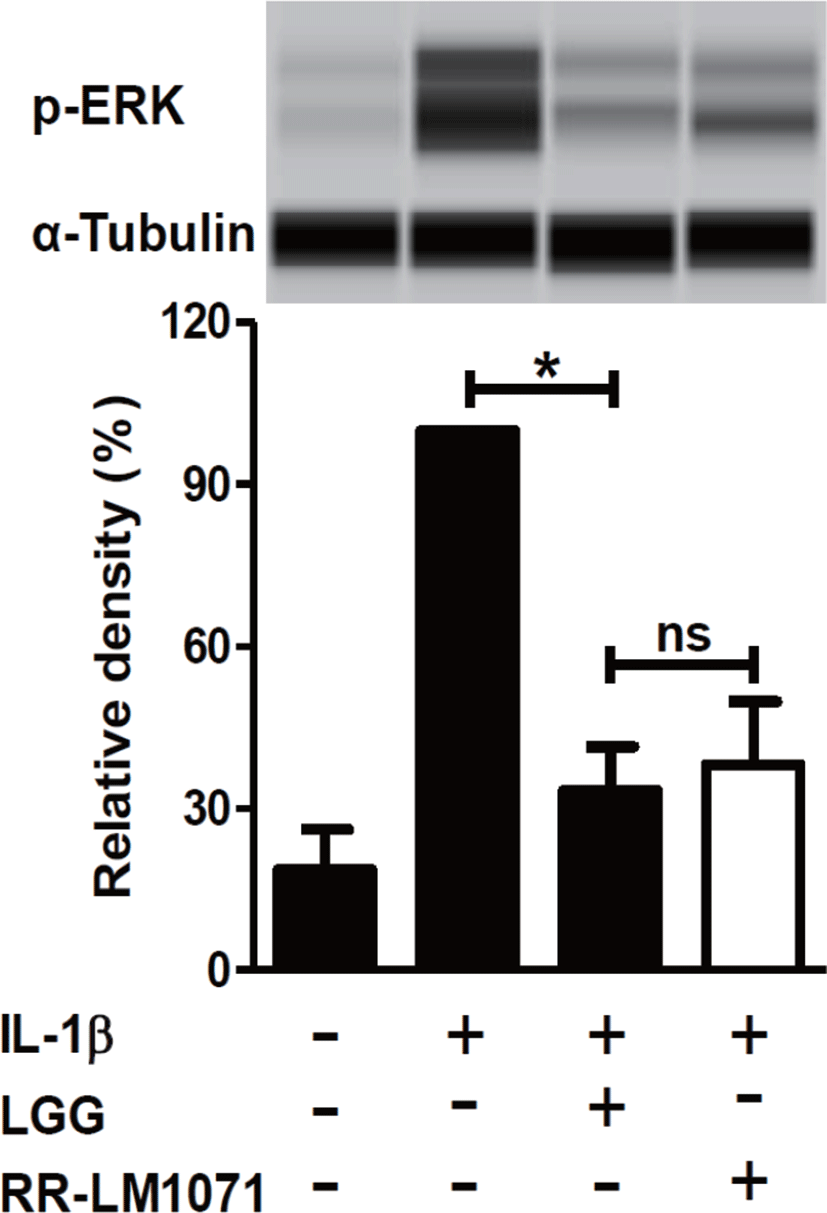
LGG is known to have possible benefits in inflammatory diseases, including UC, acute gastroenteritis, and atopic dermatitis [38]. IBD, including CD and UC, is a multifactorial disease that leads to the loss of immune tolerance mediated by various molecules. Cytokines are one of the most important factors in the immune network between immune cells and epithelial cells in the intestine [39,40]. Activated macrophages are a major contributor to the production of cytokines, including IL-1β and TNF-α. IL-1β is a proinflammatory mediator that plays a central role in intestinal inflammation in the gut [21]. In this context, we tested whether a non-toxic concentration (1 × 108 CFU/mL) of RR-LM1071, determined by the WST assay (data not shown), could regulate the expression of cytokines in IL-1β-induced HT-29 cells. Using real-time PCR analysis, RR-LM1071 was found to highly suppress the mRNA expression of IL-6, TNF-α, and IL-4 in IL-1β-induced HT-29 cells (Fig. 2). Furthermore, RR-LM1071 significantly inhibited the IL-1β-induced phosphorylation of p44/42 MAP kinase (Fig. 3). These results suggest that RR-LM1071 may inhibit IL-1β-induced cytokine expression via the MAP kinase pathway in HT-29 cells.
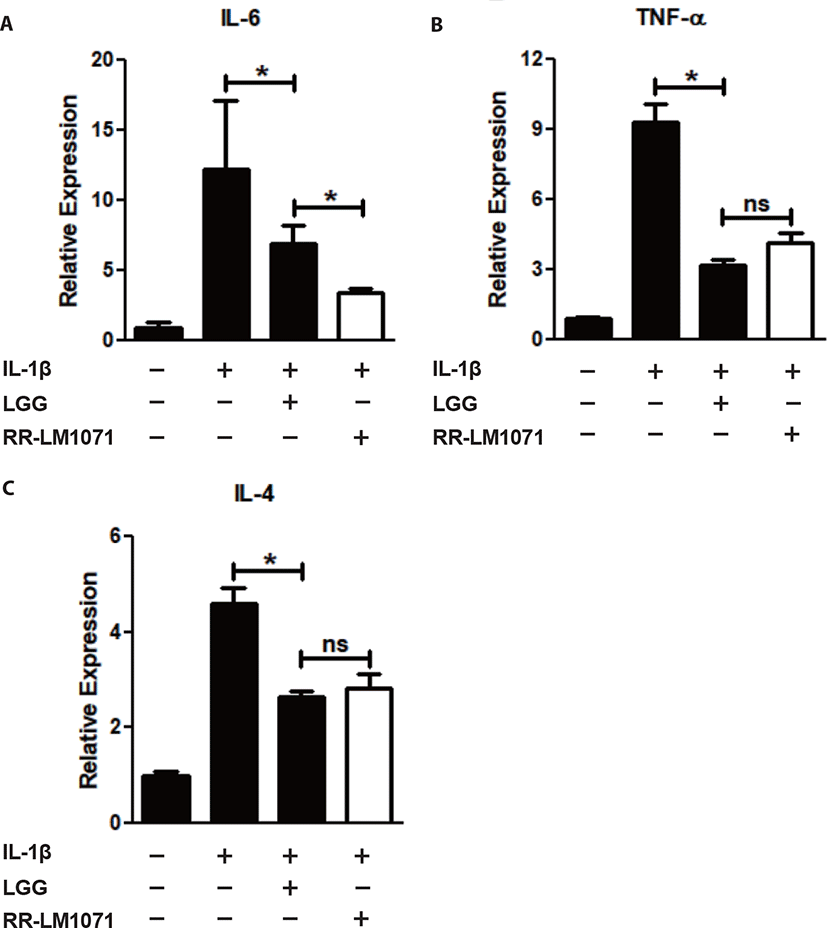
Unexpectedly, the mRNA expression of IL-6 by RR-LM1071 was found to be significantly reduced compared to LGG. However, both TNF-α and IL-4 production did not show any statistical differences compared to LGG. IL-6 is a pleiotropic cytokine that leads to the development of IBD by means of its soluble IL-6 receptor (sIL-6R). IL-6 and sIL-6R levels are known to be correlated with the pathogenesis of UC and DC [41–43]. Therefore, our findings suggest that RR-LM1071 could be a better choice than LGG for preventing IBD.
Although the findings presented in this study point to the involvement of MAP kinase activation, which is responsible for the activation of inflammatory genes, future studies will need to confirm whether RR-LM1071 specifically regulates the pathway in HT-29 cells, as well as evaluate whether other pathways, such as the NF-κB pathway mediated by IL-1β, are involved.
CONCLUSION
In this study, the safety of RR-LM 1071 isolated from human breast milk was demonstrated by evaluating its hemolytic activity, biogenic amine production, and antibiotic resistance. In addition, RR-LM1071 was found to have more affinity for HT-29 cells compared to LGG in adhesive tests. Under inflammatory conditions induced by IL-1β in HT-29 cells, RR-LM1071 suppressed the expression of inflammatory cytokines, namely IL-6, TNF-α, and IL-4, via the MAP kinase pathway. Collectively, our results demonstrate that RR-LM1071 would be an effective and safe probiotic in humans, thus making the case for its use in the development of health supplements and therapeutic treatments for IBD.

