INTRODUCTION
Productivity and feed efficiency are the basic prerequisites for profitable swine production. In past decades, it was common for sows to receive a single diet through both pregnancy and lactation and this approach remains predominant in some regions where protein sources are readily available and affordable. However, today, sows are typically provided with a dual-diet regimen. During gestation, they were typically fed a diet lower in energy and protein to prevent excessive weight gain and maintain reproductive health. But when they enter the transition period (i.e., from gestation to lactation) they are subjected to have high-energy and protein diet to support the increased nutritional demands associated with the rapid development of the fetus and to optimize their performance during farrowing [1]. Earlier studies [2,3] demonstrate that a high-fiber diet provided to sows up to the point of farrowing has minimized the constipation and enhanced the farrowing outcomes. Energy requirements of sows during gestation and lactation are still difficult to assess because of the confounding effects of reproductive cycle. Particularly, during lactation period sows were offered with less fiber and higher levels of metabolic energy, and protein diet [4]. Energy intake during pregnancy influences voluntary expenditure during lactation at the same time energy intake during lactation influences to maximize subsequent reproductive performance [5]. This increased reproduction performance intensifies the demands of sows, requiring them not only to successfully farrow but also to supply the essential nutrients to support piglet growth and survival. As a result, swine producers and researchers were driven to identify an optimal nutrient composition for gestation and lactation to improve feed intake, reproductive success, and healthy offspring.
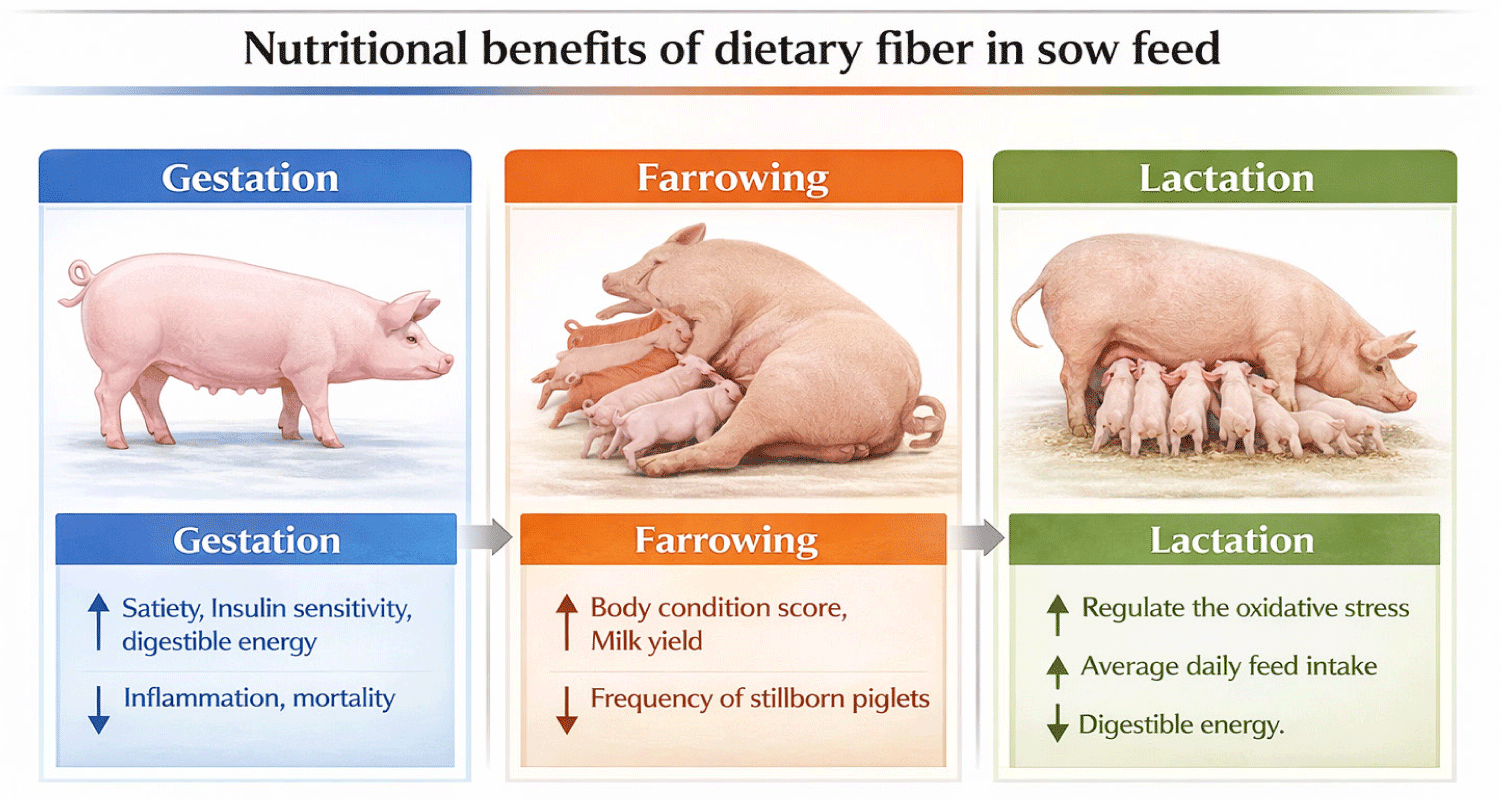
Dietary fiber (DF) often called as the “seventh largest nutrient,” has been identified as an essential and sustainable nutritional source [6]. Emerging data proved that dietary fiber during gestation plays a crucial role in optimizing sow performance and producing healthier piglets [7]. Incorporating appropriate amounts of fiber into the diet of gestation sows can help to maintain proper body condition and promote feed intake during lactation [1]. This, in turn, can help to reduce farrowing complications related to overweight, shorten farrowing duration, and improve lactation performance [8]. In recent years, a diverse range of fibrous feedstuffs were incorporated in pig diets such as distillers dried grains with soluble (DDGS), soybean hulls, wheat bran, sunflower meal, and sugar beet pulp [9]. As global demand for livestock feed rises, novel co-products like copra meal, palm kernel, rice bran, and canola meal are gaining prominence. Yet it is crucial to recognize that these fiber sources exhibit substantial variations in their nutritional composition, particularly in terms of fatty acid profiles. Also, the practical guidelines for incorporating these fiber feedstuffs into sow diets remain undefined and need specific recommendations. Thus, this review aims to explore the current information of the use of fiber-rich ingredients in contemporary sow diets and emphasizing its role in enhancing productivity by improving nutrient utilization and overall health (Fig. 1).
DIETARY FIBER: A GENERAL DESCRIPTION
Fiber, a carbohydrate and an anti-nutritional component is usually hard to digest by the endogenous enzymes and passes through the digestive system [10] of the host. Instead, they were fermented by micro-organisms within the colon into short chain fatty acids. DF is the primary component of fiber-rich feeds, constituting more than 40% of the total dry matter (DM) [11]. These fiber-rich feeds are cost-effective and plant-based which include barley, corn, wheat bran, soybean hulls, canola meal, and DDGS [12]. The main component of DF includes pectin, lignin, hemicellulose, cellulose, fructan, oligosaccharides, and resistant starch [13]. Previously, Sapkota et al. [14] demonstrated that DF has minimized the behavioral issues and enhanced the well-being of gestation sow by reducing their stress level. However, Portal et al. [15] reported that sows fed high fiber gestation diet had no better reproductive performance. While Rosero [16] found reduced wean-to-estrus interval in sow during the peripartum period when DF was top-dressed. Also, Li et al., [17] reported that DF had reduced nutrient digestibility and inhibited energy deposition in pig diets. Though DF inclusion offers benefits beyond lower feed costs ongoing research in animal nutrition has begun to emphasize its positive effects by showcasing its potential benefits for enhancing pigs’ overall health and performance.
FIBER: CLASSIFICATION, TYPES, AND MODE OF ACTION
Fibers can be classified based on several key characteristics such as structure, solubility, fermentability, water-holding capacity, digestible and indigestible fractions, and viscosity. Soluble dietary fiber (SDF), such as pectins, hemicelluloses, arabinoxylans, xyloglucans, and fructooligosaccharides, are rapidly fermentable [18] while insoluble dietary fiber (ISDF), like cellulose, lignin, and different forms of resistant starches are slowly fermentable [19]. The functional properties of these fibers are largely defined by their solubility and fermentability. Solubility refers to the ability of fiber to dissolve in water. The degree of fiber solubility may stimulate the viscosity of the ileum and slow down the digestive process which results in reducing transit time and limiting the availability of enzymes for nutrient absorption [20]. Excess solubility is therefore undesirable as it can impair overall nutrient digestibility. Whereas insoluble fibers absorb water rather than dissolve. This water absorption helps maintain proper hydration of the digesta as it moves through the digestive system, and some of this water is released in the large intestine, where it may re-enter the body’s circulatory system. However, we should not exclude all soluble fibers because many are fermentable, playing a vital role in gut health. Concurrently, ISDF can promote gut motility when consumed at the right concentration. This complex process involves both inert and fermentable fibers, with the latter providing nourishment for gut microbiota, which in turn produce neuro-mediators and regulate gut function. Most bacteria degrading DF are beneficial and can ferment DF into organic acid, thereby lowering the pH of the intestinal lumen, and inhibiting the proliferation of pathogenic bacteria [18]. Previously, Wu et al. [21] reported that xylan promoted the proliferation of Bifidobacterium in weaning pigs. The fibrous components extracted from feeds or feedstuffs are generally classified into three main types: total dietary fiber (TDF), crude fiber (CF), and neutral detergent fiber (NDF), and these fibers were often measured by different approaches: a) enzymatic-gravimetric method for TDF; b) chemical-gravimetric method for CF; and c) Van Soest method for NDF [22]. The detection methods for CF and NDF often exclude SDF components, while TDF analysis encompasses all types of fiber including SDF and IDF. DF enhances the beneficial bacteria in the lower gastrointestinal (GI) tract by fermenting carbohydrates and produces short-chain fatty acids (SCFAs), particularly butyrate which supports GI and overall productivity in sows. In the earlier study, Shang et al. [23] demonstrated that TDF increases beneficial bacteria in the hindgut, such as the genus Christensenellaceae, which is associated with improved gut health and SCFA production. Normally fiber cannot be digested by the animals, but it could provide feelings of fullness after feeding without spiking blood sugar or providing extra calories [24]. Also, it mitigates the symptoms of constipation by alleviating stress. Even if the addition of dietary fiber plays an important role in alleviating sow hunger and reducing abnormal behavior, it may be affected by the type of fiber and the inclusion level [25] thus incorporating DF into sow diets necessitates a clear understanding.
ROLE AND IMPACT OF FIBER IN SOW FEED
Gestation sows are often subjected to a strict feeding and this controlled feeding regime might often lead to constipation and stereotypical behaviors such as sham chewing (chewing motion unrelated to eating), bar biting, and nosing or licking the floor in the absence of feed [26]. A previous study insists that feeding 5% resistant starch during the gestation period has enhanced postprandial satiety by alleviating stress and reducing abnormal behaviors [27]. Additionally, Ferguson et al. [28] reported that providing gilts with a high-fiber diet enhances follicle quality, oocyte maturation, and early embryo survival which seems to be linked with changes in estradiol (E2) and luteinizing hormone (LH) profiles. In 2023, Qin et al. [13] reported that adding 10.8%, 15.8% and 20.8% NDF to sows on different parity from day 1 through to day 90 of gestation has improved their reproductive performance. Similarly, Noblet and Le Goff G. [29] demonstrate that incorporating increasing levels of DF (3.3, 8.6, and 10.1 kJ for each gram of NDF, ADF, and CF, respectively) had increased digestible energy in adult sows. These effects were primarily attributed to the enhanced degradation of dietary fiber in the hindgut of the sows. A study by Lu et al. [30] proved that sows fed higher levels of TDF had lower constipation, which in turn led to reduced farrowing time. On the other hand, Tan et al. [31] demonstrate that inclusion of dietary fiber in late gestation has reduced oxidative stress on day 1 of lactation. Also, Zhou et al. [32], addressed that adding high fiber during gestation helps sows to maintain a suitable body condition scoring. Moreover, Lowell et al. [33] noted a high metabolic energy in gestation sow fed wheat middlings and soybean hulls. Additionally, Stein and Shurson. [34] described that replacement of soybean with DDGS had not adverse impact on gestation sow performance. Meanwhile, Kim et al. [35] reported that gestating sows fed a diet containing soybean hulls exhibited greater apparent total tract digestibility of DM and energy compared to those fed a diet containing pistachio shell powder (PSP). Several studies have examined the impact of varying fiber content in the diets of pregnant sows and the growth rates of piglets. For example, Oliviero et al. [36] reported that sows fed 7% versus 3.8% crude fiber had increased the piglet weight gain from 1 to 5 d of age, while Loisel et al. [37] stated that feeding a high fiber diet from d 106 of pregnancy until parturition did not influence piglet weight gain until d 21 of lactation. Sows often experience metabolic syndrome during late gestation and early lactation due to vigorous metabolism and diminishing antioxidant capacity, with intestinal microbiota imbalance playing a significant role in this condition [38]. Promoting beneficial bacteria in the intestine may help improve metabolic syndrome through “microbiota remodeling,” thereby alleviating inflammation and oxidative stress, which effectively increases the average daily feed intake (ADFI) during lactation [39]. Research by Li et al. [12] demonstrated that maternal intake of SDF enhances intestinal health in neonates. Increasing the ratio of soluble to insoluble dietary fiber in the diets of pregnant sows improved antioxidant capacity and reduced inflammation in the colon of piglets. Briefly, maternal SDF intake benefits the intestinal health of piglets however, precise mechanisms related to various factors, including the vertical transmission of intestinal microbiota, and fetal intestinal development during pregnancy require further clarification. Overall, DF has been shown to improve the reproductive performance of sows at different stages, but the selection of fiber sources and optimal inclusion levels warrant further exploration.
The primary challenge faced by sows during parturition is the rapid depletion of energy, particularly when dealing with larger litter and prolonged farrowing times [40]. A swift farrowing process is crucial for the survival of piglets; however, prolonged farrowing can result in a higher number of piglet deaths at birth, lower survival rates, increased postpartum oxidative stress, and a greater incidence of anorexia in sows. Ultimately, these factors contribute to reducing the performance of the litter [41]. These challenges can be exacerbated by several factors: constipation, oxidative stress, and insulin resistance, which may result in inadequate physical endurance during farrowing [17,41,42]. Therefore, ensuring proper nutrition for sows during this critical period is paramount and has become a key focus for swine researchers. Earlier studies indicate that when the time between the last meal consumed and farrowing extends beyond approximately four hours, the length of farrowing increases, potentially leading to energy depletion [17]. To prevent sows from being depleted during farrowing, it is recommended to feed them three meals a day. Accordingly, adequate energy supply to sows is vital to reduce the farrowing length and stillborn piglets. The addition of fiber into sow diet during the last two weeks before parturition has been shown to decrease the frequency of stillborn piglets [43]. Feyera et al. [3] reported that sows fed high-fiber diet during the perinatal period alleviates prolonged farrowing duration by softening feces and providing energy from their hindgut. We proposed that above mentioned research outcomes might be due to fiber fermented in the hindgut which ensures to have constant energy uptake from the gastrointestinal tract and thus contributes to stable blood glucose level even though the sows are not able to eat while giving birth. So far, several studies have demonstrated the effects of DF addition on sow reproductive performance. However, only a few have explored the mechanisms by which DF supplementation enhances their fertility. Placenta plays a crucial role in material exchange between the fetus and the mother, and placental insufficiency is a common cause of low litter size, birth weight, and uniformity of piglets within a litter. A recent study indicated that dietary DF during gestation has resulted in three successive parities, high litter size and weight, as well as increased placental weight in the 2nd and 3rd parities [44]. This suggests that DF during gestation may enhance sow reproductive performance by improving placental development and function.
Lactation is a critical phase in the reproductive cycle of sows [1], and their feed intake during this period is more crucial for their subsequent performance [45] as well as the growth and development of their suckling’s [46]. Previously, Shang et al. [23] investigated the effect of sugar beet pulp (SBP) and wheat bran (WB) in sows and found that maternal feeding of SBP improved the weaning weight of piglets, whereas WB had no significant impact on their growth rate. Additionally, Kim et al. [35] found low energy digestibility in lactation sow by the inclusion of PSP (high in IDF). Based on these findings, we hypothesize that soluble fiber sources like SBP may be more effective than insoluble fiber sources like WB and PSP in supporting lactation performance. During early lactation, maternal nutrition plays an important role in regulating the immune development of their offspring as they largely rely on the colostrum (first source of nutrients and energy) supplied from their dam. Prolactin, an essential hormone, is responsible for initiating and maintaining milk production [47]. Also, it plays a vital role in cell proliferation, mammary gland development, and milk secretion which ultimately helps to deliver nutrients to suckling and improve their survival rates [7]. Such prolactin hormone showed tendency to increase when gestation sows were offered with high fiber diet [48], however some studies showed no difference in the prolactin concentrations with high fiber diet. We hypothesize that these inconsistencies may be attributed to maternal obesity [49] and offering a high-fiber diet to gestating sows may help reduce obesity, thereby boosting prolactin levels in the serum to produce sufficient milk during lactation. Oxytocin is a neurohypophysial hormone that plays a key role in regulating parturition and lactation, including the initiation of uterine contractions and milk secretion [50]. Li et al. [51] reported that sows fed a diet supplemented with 2.26% inulin and 18.2% cellulose during gestation showed increased plasma oxytocin levels. This increase was linked to postprandial satiety resulting from the consumption of a high-dietary fiber diet [52]. Though the concept of fetal programming through maternal nutrition is well-established yet specific mechanisms by which maternal DF intake benefits the intestinal health of piglets remain unexplored, highlighting the need for further research. In recent times, functional fiber such as chitosan oligosaccharides (COS), konjac glucomannan, and inulin have been shown to effectively manage oxidative stress in lactating sows [41,42]. COS, a depolymerized form of chitosan, exhibits excellent water solubility and a range of biological activities, including immune stimulation, anti-inflammatory, and antioxidant properties [53]. These characteristics suggest that COS might play a beneficial role in sows, particularly in neutralizing reactive oxygen species (ROS) and mitigate oxidative damage within their bodies [21]. Also, by scavenging free radicals, COS supports sows to enhance their immune system and protects against cellular damage during pregnancy and lactation. Konjac glucomannan is a natural neutral polysaccharide extracted from konjac tubers [54]. Over the decades, it has been used as a food additive and dietary supplement [55]. The inclusion of 0.25% konjac glucomannan to late gestation sows reveal better lactation feed intake [56] while some studies showed no improvements [57] the inconsistent results may be attributed to factors like leptin hormone secretion, which can suppress appetite. Further research is needed to fully understand the mechanisms and optimize the use of these functional fibers in enhancing the performance of sows during lactation.
CONCLUSION
Effective fiber management in sow diets promotes satiety, supports gut health, stabilizes energy levels, and enhances reproductive outcomes. Though dietary fiber contributes to the well-being of both the sow and piglets, determining the ideal ratio of soluble to insoluble fiber is still challenging. Therefore, further investigation is necessary to understand how processing methods and the timing of fiber supplementation can enhance its nutritional value, enabling the development of effective feeding standards to optimize sow performance.

