INTRODUCTION
The gut is essential for nutrient absorption, and the development of the intestinal system can enhance nutrient absorption, growth performance, and animal health [1]. The poultry’s digestive tract contains many microorganisms, commonly called microbiota. The gut microbiome regulates gut health. A balanced gut microbial population enhances feed digestibility and efficiency, thereby enhancing growth and feed conversion [2]. Various feed additives in poultry diets can affect the gut microbiome, with a few specifically used to modulate the gut microbiome [3].
For decades, antibiotics have been used in the poultry industry to improve production, growth, and health, thereby increasing economic benefits [4,5]. Although antibiotics are crucial for combating bacterial infections, they can also have unintended consequences. For example, they can result in increased antibiotic resistance, food and egg contamination, and environmental pollution. Therefore, in 2006, the European Union banned non-therapeutic antibiotics for growth and production. Korea adopted a similar approach in 2011 [6]. Consequently, there is a need to develop alternatives to antibiotics, such as probiotics, to ensure the continued efficacy of antimicrobial agents.
Probiotics are defined as living microorganisms that, when administrated in adequate amounts, confer a health benefit on the host and are widely used as feed additives in the poultry industry to improve health and welfare [7]. They offer numerous benefits, including stimulation of host intestinal microorganisms and immune modulation [8]. Recently, probiotics have gained popularity in the poultry industry as substitutes for antibiotics in nutritional supplements and feed additives [9]. These universal feed additives can be combined with other additives to improve the performance and health of poultry [10]. Their beneficial effects are observed directly in the gastrointestinal tract and indirectly through poultry immunomodulation [11]. Probiotic-fed flocks exhibit enhanced laying performance and egg quality, increased daily gain, and improved feed conversion ratio (FCR) [12,13]. Additionally, probiotics accelerate the maturation of gut microbiota in broiler chickens [14,15]. Therefore, probiotics have the potential to enhance the productivity and overall health of the poultry industry.
Bifidobacteria, lactobacilli, and Saccharomyces boulardii are the most commonly used microorganisms in the production of probiotics. Probiotics are more effective in controlling microorganisms and less harmful to the environment compared to antibiotics. In poultry, probiotics can enhance growth performance and health by improving feed intake and efficiency, maintaining gut integrity, and promoting gut health [11]. Among these, Bacillus-based probiotics are particularly effective in enhancing health [16]. Bacillus spp. are used to improve production efficiency, boost the immune system, alter the intestinal environment, address metabolic and inflammatory problems, improve cholesterol profiles, and prevent or treat autoimmune diseases in broiler chickens [17]. These strains support chickens’ growth, digestion, and overall health, promoting a healthy gut [18]. Among them, B. subtilis has attracted significant attention in probiotic supplementation research due to its high resistance and survival in the hostile environment of the gastrointestinal tract [19]. Additionally, Bacillus velezensis is known to facilitate plant growth by inhibiting plant pathogens, and its potential as a probiotic in animal feed has also been evaluated [20].
Essential oils (EOs) are volatile, aromatic compounds synthesized by plants with antimicrobial, antifungal, anti-inflammatory, antioxidant, and antiviral properties [21,22]. Due to these characteristics, several studies have demonstrated their potential as alternatives to antibiotics [23,24]. In particular, essential oils have been shown to improve gut health, thereby enhancing growth performance and immune function in broilers [25,26] Additionally, the supplementation of EOs has been shown to enhance immune capacity and positively affect carcass characteristics in broilers. In this context, future studies should focus on identifying novel strategies to maintain animal health and well-being. Therefore, this study aimed to assess the effects of probiotic and EO supplementation on the growth performance and microbiome composition of broiler chickens.
MATERIALS AND METHODS
All animal procedures were reviewed and approved by the National Institute of Animal Science (NIAS) Animal Use and Care Committee in Korea (NIAS-2021-0508). A total of 300 one-day-old broiler chicks (arbor acres [AA]) were purchased from a commercial farm, sorted by sex (male), and weighed. Subsequently, the chicks were randomized into four experimental groups (1) control: basal diet, (2) Bacillus subtilis (BS; 3 mg/kg) + basal diet, (3) essential oil (EO; 3 mg/kg) + basal diet, and (4) Bacillus velezensis (BV; 3 mg/kg) + basal diet. The oregostim used in the EO test is a natural oregano oil extract with antibacterial, antioxidant, and gut-regenerative properties. It was purchased from SOLTON (Seoul, Korea). Each group was housed in 12 replicate cages (1.5 × 0.9 × 0.5 m) containing 6–7 birds and were reared for 35 days. The chicks received starter, grower, and finisher diets at 1, 2–3, and 4–5 weeks, respectively. Throughout the experiment, all chickens were fed and watered ad libitum. The light period was 24 hours at 40 lux from 0 to 7 d, then 19 h at 20 lux from 8 to 35 d. The experimental environment was controlled at 33 ± 1°C and 50% relative humidity, then reduced by 2°C each week to 24°C. Room temperature was measured daily to ensure consistency. The temperature was manually monitored and controlled. Thermometers and manual ventilation were used. This method is labor-intensive but effective. The ventilation system was activated to remove contaminated air and introduce fresh air. A low-power heater and humidifier were installed. The lighting system was put on a timer. Windows were insulated, and curtains were installed. On days 7 (n = 10) and 35 (n = 12), cecal contents were collected from one bird per replicate selected based on the average body weight for microbiome analysis. The control diet of the mesh type was based on maize and soybean meal (Table 1). Feed consumption and body weight per cage were measured weekly and weight gain and FCR were calculated for mortality. At 7 and 35 days of age, the chickens in the treatment groups were euthanized under carbon dioxide anesthesia. Blood was collected from the wing vein at 35 days. Cecal digesta were frozen in liquid nitrogen and stored at –80°C.
1) Vitamin premix (kg–1): vitamin A (6,250,000 IU), vitamin D3 (1,000,000 IU), vitamin E (15,000 IU), vitamin K3 (1,000 mg), vitamin B1 (500 mg), vitamin B2 (2,500 mg), vitamin B6 (2,500 mg), vitamin B12 (10 mg), pantothenic acid (600 mg), nicotinic acid (15,000 mg), folic acid (500 mg), biotin (35 mg), choline chloride (150,000 mg), iron (20,000 mg), copper (2,500 mg), zinc (25,000 mg), manganese (15,000 mg), iodine (600 mg), cobalt (400 mg), and butylated hydroxytoluene (anti-oxidant, 125,000 mg).
Blood samples were collected from wing vein into ethylenediaminetetraacetic acid (EDTA tubes; BD Vacutainer). Hematological parameters were assessed using a Mindray BC-5300 automated hematology analyzer (Mindray). The concentrations of pro-inflammatory cytokines, including interleukin 1 beta (IL-1β), IL-6, and tumor necrosis factor-alpha (TNF-α), were measured using commercially available chicken enzyme-linked immunosorbent assay kits (EK780087, EK780053, and EK780062; AFG Scientific), following the manufacturer’s protocol.
Deoxyribo nucleic acid (DNA) from cecal samples was extracted using the bead-beating plus column method with a QIAamp DNA kit (Qiagen). The samples were prepared for PacBio instrument sequencing following the single-molecule real-time (SMRT) bell template preparation guide. SMRTbell libraries were constructed by ligating hairpin adapters to double-stranded DNA ends, followed by annealing sequencing primers and polymerase to the library for SMRT sequencing using Sequel II Binding Kit 2.1 and Sequel II DNA Internal Control Complex 1.0 (PacBio). For bacterial 16S rRNA sequencing, primers 27F (5′-AGRGTTYGATYMTGGCTCAG-3’) and 1492R (5′- RGYTACCTTGTTACGACTT-3’) were used to amplify the full-length variable regions of the gene, resulting in a single amplicon of approximately 1,400 base pairs (bp). Polymerase chain reaction (PCR) amplification involved 25 cycles of denaturation at 95°C for 30 s, annealing at 57°C for 30 s, and extension at 72°C for 30 s. After cleanup, eight additional cycles were performed to attach the adapters. Negative controls were included.
The resulting amplicons were sequenced by Macrogen on an Illumina MiSeq platform (Illumina) as previously described. We assessed the size of PCR-enriched fragments using an Agilent Technologies 2100 Bioanalyzer with a DNA 1,000 chip. To ensure accurate data, we optimized cluster density in our prepared libraries using qPCR, following the Illumina guidelines. DADA2 (v. 1.20.0) was used to process Illumina sequences. First, primer sequences were removed and reads were trimmed based on length. Those with > 5 expected errors were removed. The remaining reads were dereplicated and analyzed for sequencing errors using loessErrfun. True sequence variants were inferred, and forward and reverse reads were merged to obtain complete denoised sequences. Chimeric amplicon sequence variants (ASVs) were identified and removed. The remaining reads were annotated using the Silva v. 138.1 database.
Pacific Bioscience data were demultiplexed, and consensus circular sequences (CCS) were generated using the SMRT-Link analysis software (v. 9). A mean of 19 high-fidelity passes were used. Subsequently, the obtained CCS underwent quality checks using the DADA2 R Statistics package (v. 1.20.0). Chimeric ASVs were removed, and the remaining reads were annotated using the naive Bayesian classifier from DADA2 against the Silva138.1 database that comprises the species training set from the Silva138 database. An optimal match is designated only when the discrepancy between the initial and secondary optimal matches exceeds 2%. The taxonomic annotations were subsequently used to generate contingency tables for each taxonomic rank.
Linear discriminant analysis (LDA) effect size (LEfSe) was used to analyze taxon profiles for differential abundance among the four treatment groups (LDA score > 3). Permutational multivariate analysis of variance (PERMANOVA) was used to compare beta diversity analysis and functional genetic profiles among the four treatment groups. Rarefaction curves, richness, and diversity analyses were performed using the minimum number of reads annotated at the ASV. Principal Coordinate Analysis (PCoA) was performed on Bray-Curtis distances to assess similarities between sample types and platforms. Analysis of variance was performed using PERMANOVA on distance matrices from the Vegan R package. Growth performance and blood analysis used two-way and one-way analyses of variance, respectively, with a post-hoc Tukey’s test. Significant differences (p < 0.05) were determined using Prism software (v. 9.5.1).
RESULTS
Fig. 1 illustrates probiotic treatment’s effects on broiler chickens’ growth performance. Initial body weights (40.0 ± 0.05 g) were similar among all dietary treatment groups. BW and ADG showed no significant difference among all groups for 7 days. However, the FCR exhibited an increase (p < 0.05) in all treatment groups compared to the control during the starter period (0 to 7 d). In the growing phase (8 to 21 d), the BW, ADG, and FCR were not significantly different in all diet groups. During the finishing phase (22 to 35 d), the final body weight of the BS group (1,947 ± 29.9 g) was significantly higher than that of the other groups, including the control (1,807 ± 24.4 g), EO (1,860 ± 46.6 g), and BV (1,821 ± 27.6 g) groups (p < 0.05). The BS group also had the highest ADG at the end of the study compared to the other groups (p < 0.05). The BS group had the lowest FCR compared to the other groups (p < 0.05).
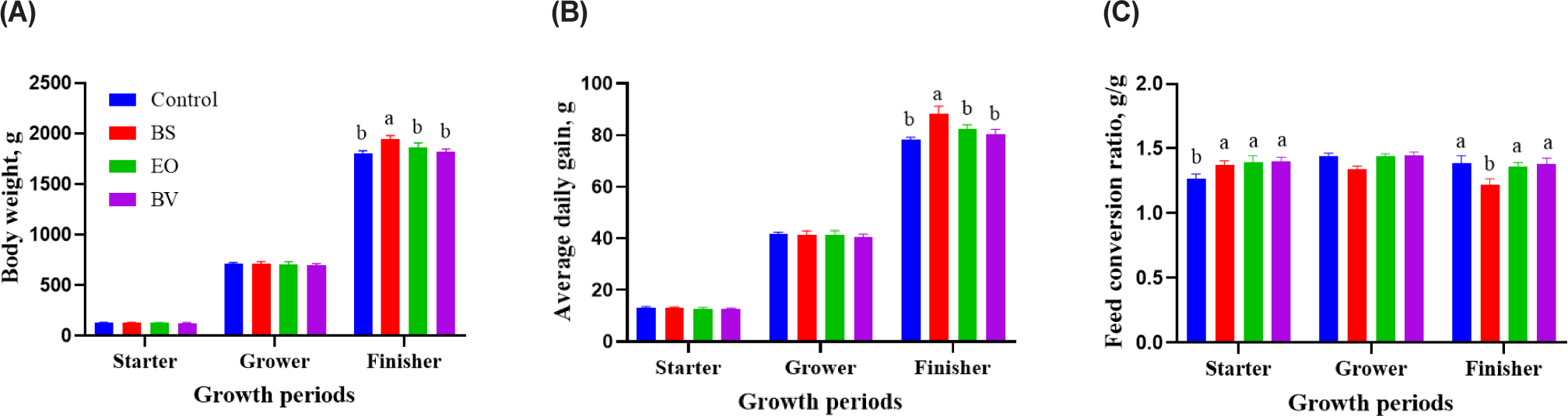
Fig. 2 illustrates the effects of probiotic treatment on the hematological and cytokine parameters of broiler chickens at 35 days. Cytokine parameters (TNF-α, IL-1B, and IL-6) were not significantly different among the probiotic-treated groups (Fig. 2A). Additionally, hematological parameters, including WBC, RBC, hemoglobin, mean corpuscular volume, and platelet counts, exhibited no significant differences among the probiotic-treated groups (Fig. 2B).

Alpha diversity of the cecal microbiota was assessed using the observed and the Chao 1 indices to analyze the effects of age and the four dietary treatments. Additionally, beta diversity was analyzed using PCoA with the Bray–Curtis index to assess differences in the microbial community composition. No significant differences in alpha or beta diversity were observed between the three probiotic treatments. However, the observed and Chao 1 indices were significantly higher in 35-day-old broiler chickens than that in the 7-day-old broiler chickens (p < 0.001; Fig. 3A and 3B). PCoA-based beta diversity analysis using the Bray–Curtis index indicated a distinct separation between the microbial communities of 7-and 35-day-old broiler chickens (p < 0.001; Fig. 3C).
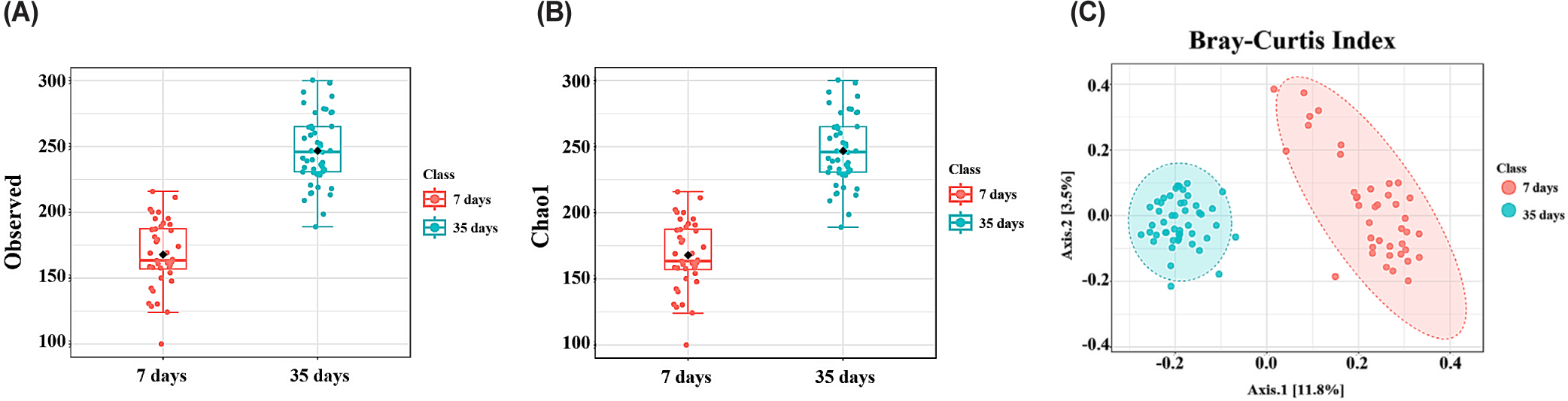
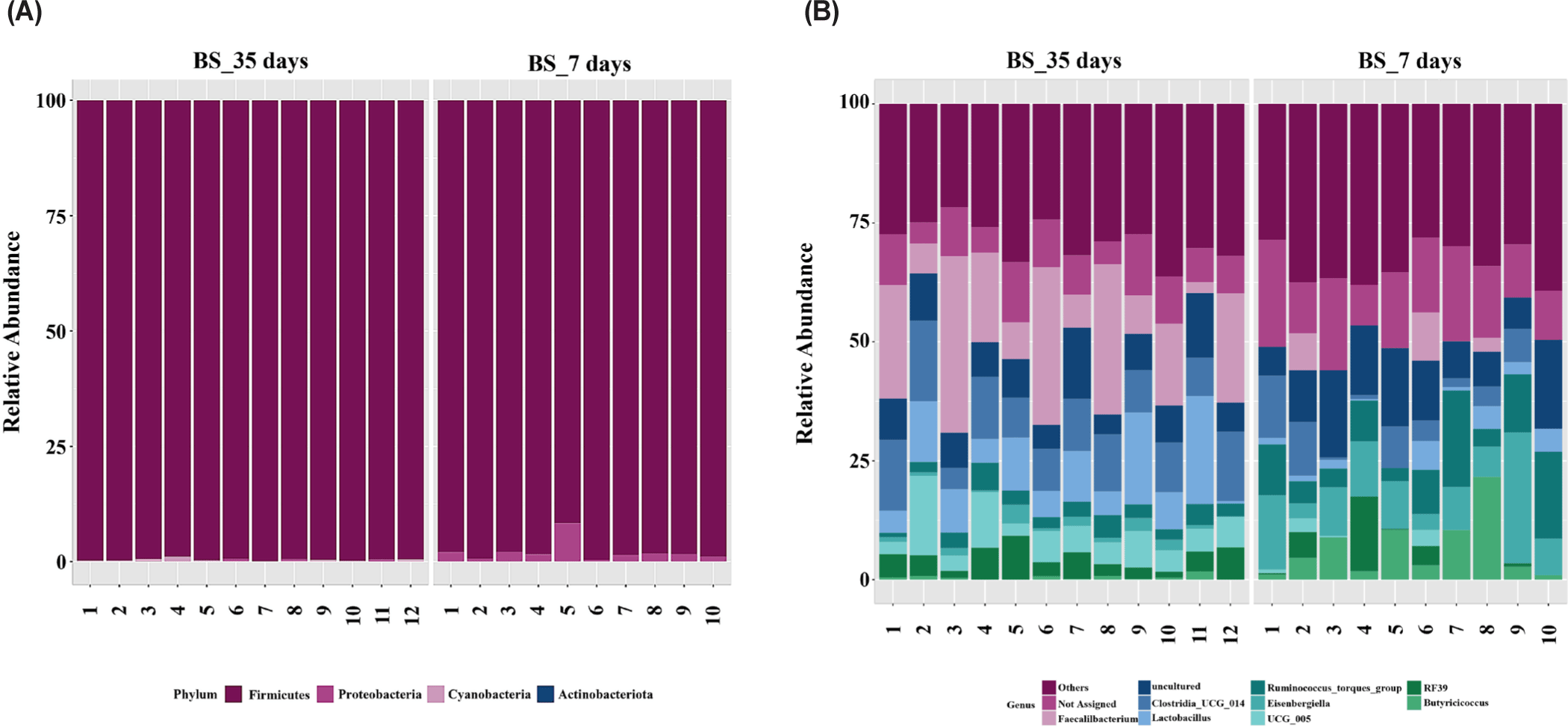
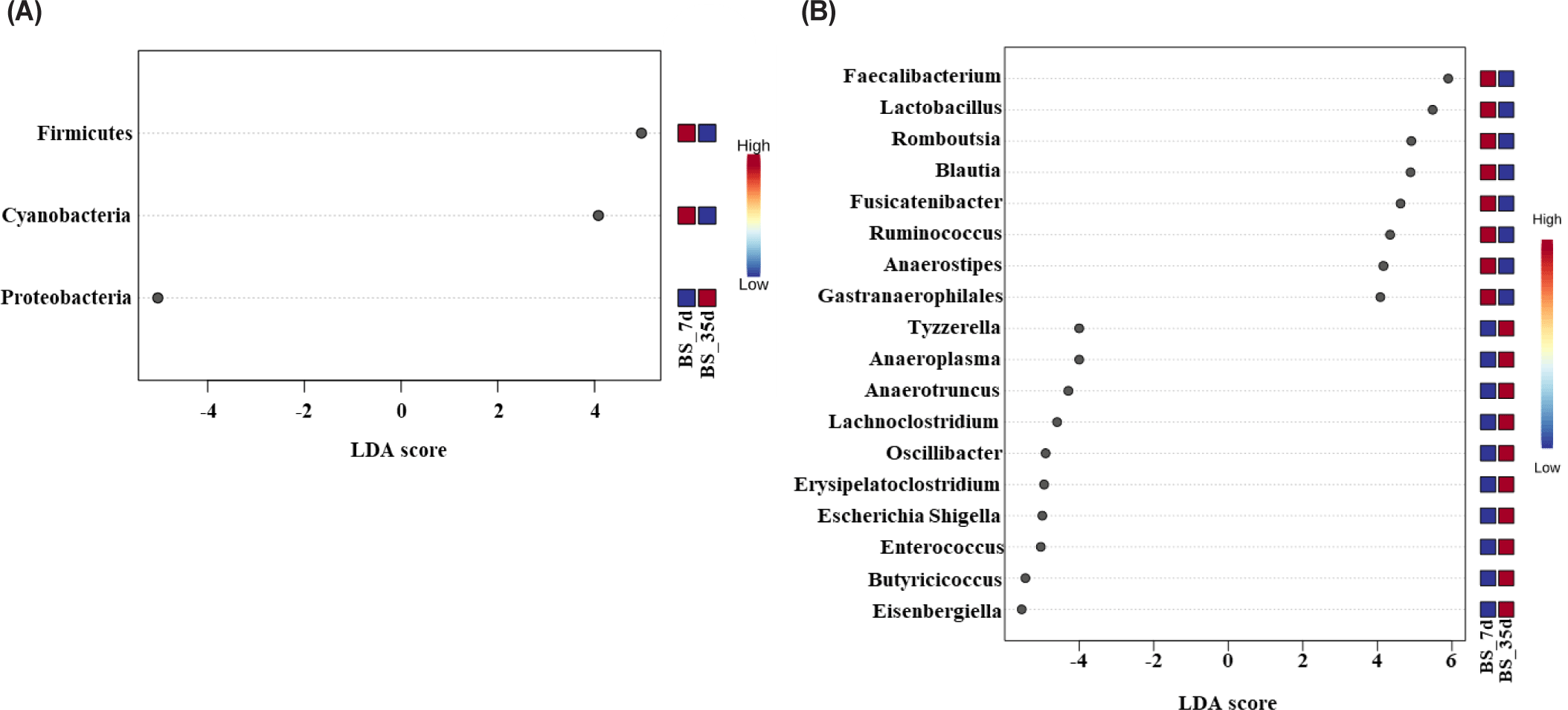
Fig. 4 illustrates taxonomic bar plots for all four dietary treatments in 7- and 35-day-old broiler chickens, demonstrating the mean relative abundance. At the phylum level, Firmicutes were dominant in both 7- (97.07%) and 35-day-old (99.13%) broiler chickens (Fig. 4A). At the genus level, Ruminococcus torques group (X), Eisenbergiella (5.44%), and Butyricicoccus (X) were dominant on day 7, and Faecalibacterium (13.98%), Clostridia_UCG_014 (X), and Lactobacillus (8.04%) were dominant on day 35 (Fig. 4B). We performed LEfSe analysis to identify differentially abundant taxa based on age and dietary treatments (Figs. 5 and 6). At the phylum level, 7-day-old broiler chickens were relatively abundant in Proteobacteria (2.89% vs. 0.40%), whereas 35-day-old broiler chickens were relatively abundant in Firmicutes and Cyanobacteria (Fig. 5A). At the genus level, 7-day-old broiler chickens were relatively abundant in Eisenbergiella, Butyricicoccus, Escherichia-Shigella, Bacillus, Enterococcus, Erysipelatoclostridium, Oscillibacter, Lachnoclostridium, Colidextribacter, Anaerotruncus, Anaeroplasma, and Tyzzerella, whereas 35-day-old broiler chickens were relatively abundant in Faecalibacterium, Lactobacillus, Romboutsia, Blautia, Ruminococcus, Fusicatenibacter, Gastranaerophilales, Roseburia, Anaerostipes, Hydrogenoanaerobacterium, Fournierella, and Anaerofilum (Fig. 5B). On day 7, the BS group was relatively abundant in the genus Eisenbergiella compared to that in the other groups (Fig. 6A). At day 35, the BS group was relatively abundant in Firmicutes and Lachnoclostridium, whereas the BV group was relatively abundant in Hydrogenoanaerobacterium (Figs. 6B and 6C).
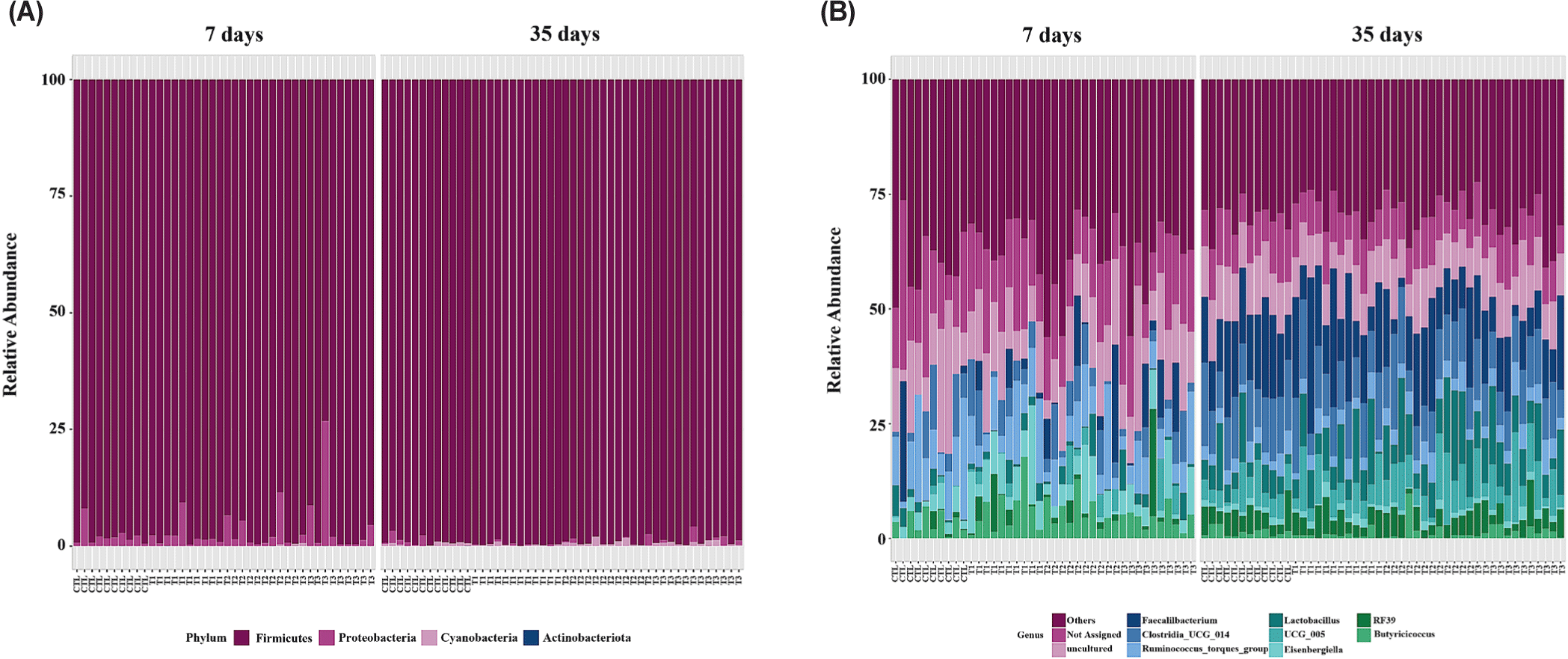

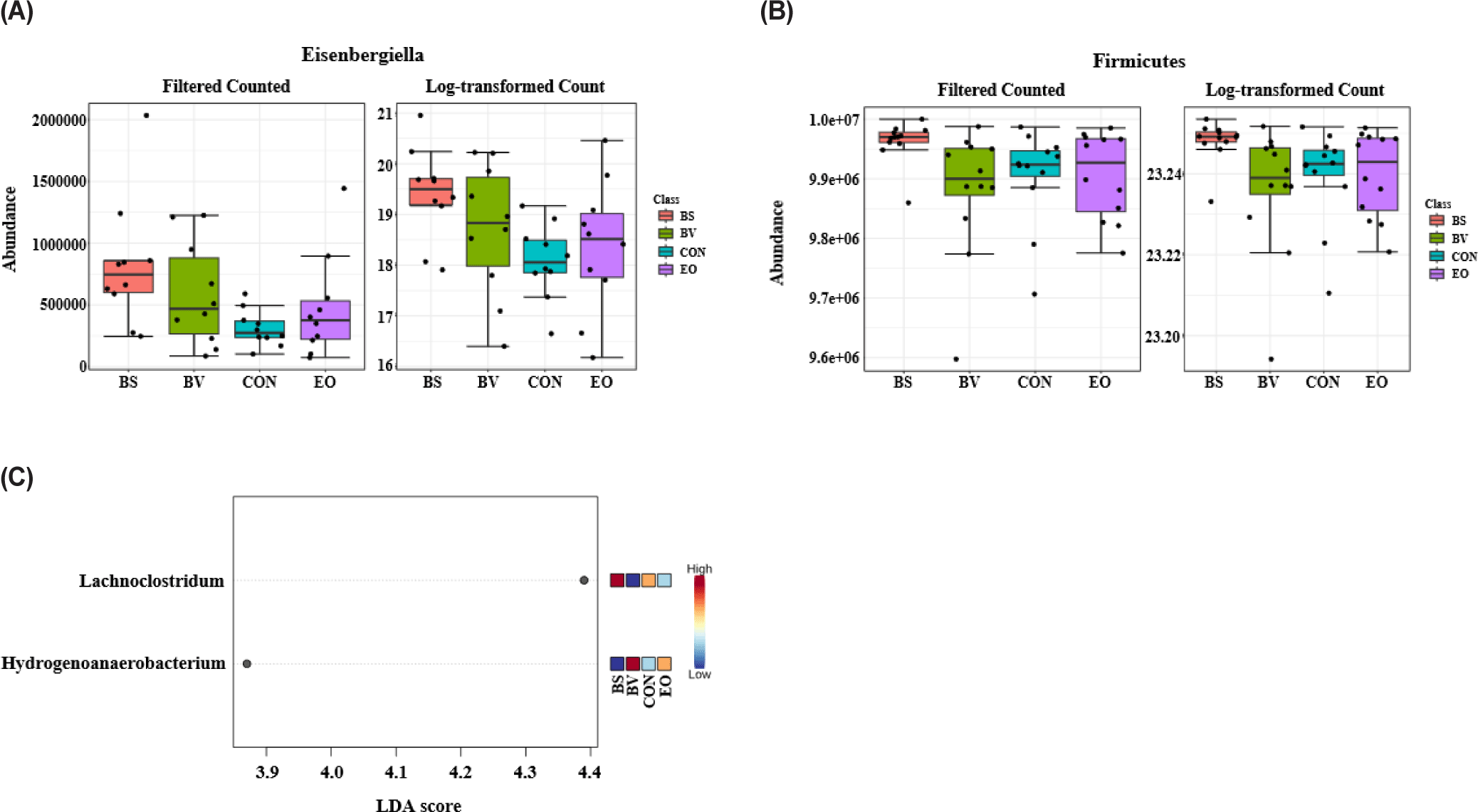
Fig. 7 illustrates the alpha diversity analysis using the observed and Chao1 indices and beta diversity analysis using PCoA with the Bray–Curtis index in the BS group. The observed and Chao1 indices were significantly higher in 35-day-old broiler chickens than that in 7-day-old broiler chickens (p < 0.001; Fig. 7A and 7B). The Bray–Curtis index indicated a distinct separation between the microbial communities of 7- and 35-day-old broiler chickens (p < 0.001; Fig. 7C).
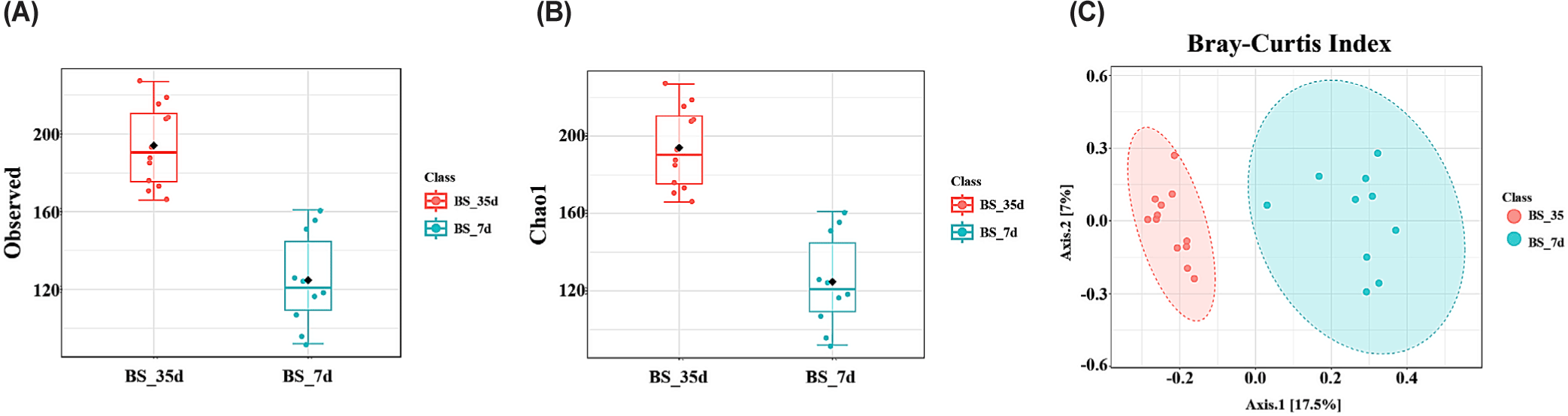
Fig. 8 illustrates the taxonomic bar plots for the BS group in 7- and 35-day-old broiler chickens, demonstrating their mean relative abundance. At the phylum level, Firmicutes were dominant in both 7- (97.75%) and 35-day-old (99.63%) broiler chickens (Fig. 8A). At the genus level, Eisenbergiella (8.24%), Ruminococcus torques group (X), and Butyricicoccus (6.38%) were dominant on day 7, and Faecalibacterium (13.98%), Clostridia_UCG_014 (X), and Lactobacillus (7.84%) were dominant on day 35. In the BS group, LEfSe analysis demonstrated that 7-day-old broiler chickens exhibited a relative abundance of Proteobacteria at the phylum level, whereas 35-day-old broiler chickens exhibited a relative abundance of Firmicutes and Cyanobacteria (Fig. 9A). At the genus level, 7-day-old broiler chickens were relatively abundant in Eisenbergiella, Butyricicoccus, Escherichia–Shigella, Enterococcus, Erysipelatoclostridium, Oscillibacter, Lachnoclostridium, Anaerotruncus, Anaeroplasma, and Tyzzerella, whereas 35-day-old broiler chickens were relatively abundant in Faecalibacterium, Lactobacillus, Romboutsia, Blautia, Fusicatenibacter, Ruminococcus, Gastranaerophilales, and Anaerostipes (Fig. 9B).
DISCUSSION
In this study, no significant differences in growth performance were observed among dietary treatments on day 7 post-hatching, except for FCR. Consistent with our findings, several studies have also failed to demonstrate significant effects of supplementing B. subtilis, essential oils, and Bacillus velezensis on growth performance at day 7 post-hatching [27–29]. Nevertheless, B. subtilis supplementation significantly enhanced weight gain and feed efficiency at the finisher phase, contributing to the improved growth performance in broilers. Molnár et al. [30] observed that providing 7.27 × 10⁹ colony-forming units (CFU)/g of B. subtilis-supplemented diets increased body weight and feed conversion ratio (FCR) in broiler chickens from days 7 to 42 post-hatching. Additionally, Amerah et al. [31] demonstrated that supplementing 1.5 × 10⁸ CFU/kg of B. subtilis improved FCR in broiler chickens at 42 days post-hatch. Consistent with previous studies, our findings highlight the positive effects of B. subtilis on growth performance. This enhancement when using B. subtilis as a probiotic supplement may result from its role in maintaining a beneficial balance in the intestinal microbiome by increasing the population of beneficial bacteria and inhibiting the growth of pathogenic bacteria [32,33].
However, in this study, supplementation with essential oils and Bacillus velezensis did not significantly affect the growth performance of broiler chickens throughout the experimental period. Similarly, Jang et al. [34] supplemented 25 and 50 mg/kg of essential oils in the diet but observed no significant difference in growth performance compared to the basal diet. In contrast, Khattak et al. [35] supplemented 100, 200, 300, 400, and 500 mg/kg of essential oils and found that all treatments improved growth performance compared to the control group after 10 days of age. In our study, 3 mg/kg of essential oil was added to the diet, which may explain the discrepancy in results due to the relatively low concentration. Tsai et al. [36] supplemented broiler chickens with 1.5 × 10⁹ CFU/mL of Bacillus velezensis for 35 days but did not observe a significant improvement in growth performance.
In contrast, Zhu La et al. [29] reported that broilers supplemented with 1 × 10⁹ CFU/mL of Bacillus velezensis for 42 days exhibited increased daily weight gain after 10 days of age and improved final body weight on day 42. Our study’s lack of significant improvement in final body weight may be attributed to the shorter testing period, 7 days less than that of Zhu La et al. [29]. Several studies have also demonstrated that probiotics improve growth performance from 21 days of age and significantly improve body weight at 42 days [27]. Furthermore, this study was conducted under laboratory-scale conditions with a fully controlled environment and feeding regimen, which may have masked the growth-promoting effects of the additives. Jang et al. [34] suggested that dietary antibiotic replacements may fail to induce improvements in growth-related parameters under well-nourished and highly controlled conditions. Thus, the effects of probiotics on growth performance, feed conversion, and productivity in farm animals remain inconsistent under certain conditions, potentially rendering their use economically unviable in specific situations [37]. Consequently, further studies focusing on appropriate dosage and experimental duration are required to validate the efficacy of essential oils and Bacillus velezensis on growth performance compared to previous studies.
In general, adequate probiotic supplementation can enhance the intestinal mucosa, providing a major barrier against pathogens [38]. These effects may contribute to various aspects of the immune response, such as regulating cytokine production [39,40]. In this study, we assessed the cytokine-modulating ability of B. subtilis, essential oils, and Bacillus velezensis supplementation in the diet of broiler chickens and observed no significant enhancement. Additionally, blood cell analysis revealed no effects of dietary treatments. Moreover, these additives do not significantly affect the hematology and cytokine regulation in broiler chickens [41]. In contrast to our results, cytokines, including TNF-α, IL-1β, and IL-6 were significantly upregulated by B. subtilis supplementation [42]. Moreover, essential oil supplementation increased cytokine levels, such as IL-1β and IFN-γ [43]. Furthermore, Bacillus velezensis regulates various cytokines, such as TNF-α, IL-1β, IL-6, and IL-10 in the blood of broiler chickens [38]. However, the studies above and our study differed in dosage, experimental period, and rearing environment (presence of litter, temperature). These differences may explain the controversial results in blood biochemical parameters [44,45]. Furthermore, in well-controlled experimental settings, reduced corticosterone levels in broilers can enhance humoral immunity by promoting the production of anti-inflammatory cytokines and immunoglobulins [46]. This may have mitigated the additive’s effects on immunological parameters in this study. Therefore, further studies are required to clarify the efficacy of these additives on broiler chickens’ immune regulation ability.
The intestinal microbiota of broiler chickens develops throughout the gastrointestinal tract and plays a crucial role in maintaining health while also influencing productivity [47]. These beneficial microbes support gut health by aiding feed digestion, nutrient absorption, immune system development, and pathogenic bacterial growth inhibition [48,49]. In this study, no significant differences were observed in alpha or beta diversity in the cecal content between the control and treatment groups. Similar to our findings, previous studies have shown that supplementation with B. subtilis, essential oils, and Bacillus velezensis did not significantly affect alpha diversity or beta diversity in broiler chickens [33,50,51]. The dynamic diversity of the gut microbiome is known to be influenced by diet and age. Still, age has been shown to have a more significant impact than feed additive supplementation [33,52]. Indeed, the microbial diversity and community composition varied significantly between 7- and 35-day-old broiler chickens across all dietary treatments. Similar patterns were observed in the BS group. Microbial diversity increases with age and forms distinct clusters in the microbiota [48,53]. Our results demonstrated that Firmicutes was the most dominant phylum across all treatments, aligning with previous studies demonstrating that Firmicutes generally comprise the majority of the cecal microbiota for short-chain fatty acid production in broiler chickens [54,55]. At the genus level, the Ruminococcustorques group was dominant in 7-day-old broiler chickens, whereas Faecalibacterium was most abundant in 35-day-old broiler chickens. Similarly, at the genus level, the Ruminococcustorques group was dominant in 21-day-old broiler chickens, with Faecalibacterium becoming more prevalent in 39-day-old broiler chickens [56]. LEfSe analysis demonstrated differentially abundant taxa between the age groups. Host age affects the diversity and stability of microbiota. In broiler chickens, the gut microbiota is dynamic during the first few weeks of life, transitioning to a mature and stable state after 21 days of age [14,57]. In this study, Proteobacteria were relatively abundant in the early stages, and the abundance of Firmicutes increased with age, aligning with the previous studies [52,57,58]. At the genus level, Eisenbergiella, Butyricicoccus, and Escherichia–Shigella were relatively abundant at 7 days of age. Eisenbergiella and Butyricicoccus are significant producers of butyric acid, an energy source for fast-growing broiler chickens [57,59]. Additionally, Escherichia–Shigella is a rapidly colonizing microbial group that can dominate the gut of early broiler chickens [60]. However, Escherichia–Shigella can cause diseases, such as colibacillosis and shigellosis, highlighting the significance of an early hatching environment in preventing harmful microorganisms [60]. Faecalibacterium, which was most abundant at 35 days of age in our study, is known to dominate the mature microbiota after 21 days [55]. Additionally, this study included two feed transitions: from starter to grower feed, and subsequently to finisher feed. These dietary alterations have likely contributed to age-related shifts in the intestinal microbiota [56]. In summary, these findings indicate that the age of broiler chickens significantly affects the composition and diversity of their intestinal microbiota.
Our findings demonstrated that B. subtilis supplementation increased the abundance of beneficial microorganisms in broiler chickens compared to other dietary treatments. On day 7, B. subtilis supplementation resulted in a relative dominance of Eisenbergiella. The abundance of Eisenbergiella was lower in the non-B. subtilis-treated group compared to that in the B. subtilis-treated group. Eisenbergiella plays a crucial role in producing butyric acid, which is the preferred energy source for intestinal epithelial cells [59]. Additionally, increasing the abundance of Eisenbergiella can improve feed efficiency and reduce FCR in broilers [61]. By day 35, B. subtilis supplementation, compared to other dietary treatments, increased the abundance of Firmicutes and Lachnoclostridium at the phylum and genus levels, respectively. Firmicutes are essential for growth that break down indigestible polysaccharides, facilitating nutrient absorption [62,63]. In this regard, the abundance of Firmicutes was shown to improve ADG and reduce FCR in broiler [64]. Lachnoclostridium can ferment dietary fiber by breaking down various indigestible polysaccharides and producing butyric and acetic acids [65]. Moreover, a previous study showed that the diet group enriched with Lachnoclostridium had improved ADG [64]. B. subtilis is known for maintaining the intestinal microbial ecosystem by enhancing mucosal immunity and regulating intestinal commensal microorganisms [66]. This may promote the growth performance broilers and may provide the basis for our study results showing improved body weight and FCR in BS group broilers. Therefore, our results demonstrate that B. subtilis supplementation modulates commensal microbiota and supports the findings of previous studies.
Our study assessed the effects of three dietary treatments B. subtilis, essential oils, and Bacillus velezensis on the growth performance, cytokine levels, and gut microbiome composition of broiler chickens over five-weeks. We observed that B. subtilis supplementation enhanced the growth performance of broiler chickens and increased the abundance of beneficial microorganisms throughout their life cycle. This highlights its potential as a promising probiotic to enhance broiler health. Additionally, we observed age-related alterations in the gut microbiome composition, indicating the significance of growth and health management throughout the broiler life cycle. However, the study did not reveal any significant effects of the three dietary treatments on the immune regulatory ability of broiler chickens, which may be attributed to various complex factors. Therefore, further studies—considering various factors—are required to fully understand the effects of probiotics on the immune capacity of broiler chickens.

