INTRODUCTION
The weaning period is one of the crucial stages in the growth and development of pigs. This is because weanling piglets are suddenly faced with complex and diverse stressors [1,2]. These challenges disrupt the physiological, immunological, and microbiological interactions that maintain gut health, leading to growth retardation, increased morbidity, and even mortality. Furthermore, incidence of post-weaning diarrhea (PWD) caused by Escherichia coli is increased due to changes in the gut ecosystem following the diet transition at weaning, and it remains a challenge on the gut health of pigs during the immediate post-weaning period [3,4]. Thus, targeted nutritional interventions to reduce colonization with pathogenic Escherichia coli through the establishment of beneficial microbes is important for the prevention of PWD. Historically, there has been usage of in-feed antibiotics for prophylactic or therapeutic treatment of the disease [5,6]. However, the overuse of in-feed antibiotics for the purpose of growth promotion was banned in the many countries due to public health concerns about development of antimicrobial resistance that can be transferred to animals and humans [7]. Therefore, there is a need for nutritional strategies that can replace in-feed antibiotics.
Probiotics (PRO) are defined as “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host” [8]. In general, PRO is known to support growth performance of animals via their modulating effects on gut microbiota. Furthermore, PRO can support gut health promotion by ameliorating diarrhea incidence, nutrient digestibility, and gut integrity as well as immune modulation [9,10]. Because the proportion of lactic acid-producing bacteria (LAB) is decreased after weaning, providing LAB in weaner diets may be beneficial for intestinal balance of weaned pigs [11]. Among different PRO, Lactobacillus spp. are decreased in the weaning period and are known to have positive effects on gut health, which play an important role in establishing a potential targeted therapy for weaned pigs with intestinal microbial imbalance [12]. Lactobacillus spp. secrete antimicrobial substances such as bacteriocins to suppress the growth and adhesion to intestinal epithelium of potential pathogenic strains, thereby regulating the intestinal microbial ecosystem [13,14]. In addition, based on this competitive exclusion, Lactobacillus strains stimulate digestive enzymes activity by fermenting available digested nutrients [15,16] and improve the morphological development of the intestinal epithelial cells [17,18]. Moreover, it regulated inflammatory cytokines by interacting with intestinal immune cells through immunomodulatory effects [19,20]. However, information is lacking on whether microbial shifts by supplementation Lactiplantibacillus plantarum (formerly Lactobacillus plantarum) strain, contributes to gut health and immune responses, and whether it can modulate gut microbiota in weanling pigs. Thus, it was hypothesized that dietary PRO addition in weaner diets will enhance intestinal health by modulating gut microbiota and immune responses, thereby promoting the growth performance in post-weaning piglets. Therefore, the objective of this study was to evaluate the effects of dietary L. plantarum CJLP 243/475 on growth performance, nutrient digestibility, immune responses, and intestinal health of weaned pigs.
MATERIALS AND METHODS
Forty newly weaned pigs ([Landrace × Yorkshire] × Duroc; initial body weight [BW] of 6.93 ± 0.27 kg; 28 days of age) obtained from a commercial farm (CJ CheilJedang) were allotted to one of two dietary treatments based on a randomized complete block design (block: initial BW). Pigs were fed either 1) a basal weaner diet based on corn and soybean meal (CON) and 2) CON added with 0.1% dietary PRO (3 × 109 CFU/g of L. plantarum CJLP 243/475, Immprober-S, CJ CheilJedang) for 5 weeks. Each dietary treatment was applied to 20 replicate pens of individually housed pigs (equal ratio of barrows and gilts per pen). Energy and nutrient content of the basal diet were formulated based on the nutrient requirements for weaned pigs (Table 1) as recommended by the National Research Council [21]. Dry matter (DM), gross energy, crude protein (CP), total calcium, and total phosphorous content of the basal diet were analyzed according to the Association of Official Agriculture Chemists (AOAC) [22] procedures at the Institute of Agricultural Science, Chungnam National University. All pigs were provided a mash form of dietary treatments and fresh water ad libitum in the same sized pen (232 × 175 cm; width × length) throughout the study. In addition, pigs were housed in ambient temperature-controlled pens at 25°C to 28°C and the humidity was set at 50% to 65%. The lighting program was maintained at light/dark intervals of 12 h. During the last week of study, chromic oxide (Daejung Chemicals & Metals) was added to the diets, as an indicator for nutrient digestibility analysis at a concentration of 2 g/kg.
1) Provided per kilogram of diet: vitamin A, 12,000 IU; vitamin D3, 2,500 IU; vitamin E, 30 IU; vitamin K3, 3 mg; D-pantothenic acid, 15 mg; nicotinic acid, 40 mg; choline, 400 mg; and vitamin B12, 12 μg; Fe, 90 mg from iron sulfate; Cu, 8.8 mg from copper sulfate; Zn, 100 mg from zinc oxide; Mn, 54 mg from manganese oxide; I, 0.35 mg from potassium iodide; Se, 0.30 mg from sodium selenite.
2) The values were calculated based on NRC [21].
3) The values were analyzed according to the AOAC [22].
The BW of the weaned pigs and their diet residuals after feeding were recorded on day 1, 7, 14, 21, and 35 and used to calculate the average daily gain (ADG), average daily feed intake (ADFI), and gain to feed ratio (G:F; feed efficiency). The fecal score of each pig was visually monitored for the first 2 weeks after weaning by two independent evaluators. The fecal score ranged from 1 to 5 (1 = hard and dry feces, 2 = soft feces, 3 = moist feces, 4 = mild diarrhea, and 5 = watery severe diarrhea). The diarrhea frequency was presented as a percentage by calculating the number of pen days with a pen average diarrhea score of 3 or higher [23]. Blood samples (5 mL each) were collected from the jugular vein of pigs in each pen using vacuum tubes (BD Vacutainer Systems) with (on day 1, 7, 21, and 35) and without (on day 1, 7, and 21) ethylenediaminetetraacetic acid (EDTA). Serum samples were obtained from non-EDTA tubes by using centrifugation (1580R, LaboGene) at 3,000×g for 15 min at 4°C and stored at –20°C for further immune responses analysis. Fecal samples for fecal microbial analysis were collected from six randomly pigs per dietary treatments on day 1, 7, and 21 via rectal stimulation using a sterile swab and stored at –80°C until metagenomic analysis [24]. For apparent total tract digestibility (ATTD) analysis, fecal samples were collected by daily rectal palpation during 3 days of final week of the experiment after 4 days of adaptation to chromium oxide-containing diets and kept at –80°C until analysis [25]. On the last day, six randomly selected pigs per dietary treatment were anesthetized by 2 mL suxamethonium chloride (Succicholine Inj., BKPharm) via intramuscular injection. After anesthesia, the pigs were euthanized by exposure to carbon dioxide. Jejunal digesta samples for microbial analysis were collected at the middle part of jejunum from the pigs and stored at –80°C until metagenomic analysis. Ileal digesta samples for apparent ileal digestibility (AID) analysis were collected between the distal ileum and ileocecal junction and stored at –80°C until chemical analysis. To analyze intestinal morphology, segments approximately 3 cm in length were collected from the duodenum, jejunum, and ileum and washed with distilled water, placed in a 50 mL conical tube and fixed with 10% neutral buffered formalin solution (BBC Biochemical) until microscopy analysis. Other ileal segments were scraped for gene expression analysis, stabilized in a 1.5 mL microtube with RNA later reagent (QIAGEN GmbH) for 24 h at room temperature, and stored at –80°C until analysis.
Diet and frozen fecal samples were forced-air dried in an oven (FC-PO-91, LabHouse) at 65°C for 72 h. Frozen ileal digesta samples were freeze-dried at a chamber pressure of 5 mTorr at room temperature for 48 h using a freeze dryer (Bondiro, Ilshin). After drying, all samples were finely ground using a coffee grinder (Electric Coffee Grinder, Hamilton Beach) for chemical analysis. The ground samples were analyzed for DM, energy by a bomb calorimeter (C2000, IKA Works), and CP using Kjeldahl method (VAPOXX, Gerhardt) based on the AOAC [22]. Chromium concentrations of all grinded samples were estimated by absorption spectrometry (Hitachi Z-5000 Absorption Spectrophotometer, Hitachi High-Technologies). The AID and ATTD of DM, energy, and CP were summarized for each dietary treatment according to previous reports [26].
For microscopy, the fixed duodenum, jejunum, and ileum tissues were implanted in paraffin, cut into thin sections, stained with hematoxylin and eosin (H&E), and sealed on slide glass. Fifteen villi and their associated crypts were selected from the H&E slides by a fluorescence microscope (TE2000, Nikon) and NIS-Elements software (v. 3.00; NIS Elements, Nikon) to measure villus height, width, area, crypt depth, villus height to crypt depth ratio (VH:CD), and number of goblet cells.
Blood profiles of weaned pigs were measured from whole blood samples in EDTA tubes using an automated hematology analyzer calibrated by porcine blood (scil Vet abc hematology analyzer, Scil animal care company) for total white blood cell (WBC) count, red blood cell (RBC) count, and hematocrit (HCT) level. The immune response of the pigs was measured from serum samples using enzyme-linked immunosorbent assay kits (R&D Systems) according to the manufacturer’s protocol. Serum concentrations of cortisol, tumor necrosis factor-alpha (TNF-α), transforming growth factor-beta1 (TGF-β1), interleukin-1beta (IL-1β), and interleukin-6 (IL-6) were estimated using a microplate reader at 450 nm (Epoch microplate spectrophotometer, Bio Tek instruments).
Expression of tight junction (TJ)-related protein and inflammatory cytokine genes (claudin-1 [CLDN1], claudin-2 [CLDN2], claudin-3 [CLDN3], claudin-4 [CLDN4], occludin [OCLN], mucin-1 [MUC1], interferon-gamma [INFG], TNFA, IL1B, IL6, and monocyte chemoattractant protein-1 [MCP1]) from the ileal mucosa of the pigs was measured by quantitative real-time polymerase chain reaction (qRT-PCR). Total RNA was extracted from ileal mucosa samples using a commercial kit (HiGene Total RNA Prep kit, BIOFACT) and the concentration measured with a spectrophotometer (NanoDrop ND-1000; NanoDrop Technologies). cDNA was synthesized using a QuantiTect Reverse Transcription kit (Qiagen). The qRT-PCR analysis was performed with a StepOnePlus RT-PCR system (Applied Biosystems) using SFCgreen I (BIOFACT), and gene-specific primers (Table 2). The 18S rRNA was used as an internal control for normalization of target gene cycle threshold (Ct) value. The relative quantification of gene expression was determined by the 2–ΔΔCt method [27].
Total genomic DNA was extracted from jejunal digesta and fecal samples using DNeasy PowerSoil kit (QIAGEN) following their protocol. The V3-V4 regions of the 16S rRNA gene were amplified with primer set by Bakt 341F and Bakt 805R [28]. The 16S rRNA gene amplicons were sequenced using an Illumina MiSeq platform (Illumina) at a biotechnology company (Macrogen). Paired-end reads were merged using FLASH v. 1.2.11 [29]. Low quality reads, ambiguous reads, and chimeric reads, were identified and removed using CD-HIT-OUT v. 4.5.4 [30]. Sequences were clustered into operational taxonomic units (OTUs) at 3% different distance cut-offs [31]. Taxonomic assignment of each OTU was performed using BLASTN v. 2.9.0 with reference to the NCBI 16S microbial database. Taxonomic composition was generated using QIIME-UCLUST. Comparison of various microbial communities was performed using QIIME (v. 1.9) based on the OTUs abundance and taxonomy information. Microbial data were normalized by data scaling using the total sum scaling before statistical comparison. Alpha diversity including observed OTUs, Chao1, Shannon, and Simpson indices were measured for within-sample richness and evenness. Beta diversity was measured between dietary treatments based on principal coordinate analysis (PCoA) plots using Bray–Curtis dissimilarity.
Data were analyzed using the GLM procedure of SAS (v. 9.4, SAS Institute), except diarrhea frequency, ileal gene expression, and gut microbiota, in a randomized complete block design (block: initial BW). The experimental unit was the pig. Diet was a main effect and initial BW was a covariate. Frequency of diarrhea was analyzed using the Proc Freq in SAS. The t-test was used for a comparison of ileal gene expression between dietary treatments. The MicrobiomeAnalyst (https://www.microbiomeanalyst.ca/) webtool was used for analysis of the gut microbial diversities statistics (alpha diversity, Kruskal-Wallis test; beta diversity, PERMANOVA) of gut microbiota. Linear discriminant analysis (LDA) Effect Size (LEfSe) analysis [32] was used to identified taxonomic biomarkers with effect size at the 2.0 LDA score threshold using Galaxy online platform (https://huttenhower.sph.harvard.edu/galaxy/). Results were presented as means ± SEM, excluding the microbial alpha diversity, which was presented as means ± SD. Statistically differences were considered significant and tendency between dietary treatments at p < 0.05 and 0.05 ≤ p < 0.10, respectively.
RESULTS
During the overall period of the study, the pigs fed PRO had increased (p < 0.05) ADG and G:F compared with those fed CON (Table 3). Dietary PRO increased (p < 0.05) ADG and ADFI from day 8 to 14 compared with CON. In addition, the PRO group tended to have a higher ADG (p = 0.057) and G:F (p < 0.05) from day 22 to 35 than CON group. There were no differences on diarrhea frequency between dietary treatments for the first 2 weeks after weaning.
Dietary PRO increased (p < 0.05) AID of CP compared with CON (Table 4), but there were no differences on ATTD of DM, CP, and energy between dietary treatments. The result of intestinal morphology was shown in Table 5. In the duodenum, the pigs fed PRO had lower (p < 0.05) crypt depth and tended to have higher (p = 0.074) VH:CD than those fed CON. In addition, the PRO group tended to have an increase (p = 0.064) villus area in the jejunum compared with the CON group. In the ileum, the PRO diet increased (p < 0.05) the number of goblet cell counts compared with the CON diet, but there was no difference on the number of goblet cell counts in the duodenum and jejunum.
Pigs fed PRO had lower (p < 0.05) number of WBC counts on day 21 than those fed CON, but tended to have higher (p = 0.059) number of RBC on day 35 (Table 6). However, there was no difference on HCT level between CON and PRO. As shown in Table 7, the pigs fed PRO had lower serum concentrations of cortisol on day 7 (p < 0.05) and TNF-α on day 21 (p = 0.097) than those fed CON, but had higher (p < 0.05) serum concentration of IL-6 on day 21. There were no differences on serum concentrations of TGF-β1 and IL-1β.
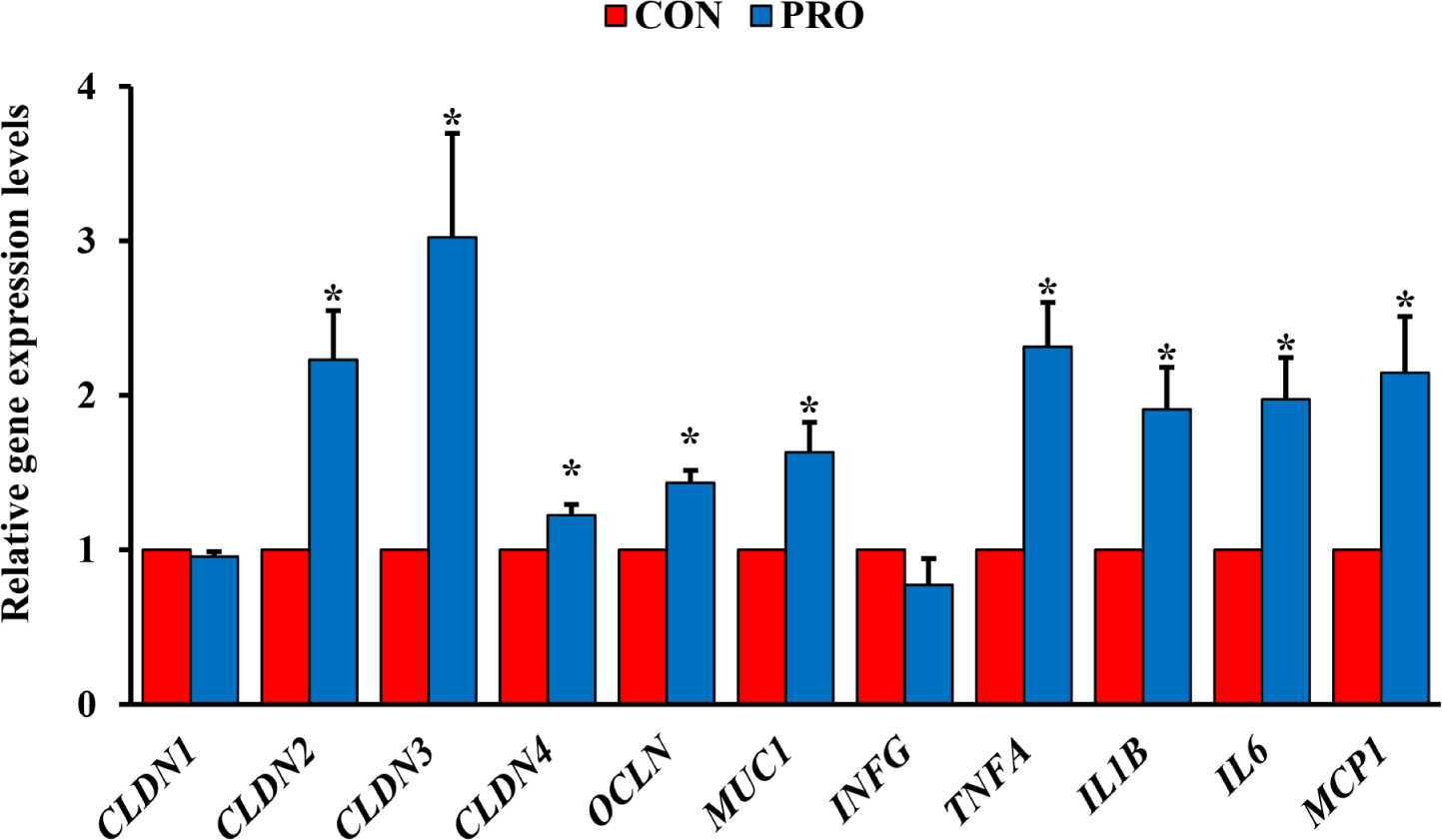
The PRO had upregulated (p < 0.05) relative expression of TJ-related protein genes in the ileum such as CLDN1, CLDN2, CLDN3, CLDN4, OCLN, and MUC1 compared with the CON (Fig. 1). Furthermore, the PRO had upregulated (p < 0.05) relative expression of inflammatory cytokine genes such as TNFA, IL1B, IL6, and MCP1 compared with the CON.
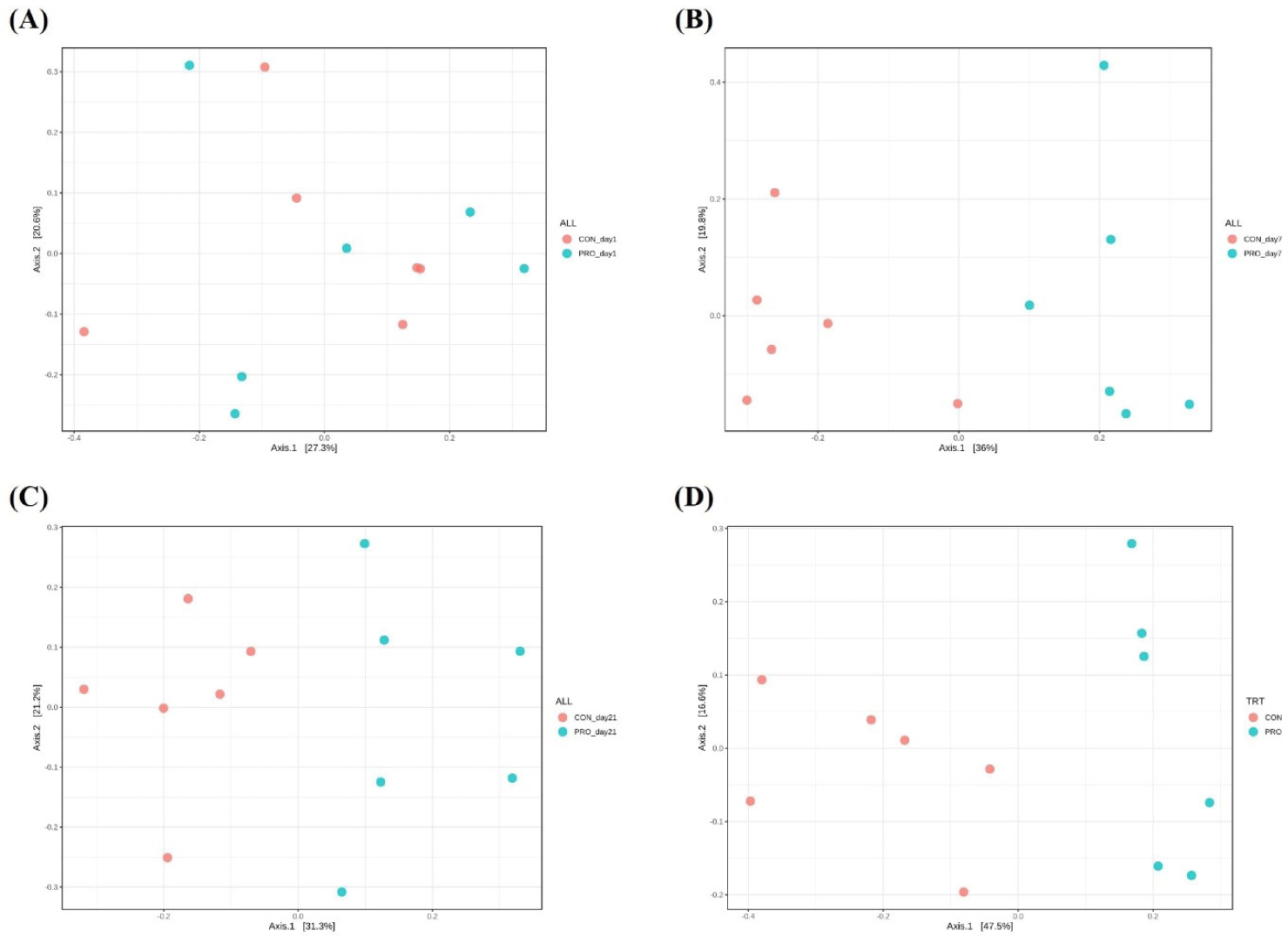
Alpha diversity indices are presented in Table 8. In fecal microbiota, there were no differences on alpha diversity indices between dietary treatments. However, in the jejunal microbiota, dietary PRO increased (p < 0.05) Shannon and Simpson indices on day 35 compared with CON. Beta diversity was presented in the PCoA plot based on Bray–Curtis distance and shown in Fig. 2. There were no differences (Fig. 2A) on Bray–Curtis distance in fecal microbial communities on day 1 between dietary treatments. However, differences on clustering of fecal samples were determined on d 7 (r2 = 0.32, p < 0.05; Fig. 2B) and day 21 (r2 = 0.26, p < 0.05; Fig. 2C) between CON and PRO. In addition, clustering of Bray–Curtis distance in jejunal microbial communities was different on day 35 between dietary treatments (r2 = 0.41, p < 0.05; Fig. 2D).
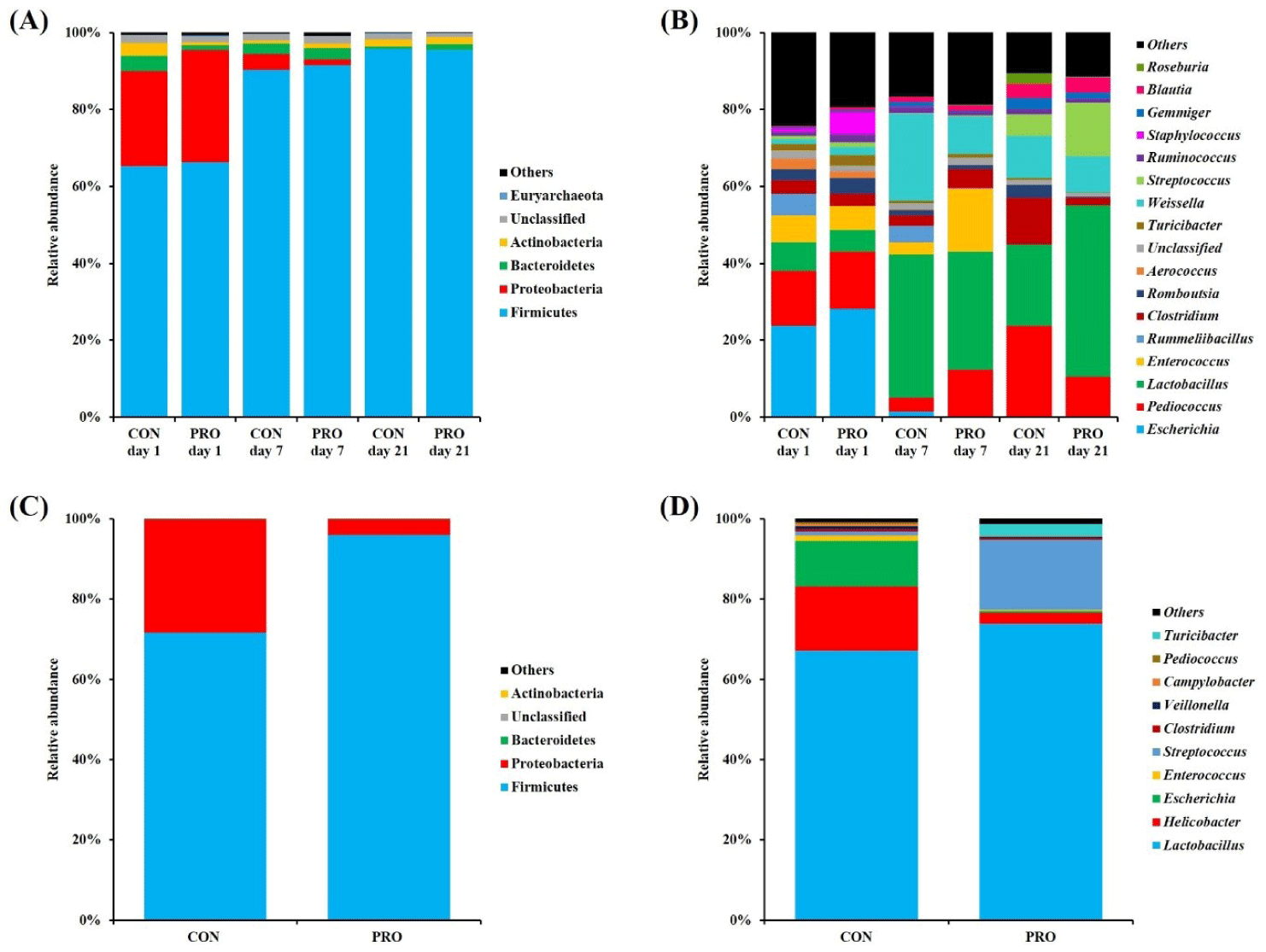
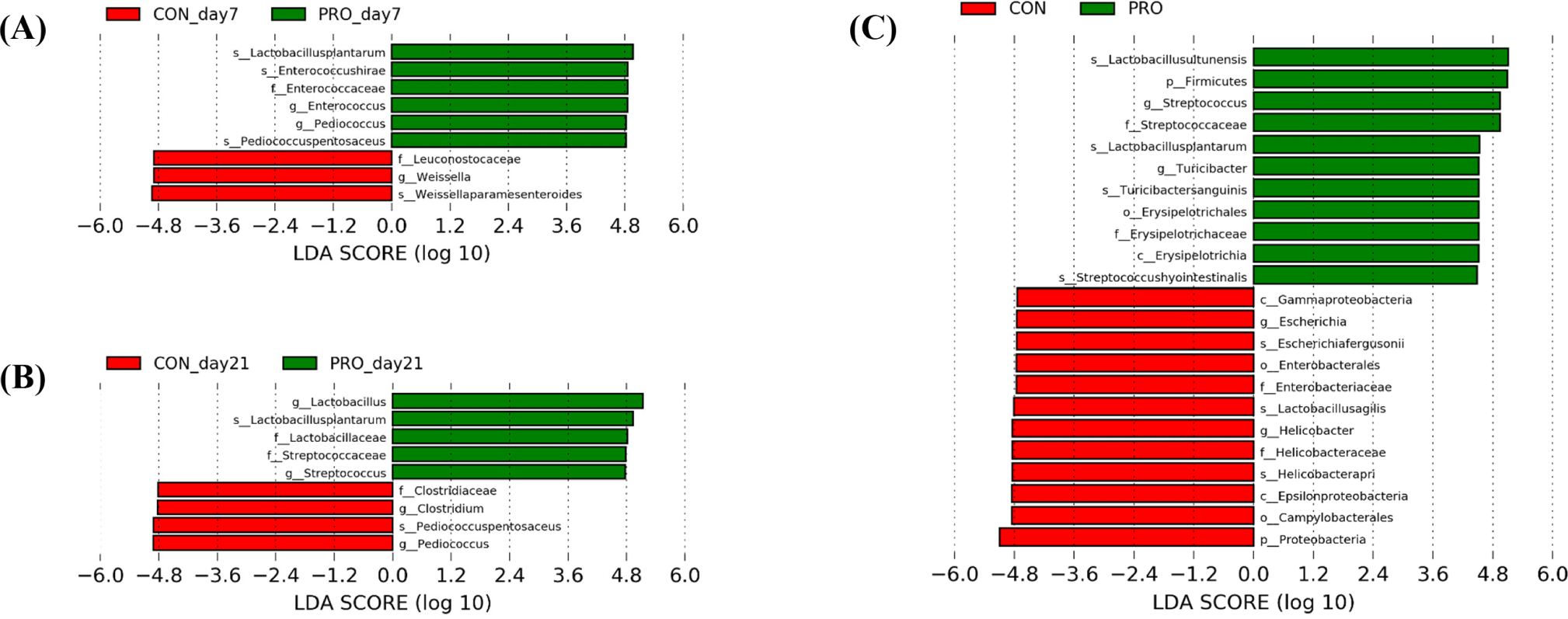
The relative abundance and differences of the gut microbiota between dietary treatments are shown in Figs. 3 and 4, respectively. In fecal microbiota, phylum Firmicutes (CON, 65.23%; PRO, 66.30%) was the most dominant in both dietary treatments at day 1, followed by Proteobacteria (CON, 24.74%; PRO, 29.18%; Fig. 3A). On day 7, Firmicutes was increased in relative proportion in both CON (90.37%) and PRO (91.51%) compared with at the beginning of the study, but Proteobacteria was decreased (CON, 4.04%; PRO, 1.46%). On day 21, Firmicutes was the most dominant phylum in both dietary treatments (CON, 95.73%; PRO, 95.51%), followed by Actinobacteria (CON, 1.79%; PRO, 1.91%). At the genus level (Fig. 3B), Escherichia was the most predominant fecal microbiota in both CON (23.73%) and PRO (28.23%) on day 1. In the CON group, genera Pediococcus (14.39%), Lactobacillus (7.41%), Enterococcus (6.89%), and Clostridium (3.47%) were followed. However, genera Pediococcus (14.82%), Enterococcus (6.30%), Staphylococcus (5.70%), and Lactobacillus (5.61%) were followed in PRO the group. On day 7, dietary PRO increased (p < 0.05) Pediococcus and Enterococcus compared with CON (12.24% vs. 3.59%; 16.47% vs. 3.19%, respectively), but Weissella was lower (p < 0.05; 9.69% vs. 22.53%; Fig. 4A). On day 21, the PRO had higher (p < 0.05) Lactobacillus (44.52% vs. 21.09%) and Streptococcus (13.85% vs. 5.55%) than the CON, but Pediococcus (10.50% vs. 23.81%) and Clostridium (1.89% vs. 12.17%) were lower (p < 0.05; Fig. 4B). In jejunal microbiota, pigs fed PRO had higher (p < 0.05) Firmicutes than those fed CON (95.91% vs. 71.72%) on day 35, but lower (p < 0.05) Proteobacteria (3.93% vs. 28.13%; Fig. 3C and Fig. 4C). At the genus level, dietary PRO increased (p < 0.05) Streptococcus (17.42% vs. 1.08%) and Turicibacter (3.02% vs. 0.11%) compared with CON, but Helicobacter (2.87% vs. 15.92%) and Escherichia (0.43% vs. 11.44%) was decreased (p < 0.05; Fig. 3D and Fig. 4C). Additionally, differences between dietary treatments at the species level identified through LEfSe analysis are shown in Fig. 4. On day 7, species L. plantarum, Enterococcus hirae, and Pediococcus pentosaceus were identified in fecal microbiota of PRO group, while species Weissella paramesenteroides were classified as microbial features in the CON group (Fig. 4A). On day 21, species L. plantarum was identified in fecal microbiota of PRO group, while species Pediococcus pentosaceus were identrified as taxonomic features in the CON group (Fig. 4B). On day 35, species Lactobacillus ultunensis, Lactobacillus plantarum, Streptococcus hyointestinalis and Turicibacter sanguinis were identified in jejunal microbiota of PRO group, while species Helicobacter apri, Lactobacillus agilis, and Escherichia fergusonii were classified as microbial features in the CON group (Fig. 4C).
DISCUSSION
The gastrointestinal (GI) tract plays a crucial role in overall health. In the weaning period, GI health is accompanied by loss of structure, barrier, local immune function and microbial dysbiosis, resulting from changes in the GI environment [1,2]. Overall, reduced digestion and absorption of nutrients leads to PWD and poor feed efficiency, thereby impairing productivity [4]. Our findings showed that dietary PRO inclusion in diet of weaned pigs enhanced growth performance compared with CON through improved intestinal health. Previous studies are consistent with our result of growth performance [19,33–35] and have reported that improvement in intestinal environment would be supported by dietary PRO. In antibiotic-free swine production, PRO have been proposed as a potential alternative for PWD caused by Escherichia coli infection [36]. PWD causes dehydration due to intestinal fluids and electrolytes secretion [36,37]. However, we found no difference on diarrhea frequency with the PRO addition. Additionally, the HCT, as an indicator that increases with diarrhea, did not differ between CON and PRO. The effects of dietary PRO on PWD are inconsistent, which could be associated with PRO dosage and healthy normal or challenge conditions [17,18,38,39]. Therefore, the present growth performance result would be supported by focusing on another potential mechanism of dietary PRO in weaned pigs.
Weaned pigs suffer from accelerated weaning stress on their immature intestine, which impaired physiological structure and function: villus atrophy and crypt hyperplasia as a structural change, and digestive enzyme activity decrease as a functional change [40,41]. Collectively, post-weaning GI disturbances affect long-term growth rate and feed efficiency of pigs. In the present study, we determined that the AID of CP was improved, which may be due to the result of probiotic Lactobacillus strain used in the experiment affecting the activity of digestive enzymes in the GI tract. This is because PRO not only increases the utilization of nutrients by producing nutrient-decomposing enzymes, such as proteolysis, but also stimulates digestive enzymes by fermenting digesta through increase beneficial microbiota [15,33]. In general, the small intestinal morphological indices are useful parameters of surface area for nutrient absorption and intestinal health. After weaning, immature enterocyte differentiation leads to a decrease in villus height and an increase in crypt depth, which inhibits digestive enzyme activity and nutrient absorption [42]. Additionally, these morphological changes in the intestine were associated with the low feed intake after weaning [42]. Our experiment confirmed that dietary PRO enhanced intestinal morphology by decreasing duodenal crypt depth and tending to increase jejunal villus area. In the crypts, intestinal stem cells continuously differentiate for villus development. However, as mentioned earlier, the structural appearance of the immature intestine in post-weaning pigs is characterized by shedding of villi and increased crypt depth. Thus, the improvement of crypt depth has a positive effect on villus development for nutrient absorption. Previous studies in weaned pigs were also consistent with the PRO efficacy on intestinal morphology, with positive effects varying across the small intestine segments [17,34,39]. Our results indicate that addition of dietary PRO may have produced and/or stimulated nutrient-digesting enzymes through the growth and development of beneficial microbiota including Lactobacillus strains in the GI tract. In addition, increased feed intake in the early post-weaning period may have stimulated the intestinal morphological development of pigs.
PRO have been suggested to have beneficial effects on intestinal barrier function, such as enhance TJ integrity and defense against pathogen invasion in intestinal epithelial cell lines [43,44]. Disruption of the TJ barrier, which composed of transmembrane proteins such as the CLDN family and OCLN, and intracellular scaffold proteins such as zonula occludens, causes paracellular permeability in gut [44]. Mucin secreted by goblet cells is a major component of the intestinal mucus layer, which serves as the first line of host defense [45]. In this study, the expression of CLDN family (CLDN2, CLDN3, and CLDN4) and OCLN genes in the ileum were upregulated in the PRO. Moreover, the upregulation of MUC1 gene in dietary PRO was consistent with the improvement in the number of goblet cells in the ileum. It has been reported that PRO prevents pathogen binding in the epithelial layer because it induces qualitative changes in mucin by mucin gene expression [45]. Moreover, dietary PRO can protect intestinal barrier function through maintenance of TJ-related protein genes expression from pathogenic bacteria [46]. Thus, our findings suggest that dietary PRO enhanced intestinal integrity by stimulating mucus secretion in the ileum, thereby inhibiting pathogenic penetration and reducing intestinal permeability. In addition, the PRO addition may have improved the barrier function of the intestinal epithelium by dominating in the intestine and protecting potential opportunistic pathogens and/or microbiota from adhering to the intestinal epithelium through competitive exclusion in the intestinal microecosystem.
Weaning period elevates the systemic cortisol concentration, as a stress indicator, and induces activation of central stress pathway [47,48]. In addition, an increased WBC counts indicate systemic inflammation [49]. Since cytokines maintain local/systemic immune homeostasis by regulating inflammation in immune responses, changes in levels after weaning can support the host health condition through the immune system. In this study, we found that dietary PRO alleviated early-weaning stress of piglets via serum cortisol level. Moreover, concentration of serum TNF-α, a pro-inflammatory cytokine that is activated by macrophages and triggers an inflammatory response, was reduced by PRO addition. Under various conditions, IL-6 is classified as either pro- or anti-inflammatory responses and it was increased in the serum of PRO pigs in this study. The anti-inflammatory/regenerative properties of IL-6 are mediated by classic signaling, but the pro-inflammatory properties of IL-6 are mediated by trans-signaling in chronic inflammatory diseases [50]. In the present study, not only clinical symptoms of chronic inflammation not observed throughout the experiment, but the WBC counts in the PRO group also decreased as the experiment progressed. On this basis, we considered increased serum IL-6 in PRO to have anti-inflammatory properties. Therefore, dietary PRO appears to modulate the systemic immunity of weaned pigs through anti-inflammatory effects of circulating cytokines. In the GI immune system of pigs, weaning transition is associated with inflammation due to upregulated pro-inflammatory cytokines [51]. Pro-inflammatory cytokines are known to play a role in increasing intestinal permeability by disrupting TJ barriers [52]. Interestingly, however, dietary PRO upregulated expression of ileal pro-inflammatory cytokine and chemokine genes (TNFA and IL1B; MCP1, respectively). On the other hand, the anti-inflammatory cytokine IL6 has been suggested to be effective in regenerating intestinal epithelial cells and protecting the intestinal barrier [50,52], and was also upregulated in the ileum. Through our results, dietary PRO activated the mucosal immune system as an immunostimulatory function through cytokines induction, but this did not deteriorate intestinal barrier permeability. Cytokines are mediators that regulate intestinal mucosal barrier function and have diverse effects on intestinal permeability, including nutrients and ion transport [53,54]. Additionally, because cytokine-induced barrier modification can restructure TJ protein expression [55], it is possible that upregulation of inflammatory cytokine affects upregulation of proteins composing TJ barrier. Moreover, intestinal cytokine-induced responses may be associated with feedback regulation of changes in the intestinal environment, such as microbial modifications following PRO addition. However, in order to clearly identify these effects, the relationship between intestinal cytokine profiles and TJ barrier functions needs to be supported by further studies. In addition, unlike systemic immune responses, cytokine stimulation in local immune responses may be the results of PRO-induced changes in the gut microbial environment and antimicrobial substances it can produce and secrete.
The present study showed dietary PRO did not alter fecal alpha diversity, but fecal microbial communities were distinctly separated in beta diversity by dietary PRO supplementation. Moreover, dietary PRO not only changed the jejunal alpha diversity but clustered the beta diversity on day 35 of the study. These results indicate that dietary PRO contributes to changes in alpha diversity with prolonged exposure effects to the gut. Furthermore, our results are consistent with previous report suggesting that alpha diversity was negatively correlated with average daily gain and body fat [56]. These results may be due to the potential mode of action that dietary PRO can inhibit pathogens [57]. However, in understating microbial diversities, it is important to identify whether the distribution of strains that have any effects on host health, such as potentially harmful or beneficial microbiota, has changed, rather than simply increasing or decreasing diversities. Therefore, dietary PRO supplementation may positively affect growth and intestinal health of post-weaning pigs by altering microbial abundance to utilize nutrients or energy source and produce metabolites. Overall, differences on taxonomic abundances of gut microbiota were expected due to altered microbial diversity and dissimilarity by dietary PRO addition.
In fecal taxonomic abundance, Enterococcus, Streptococcus, and Lactobacillus were clearly enriched in the PRO group compared with the CON group. These genera are included in LAB and are known to be beneficial in pigs. However, some species of Enterococcus spp. have been reported to be potential harmful to the host. In particular, Enterococcus hirae is known to cause enteroadherent infection and diarrhea in animals [58,59], and this species was identified in the PRO group through LEfSe analysis. This may be related to the lack of treatment group differences in clinical PWD in the early post-weaning period. However, predominance of the PRO strain may have contributed to the suppression of Enterococcushirae adhesion to the GI tract of weaned pigs due to its pathogenic inhibitory properties by antimicrobial peptides secretion. This is because as dietary PRO intake continued, Lactobacillus (L. plantarum) and Streptococcus, were predominated in the PRO group, whereas Clostridium predominated in the CON group. Genus Clostridium includes pathogenic strains such as Clostridiumperfringens and Clostridiumdifficile, which can cause clostridial diarrhea in pigs [60]. On the other hand, it has been reported that Streptococcus is correlated with the BW gain of pigs [61,62], which is consistent with our experiment. Therefore, our result suggests that dietary PRO altered gut microbiota by predominating in the gut and suppressing potential pathogenic strains, resulting in positive effects on the growth of weaned pigs. In the jejunum, phylum Firmicutes was enriched in the PRO group compared with the non-PRO group, whereas the phylum Proteobacteria was less abundant. In general, Firmicutes play important roles in the breakdown of complex plant carbohydrates and are dominant under healthy conditions [63]. However, Proteobacteria include opportunistic pathogens such as Escherichia, Salmonella, and Helicobacter, and are associated with intestinal inflammation or dysbiosis [64,65]. In pigs fed CON, Helicobacter apri and Escherichia fergusonii were identified by LEfSe analysis, both of which are clinically pathogenic. On the other hand, L. plantarum, Lactobacillus ultunensis, Streptococcus hyointestinalis, and Turicibacter sanguinis were characterized in the PRO group. Streptococcus hyiointestinalis produces bacteriocins and exhibits broad antimicrobial activity against gram-positive bacteria [66,67]. In addition, intestinal Turicibacter sanguinis play important roles in intestinal lipid and steroid metabolism [68,69] and is involved in serotonin production [70]. These findings revealed the lower abundances of Escherichia and Helicobacter and their belonged phylum Proteobacteria in the jejunum of PRO pigs than CON pigs, indicating inhibitory effects of dietary PRO on these opportunistic pathogens through gut enrichment of probiotic L. plantarum. Moreover, dietary PRO may help gut eubiosis by interacting with beneficial microbiota. Therefore, these fecal and jejunal microbial changes suggest that dietary PRO contributed to healthy and balanced alteration in the intestinal environment of weaned pigs, which promoted intestinal integrity and affected nutrient utilization of the host. Further studies evaluating the gut metabolites and functional profiles of probiotic L. plantarum would be helpful to determine the potential functionality of dietary PRO in pigs.
CONCLUSION
This study demonstrated that probiotic L. plantarum CJLP 243/475 supplementation in weaner diet enhanced the growth performance of weaned pigs. The potential mechanisms of beneficial effect involve improving intestinal morphology and intestinal integrity by upregulating expression of TJ protein genes, and modifying host-microbiota interaction by altering gut microbial communities. In addition, probiotic L. plantarum CJLP 243/475 alleviated systemic inflammation in early weaning caused by weaning stressors and modulated local immune responses by upregulating gene expression of inflammatory cytokines in the ileum. These results support our hypothesis that probiotic L. plantarum CJLP 243/475 could enhance intestinal health by modulating gut microbiota, thereby improving growth performance and modulating immunity system of weaned pigs. Further studies will consider evaluating the correlation between gut microbial changes and gut-immune health indicators to establish a potential mechanism of L. plantarum CJLP 243/475 on the health of pigs.

